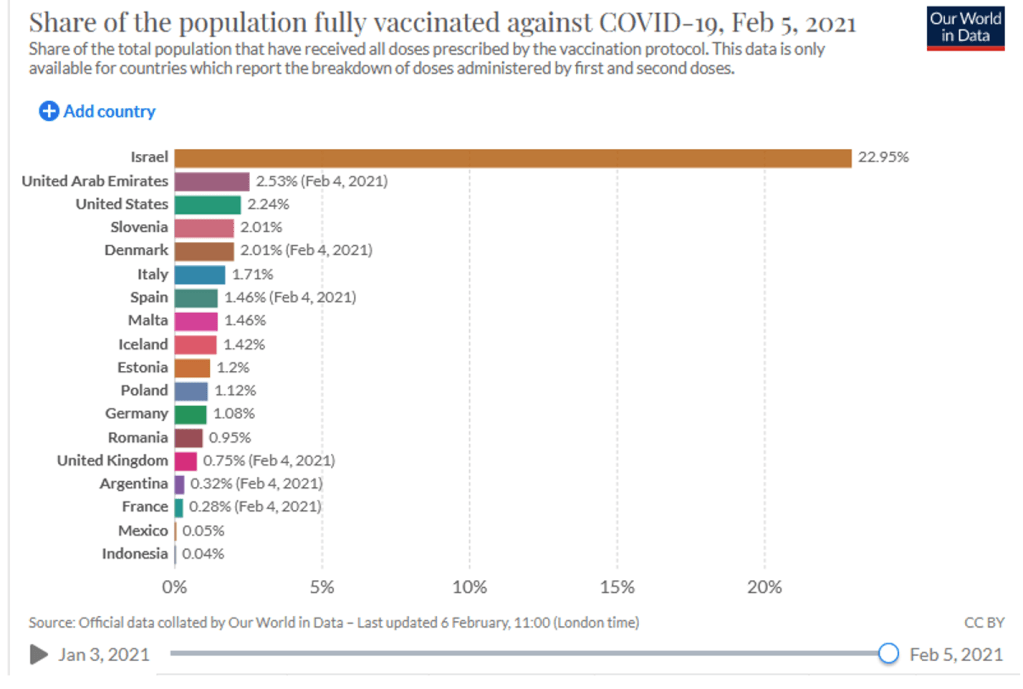
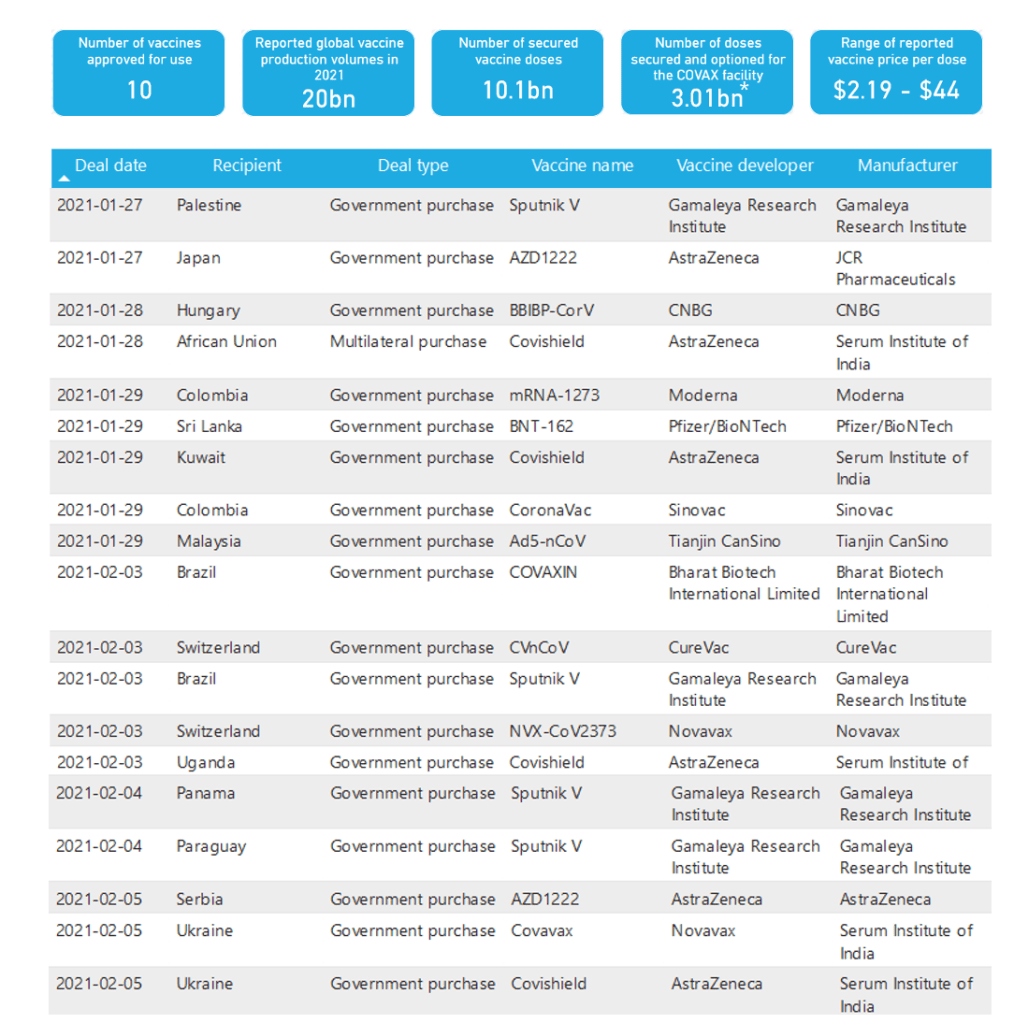
Organization Announcements
Paul G. Allen Frontiers Group [to 6 Feb 2021]
https://alleninstitute.org/what-we-do/frontiers-group/news-press/
News
Press Release
New Allen Distinguished Investigators will tackle unanswered questions about metabolism and the immune system
February 3, 2021
Awards announced today by The Paul G. Allen Frontiers Group will fund research in health, disease, and technology development all centered on the emerging field of immunometabolism
BARDA – U.S. Department of HHS [to 6 Feb 2021]
https://www.phe.gov/about/barda/Pages/default.aspx
BARDA News
No new digest content identified.
BMGF – Gates Foundation [to 6 Feb 2021]
http://www.gatesfoundation.org/Media-Center/Press-Releases
Press Releases and Statements
No new digest content identified.
Bill & Melinda Gates Medical Research Institute [to 6 Feb 2021]
https://www.gatesmri.org/
The Bill & Melinda Gates Medical Research Institute is a non-profit biotech organization. Our mission is to develop products to fight malaria, tuberculosis, and diarrheal diseases—three major causes of mortality, poverty, and inequality in developing countries. The world has unprecedented scientific tools at its disposal; now is the time to use them to save the lives of the world’s poorest people
No new digest content identified.
CARB-X [to 6 Feb 2021]
https://carb-x.org/
News
02.03.2021 |
CARB-X is funding French biotech Mutabilis to develop a new class of antibacterials to treat infections caused by Carbapenem-resistant Enterobacterales (CRE) bacteria
CARB-X is awarding Mutabilis, a biopharmaceutical firm based in Romainville, France, up to US$6.4 million to develop a new drug to treat infections caused by Carbapenem-resistant Enterobacterales (CRE) bacteria.
02.01.2021 |
CARB-X is funding the development of Avails Medical’s rapid antibiotic susceptibility testing technology to identify the most effective antibiotic therapy for serious blood infections
CARB-X is funding Avails Medical, a Menlo Park, CA, USA, diagnostics company, to develop an electronic antibiotic susceptibility testing (AST) device to speed up health care providers’ ability to determine the most effective antibiotic treatment for serious and life-threatening blood infections.
Center for Vaccine Ethics and Policy – GE2P2 Global Foundation [to 6 Feb 2021]
https://centerforvaccineethicsandpolicy.net/
News/Analysis/Statements
No new digest content identified.
CEPI – Coalition for Epidemic Preparedness Innovations [to 6 Feb 2021]
http://cepi.net/
Latest News
03 Feb 2021
COVAX publishes first interim distribution forecast
The forecast provides interim information on early projected availability of doses in first half of 2021 to COVAX Facility participants.
[See COVID above for detail]
01 Feb 2021
A leap forward in vaccine technology
By building on the lessons learned in 2020, it should be possible in the long term to compress vaccine development timelines still further
01 Feb 2021
Preparing for the next “Disease X”
Why the world needs to prepare for the next pandemic
01 Feb 2021
CEPI and Dynavax collaborate to secure adjuvant for COVID-19 vaccines in 2021
Agreement supports the supply of Dynavax’s CpG 1018 vaccine adjuvant in 2021 for CEPI-funded COVID-19 vaccine development programmes
Duke Global Health Innovation Center [to 6 Feb 2021]
https://dukeghic.org/
Launch and Scale Speedometer
No new analysis identified.
EDCTP [to 6 Feb 2021]
http://www.edctp.org/
The European & Developing Countries Clinical Trials Partnership (EDCTP) aims to accelerate the development of new or improved drugs, vaccines, microbicides and diagnostics against HIV/AIDS, tuberculosis and malaria as well as other poverty-related and neglected infectious diseases in sub-Saharan Africa, with a focus on phase II and III clinical trials
30 January 2021
World NTD Day 2021 – a global call to End the Neglect
Emory Vaccine Center [to 6 Feb 2021]
http://www.vaccines.emory.edu/
Vaccine Center News
No new digest content identified.
European Commission [to 6 Feb 2021]
http://europa.eu/rapid/search-result.htm?query=18&locale=en&page=1
No new digest content identified.
European Medicines Agency [to 6 Feb 2021]
http://www.ema.europa.eu/ema/
News & Press Releases
News: EMA reviewing data on monoclonal antibody use for COVID-19
Last updated: 04/02/2021
News: EMA COVID-19 assessments ‘OPEN’ to non-EU regulators
Last updated: 04/02/2021
News: EMA starts rolling review of Novavax’s COVID-19 vaccine (NVX-CoV2373)
Last updated: 03/02/2021
EMA’s human medicines committee (CHMP) has started a rolling review of NVX-CoV2373, a COVID19 vaccine being developed by Novavax CZ AS (a subsidiary of Novavax, Inc.).
The CHMP’s decision to start the rolling review is based on preliminary results from laboratory studies (non-clinical data) and early clinical studies in adults. These studies suggest that the vaccine triggers the production of antibodies and immune cells that target SARS-CoV-2, the virus that causes COVID19.
The company is currently conducting trials in people to assess its safety, immunogenicity (how well it triggers a response against the virus) and its effectiveness against COVID-19. EMA will evaluate data from these and other clinical trials as they become available.
The rolling review will continue until enough evidence is available for a formal marketing authorisation application…
News: EMA starts rolling review of REGN-COV2 antibody combination (casirivimab / imdevimab)
Last updated: 01/02/2021
News: Meeting highlights from the Committee for Medicinal Products for Human Use (CHMP) 25-29 January 2021 (updated)
CHMP, Last updated: 01/02/2021
News: EMA recommends COVID-19 Vaccine AstraZeneca for authorisation in the EU (updated)
Last updated: 29/01/2021
Update: COVID-19 Vaccine AstraZeneca is now authorised across the EU. This follows the granting of a conditional marketing authorisation by the European Commission on 29 January 2021.
European Vaccine Initiative [to 6 Feb 2021]
http://www.euvaccine.eu/
Latest News
EVI
Feb 4, 2021
VAC2VAC at the scientific conference “Towards replacement of animals for scientific purposes”
FDA [to 6 Feb 2021]
https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/default.htm
Press Announcements /Selected Details
February 5, 2021 – Coronavirus (COVID-19) Update: February 5, 2021
:: On Feb. 4, the FDA revised the Letter of Authorization for COVID-19 convalescent plasma to limit the authorization to the use of high titer COVID-19 convalescent plasma for the treatment of hospitalized patients with COVID-19 early in the disease course and to those hospitalized patients who have impaired humoral immunity and cannot produce an adequate antibody response. Data indicates that plasma with low levels of antibodies may not be effective in treating COVID-19. The revision was based upon data from new clinical trials analyzed or reported since the original EUA was issued in August 2020.
FDA – COVID-19 Vaccines [to 6 Feb 2021]
www.fda.gov/covid19vaccines
News and Updates; Upcoming Events
02/05/2021
Coronavirus (COVID-19) Update
The FDA has scheduled a meeting of its Vaccines and Related Biological Products Advisory Committee; alerted health care professionals and compounders of potential risks associated with compounding remdesivir drug products; revised the Letter of Authorization for COVID-19 convalescent plasma; and issued a statement that FDA continues important work to support medical product development to address new virus variants.
02/04/2021
Coronavirus (COVID-19) Update: FDA Announces Advisory Committee Meeting to Discuss Janssen Biotech Inc.’s COVID-19 Vaccine Candidate
The FDA has scheduled a meeting of its Vaccines and Related Biological Products Advisory Committee (VRBPAC) on Feb. 26, 2021, to discuss the request for emergency use authorization (EUA) for a COVID-19 vaccine from Janssen Biotech Inc.
02/04/2021
Coronavirus (COVID-19) Update: FDA Continues Important Work to Support Medical Product Development to Address New Virus Variants
As the public health agency responsible for regulating medical products, we must ensure that health care providers have the most up-to-date diagnostics, treatments and vaccines in their toolbox to fight this pandemic.
Fondation Merieux [to 6 Feb 2021]
http://www.fondation-merieux.org/
News, Events
No new digest content identified.
Gavi [to 6 Feb 2021]
https://www.gavi.org/
News Releases
COVAX publishes first interim distribution forecast
3 February 2021
[See COVID above for detail]
GHIT Fund [to 6 Feb 2021]
https://www.ghitfund.org/newsroom/press
GHIT was set up in 212 with the aim of developing new tools to tackle infectious diseases that
Press Releases
No new digest content identified.
Global Fund [to 6 Feb 2021]
https://www.theglobalfund.org/en/news/
News
Mozambique and Global Fund Launch New Grants to Fight HIV, TB and Malaria and Strengthen Systems for Health
05 February 2021
The Global Fund, the Government of Mozambique and health partners in Mozambique today launched the implementation of six new grants to fight HIV, TB and malaria and build resilient and sustainable systems for health
News
Global Fund Engages Partners to Develop New Strategy
02 February 2021
More than 300 representatives from across the world convened virtually today to kick off the Partnership Forums, a series of consultations to help shape the next multi-year Global Fund strategy.
Global Research Collaboration for Infectious Disease Preparedness [GloPID-R] [to 6 Feb 2021]
https://www.glopid-r.org/news/
News
No new digest content identified.
Hilleman Laboratories [to 6 Feb 2021]
http://www.hillemanlabs.org/
No new digest content identified.
Human Vaccines Project [to 6 Feb 2021]
http://www.humanvaccinesproject.org/media/press-releases/
Press Releases
Global COVI D Lab Meeting
February 25th speaker: Bali Pulendran, M.D., Ph.D., Stanford University
Feb 25, 2021
Global COVID Lab Meeting
Feb 11, 2021 speaker: Christos Kyratsous, Ph.D., Regeneron Pharmaceuticals
Special Issue 5: Two More Vaccines Found Effective, but Less So Against New Variants
Feb 01, 2021
By Kristen Jill Abboud, Science Writer/Editor
IAVI [to 6 Feb 2021]
https://www.iavi.org/newsroom
PRESS RELEASES/FEATURES
February 3, 2021
First-in-human clinical trial confirms novel HIV vaccine approach developed by IAVI and Scripps Research
The experimental vaccine primed the immune system as the first stage in the production of broadly neutralizing antibodies.
NEW YORK and LA JOLLA, CA — FEBRUARY 3, 2021 — A Phase I clinical trial testing a novel vaccine approach to prevent HIV has produced promising results, IAVI and Scripps Research announced today. The vaccine showed success in stimulating production of rare immune cells needed to start the process of generating antibodies against the fast-mutating virus; the targeted response was detected in 97% of participants who received the vaccine…
February 3, 2021
Evolving access pathways for long-acting HIV prevention products
February 2, 2021
IAVI Remembers Philip Russell, Vaccine Scientist, Global Health Leader
January 25, 2021
Merck and IAVI Discontinue Development of COVID-19 Vaccine Candidate V590
International Coalition of Medicines Regulatory Authorities [ICMRA]
http://www.icmra.info/drupal/en/news
Selected Statements, Press Releases, Research
No new digest content identified.
International Generic and Biosimilar Medicines Association [IGBA]
https://www.igbamedicines.org/
News
Press Releases/Announcements
No new digest content identified.
IFFIm
http://www.iffim.org/
Press Releases/Announcements
Italy reaffirms support of Gavi and CEPI with €5 million grant to IFFIm
26 Jan 2021
Italy’s new grant will provide immediate funding for CEPI’s COVID-19 vaccine research and development programme.
IFRC [to 6 Feb 2021]
http://media.ifrc.org/ifrc/news/press-releases/
Selected Press Releases, Announcements
COVID-19: Warning of “deadly consequences” of vaccine inequality, IFRC launches plan to help vaccinate 500 million people
Geneva, 4 February 2021 – The overwhelming majority of COVID-19 vaccine doses administered so far have been delivered in high-income countries, according to analysis by the International Federation of Red Cross and Red Crescent Societies (IFRC). Nearly …
[See COVID above for detail]
Lebanon, Middle East and North Africa
Six months after Beirut Blast: Deteriorating humanitarian situation needs global solidarity
The Lebanese Red Cross (LRC) and the International Federation of Red Cross and Red Crescent Societies (IFRC) appeal for continued global solidarity with the Lebanese people who are suffering from a multi-layered humanitarian crisis. Six months after th …
4 February 2021
Institut Pasteur [to 6 Feb 2021]
https://www.pasteur.fr/en/press-area
No new digest content identified.
IRC International Rescue Committee [to 6 Feb 2021]
http://www.rescue.org/press-release-index
Media highlights [Selected]
Press Release
IRC applauds Biden’s Refugee Admissions Executive Order, helping restore America’s global humanitarian leadership
February 4, 2021
IVAC [to 6 Feb 2021]
https://www.jhsph.edu/research/centers-and-institutes/ivac/index.html
Updates; Events
No new digest content identified.
IVI [to 6 Feb 2021]
http://www.ivi.int/
Selected IVI News, Announcements, Events
No new digest content identified.
JEE Alliance [to 6 Feb 2021]
https://www.jeealliance.org/
Selected News and Events
No new digest content identified.
Johns Hopkins Center for Health Security [to 6 Feb 2021]
https://www.centerforhealthsecurity.org/news/center-news/
Center News
No new digest content identified.
MSF/Médecins Sans Frontières [to 6 Feb 2021]
http://www.msf.org/
Latest [Selected Announcements]
Access to medicines
MSF urges wealthy countries not to block COVID-19 patent waiver
Press Release 3 Feb 2021
Ahead of the next round of talks at the World Trade Organization (WTO) to discuss a proposal by South Africa and India to waive monopolies on COVID-19 medical tools during the pandemic, Médecins Sans Frontières (MSF) called on the wealthy countries opposing the proposal not to block it and ruin its lifesaving potential for billions of people in the rest of the world…
Coronavirus COVID-19 pandemic
Ravaged by new strain, southern Africa must get COVID-19 vaccines
Press Release 3 Feb 2021
:: An infectious new strain of COVID-19 is spreading quickly through southern African countries, leaving health systems struggling to cope.
:: Cases in Mozambique, Eswatini and Malawi are currently at levels exponentially higher than during the first wave of the pandemic.
:: With no COVID-19 vaccines yet available in these countries – partly due to hoarding from wealthy ones – frontline healthcare workers are exposed and are getting sick.
:: MSF is urging wealthy country governments and pharma companies to prioritise vaccines for the healthcare staff in these and other lower-income countries.
National Vaccine Program Office – U.S. HHS [to 6 Feb 2021]
https://www.hhs.gov/vaccines/about/index.html
Upcoming Meetings/Latest Updates
No new digest content identified.
NIH [to 6 Feb 2021]
http://www.nih.gov/news-events/news-releases
News Releases
Fecal microbiota transplants help patients with advanced melanoma respond to immunotherapy
February 4, 2021 — Study is one of the first to demonstrate in patients that altering the composition of the gut microbiome can improve the response to immunotherapy.
Intranasal influenza vaccine spurs strong immune response in Phase 1 study
February 3, 2021 — The vaccine platform could be highly adaptable for use against other viruses.
PATH [to 6 Feb 2021]
https://www.path.org/media-center/
Press Release
No new digest content identified.
Sabin Vaccine Institute [to 6 Feb 2021]
http://www.sabin.org/updates/pressreleases
Statements and Press Releases
No new digest content identified.
UNAIDS [to 6 Feb 2021]
http://www.unaids.org/en
Selected Press Releases/Reports/Statements
1 February 2021
We are off track to meet the 2020 targets on the number of voluntary medical male circumcisions
UNHCR Office of the United Nations High Commissioner for Refugees [to 6 Feb 2021]
http://www.unhcr.org/en-us/media-centre.htmlS
Selected Announcements
UNHCR and IOM call for improved safeguards for the displaced on the frontlines of climate emergency
4 Feb 2021
The European Union, UNICEF and UNHCR join efforts to protect children on the move in Central America, Mexico and Southern Africa 2 Feb 2021
UNICEF [to 6 Feb 2021]
https://www.unicef.org/media/press-releases
Selected Press releases, Statements
Statement 02/03/2021
UNICEF Executive Director Henrietta Fore’s remarks at briefing on COVAX publishing interim distribution forecast
As delivered
[See COVID above for detail]
Unitaid [to 6 Feb 2021]
https://unitaid.org/
Featured News
05 February 2021
Unitaid’s response to COVID-19 praised in UK government review
03 February 2021
New patient-friendly tuberculosis preventive treatment to be rolled out in five high-burden TB countries at affordable price
:: Fixed-dose combination treatments reduce the pill burden from nine to three pills a week for adults and prevent TB in those at highest risk of developing the disease
:: Ethiopia, Ghana, Kenya, Mozambique, and Zimbabwe will be the first countries out of a total of 12 to provide the new regimen at a US$15 price thanks to funding from Unitaid, PEPFAR and the Global Fund to Fight AIDS, Tuberculosis and Malaria
02 February 2021
Unitaid statement regarding Ivermectin as a potential COVID-19 treatment
Ivermectin, as well as other repurposed products, has been suggested as a potential treatment for COVID-19 based on preliminary promising evidence – further data is needed to support a definitive recommendation either for or against its use for COVID-19.
Unitaid has collaborated with the University of Liverpool to conduct the preliminary desk analysis of existing trials evaluating ivermectin in different countries of the world, in order to facilitate a review by WHO.
The preliminary analysis has incorporated data from randomised clinical studies that have been completed in Bangladesh, Egypt, Iran, India, Iraq, Lebanon, Pakistan, Turkey, Nigeria, Argentina, Mexico, and Spain.
In the coming weeks, results from additional trials in other countries are expected, and an in-depth analysis will be conducted by WHO to determine next steps, including the potential need for further targeted clinical studies.
Vaccination Acceptance Research Network (VARN) [to 6 Feb 2021]
https://vaccineacceptance.org/news.html#header1-2r
Announcements
No new digest content identified.
Vaccine Confidence Project [to 6 Feb 2021]
http://www.vaccineconfidence.org/
News, Research and Reports
No new digest content identified.
Vaccine Education Center – Children’s Hospital of Philadelphia [to 6 Feb 2021]
http://www.chop.edu/centers-programs/vaccine-education-center
News
No new digest content identified.
Wellcome Trust [to 6 Feb 2021]
https://wellcome.ac.uk/news
News
Opinion
Four reasons why we need multiple vaccines for Covid-19
Having a range of Covid-19 vaccines available for people to use around the world will be essential to bringing the pandemic under control. Here’s why.
Charlie Weller, Head of Vaccines Programme
:: Going into 2021, a handful of vaccines to protect against Covid-19 were already approved for use in parts of the world.
:: So why, with highly effective vaccines already in use, is it important to keep working on and investing in the hundreds of Covid-19 vaccines still in development?
The Wistar Institute [to 6 Feb 2021]
https://www.wistar.org/news/press-releases
Press Releases
No new digest content identified.
WFPHA: World Federation of Public Health Associations [to 6 Feb 2021]
https://www.wfpha.org/
Latest News
Which Vaccines and Why: The Evidence and the Deals
Feb 5, 2021
Operation Vaccination: An Analysis of Global COVID-19 Vaccine Rollout Strategies
Jan 29, 2021 | News
Operation Vaccination: An Analysis of Global COVID-19 Vaccine Rollout Strategies
The COVID-19 pandemic is the greatest public health crisis of our lifetimes. Mass immunization is the best chance we have of conquering the disease, but it presents a fiercely complex challenge for governments, healthcare systems and populations.
This weekly post will explore the policy, logistics, obstacles and controversies involved in procuring and distributing vaccines against COVID-19. Under investigation are 9 countries across 5 continents (Brazil, India, Indonesia, Ireland, Israel, Nigeria, Taiwan, the UK and the US), as well as COVAX, the global vaccine sharing initiative.
These articles are a means for the WFPHA to keep readers up to date with how the COVID-19 vaccine race is developing across the world. Each week will cover a different theme as it applies to each of the countries being covered:
COVAX
Brazil
India
IDN
IRL
Israel
NGA
TW
UK
US
World Organisation for Animal Health (OIE) [to 6 Feb 2021]
https://www.oie.int/en/for-the-media/press-releases/2021/
Press Releases
No new digest content identified.
::::::
ARM [Alliance for Regenerative Medicine] [to 6 Feb 2021]
Press Releases – Alliance for Regenerative Medicine (alliancerm.org)
Press Releases
Alliance for Regenerative Medicine Responds to EU Beating Cancer Plan
February 3, 2021
WASHINGTON, DC and BRUSSELS, BELGIUM
The Alliance for Regenerative Medicine (ARM) – the ‘global voice’ of the regenerative medicine and advanced therapies sector – applauds the EU Beating Cancer Plan’s identification of Advanced Therapy Medicinal Products (ATMPs) as part of a comprehensive cancer solution. ATMPs include cell and gene therapies that have already revolutionized the treatment of some aggressive forms of blood cancers and hold great promise for addressing other types of cancer.
BIO [to 6 Feb 2021]
https://www.bio.org/press-releases
Press Releases
No new digest content identified.
DCVMN – Developing Country Vaccine Manufacturers Network [to 6 Feb 2021]
http://www.dcvmn.org/
News; Upcoming events
No new digest content identified.
ICBA – International Council of Biotechnology Associations [to 6 Feb 2021]
https://internationalbiotech.org/news/
News
No new digest content identified.
IFPMA [to 6 Feb 2021]
http://www.ifpma.org/resources/news-releases/
Selected Press Releases, Statements, Publications
Patients, product development partnerships and private health sector representatives call for the rapid ratification of the African Medicines Agency Treaty to speed up timelines to vaccines and medicines approval
London / Geneva, 5 February 2021 – On the eve of the 34th African Union Summit, two years after the Treaty to establish an African Medicines Agency was agreed, over 40 leading organizations who represent hundreds of thousands of patients, product development partnerships, youth groups, researchers and industry leaders, are urging Heads of State and Government to ratify the Treaty as a matter of priority. Delaying the establishment of an African Medicines Agency (AMA) undermines the timely access to effective, quality therapies, and vaccines for all patients across Africa.
The COVID-19 pandemic has highlighted the importance of regulatory harmonisation in the context of public health emergencies and the urgent need for a competent continent-wide regulatory authority to approve and monitor vaccines, repurposed medicines, innovative medicines and health technologies, in a timely manner.
Two years after African Union Heads of State and Government endorsed the treaty establishing the AMA, legal commitment from more countries remains to be secured for the continent-wide regulatory agency to become a reality. The Treaty has so far been signed by 19 countries1, and 8 out of the necessary 15 have already ratified it 2.
The agency would follow a similar mandate to other global medicines regulatory agencies, such as the European Medicines Agency and the U.S. Food and Drug Administration. The AMA would provide an enabling environment for research and development, country participation in clinical research and other scientific development activities, as well as open more opportunities to boost local manufacturing capacities. Furthermore, the AMA would help effectively manage the prevalence of substandard and falsified medical products, currently inflicting severe pain and harm on patient’s lives, due to porous borders and ineffective regulatory control…
Call to Heads of State of the African Union to Ratify the African Medicines Agency
05 February 2021
PhRMA [to 6 Feb 2021]
http://www.phrma.org/
Selected Press Releases, Statements
Takeda Pharmaceuticals U.S.A. President Ramona Sequeira Becomes PhRMA Board Chair-Elect
February 3, 2021