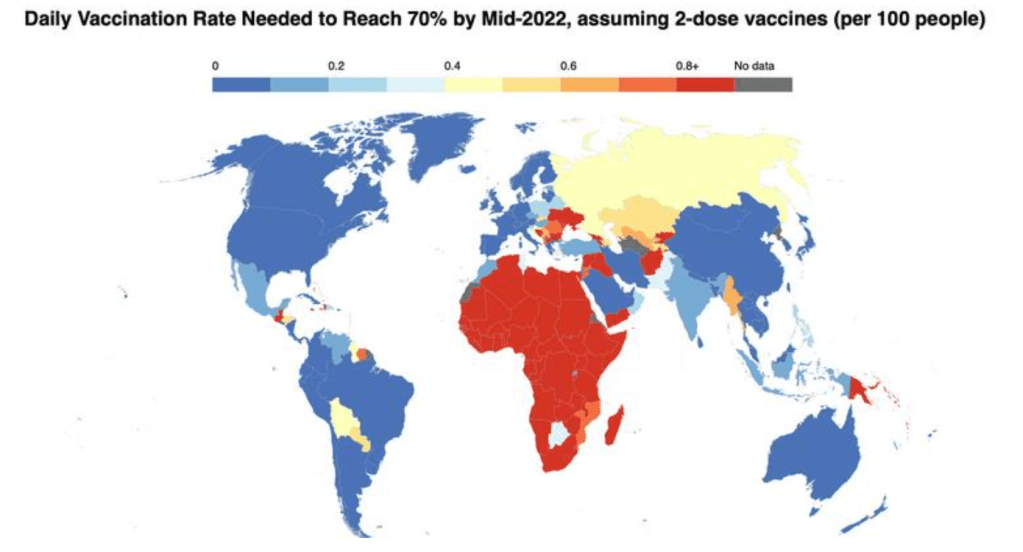
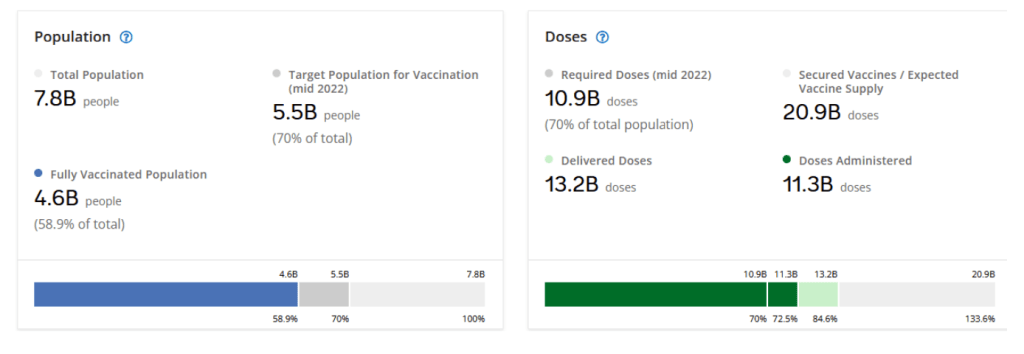
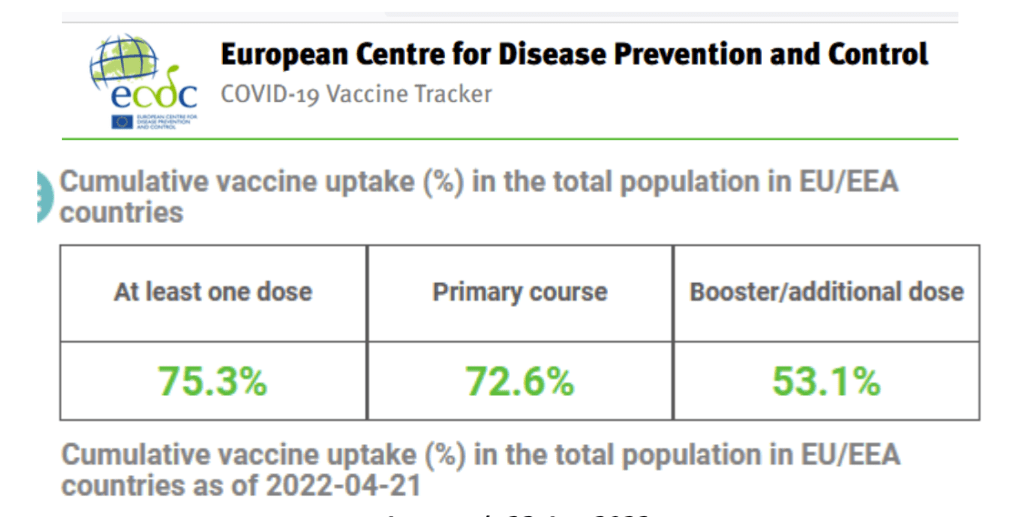
Organization Announcements
Editor’s Note:
Careful readers will note that the number and range of organizations now monitored in our Announcements section below has grown as the impacts of the pandemic have spread across global economies, supply chains and programmatic activity of multilateral agencies and INGOs.
Airfinity [to 23 Apr 2022]
https://www.airfinity.com/insights
INSIGHTS & COMPANY NEWS
Airfinity downgrades its global COVID-19 vaccine revenue forecast to $64.1B
Published by Airfinity
Apr 10, 2022
Airfinity has downgraded its global sales forecast for COVID-19 vaccines this year from $80.8bn to $64.1bn.
Paul G. Allen Frontiers Group [to 23 Apr 2022]
https://alleninstitute.org/news-press/
News
No new digest content identified.
BMGF – Gates Foundation [to 23 Apr 2022]
https://www.gatesfoundation.org/ideas/media-center
Press Releases and Statements
No new digest content identified.
Bill & Melinda Gates Medical Research Institute [to 23 Apr 2022]
https://www.gatesmri.org/news
The Bill & Melinda Gates Medical Research Institute is a non-profit biotech organization. Our mission is to develop products to fight malaria, tuberculosis, and diarrheal diseases—three major causes of mortality, poverty, and inequality in developing countries. The world has unprecedented scientific tools at its disposal; now is the time to use them to save the lives of the world’s poorest people
No new digest content identified.
CARB-X [to 23 Apr 2022]
https://carb-x.org/
News
No new digest content identified.
Center for Vaccine Ethics and Policy – GE2P2 Global Foundation [to 23 Apr 2022]
https://centerforvaccineethicsandpolicy.net/
News/Analysis/Statements
:: Past weekly editions and posting of all segments of Vaccines and Global Health: The Week in Review are available here.
:: Informed Consent: A Monthly Review – April 2022 is now posted here
:: Statement on ICF [Informed Consent Form] Posting for Clinical Trials Involving Gene Editing/Gene Therapies – Invitation to Endorse
:: Patients, Politics, Pressure :: Pharma Stays Steady [Mostly] In Russia Sanctions Context
Observations at 30 March 2022 David R Curry
CEPI – Coalition for Epidemic Preparedness Innovations [to 23 Apr 2022]
http://cepi.net/
Latest News
No new digest content identified.
DARPA – Defense Advanced Research Projects Agency [U.S.] [to 23 Apr 2022
https://www.darpa.mil/news
News
No new digest content identified.
Duke Global Health Innovation Center [to 23 Apr 2022]
https://dukeghic.org/
Our Blog
No new digest content identified.
EDCTP [to 23 Apr 2022]
http://www.edctp.org/
The European & Developing Countries Clinical Trials Partnership (EDCTP) aims to accelerate the development of new or improved drugs, vaccines, microbicides and diagnostics against HIV/AIDS, tuberculosis and malaria as well as other poverty-related and neglected infectious diseases in sub-Saharan Africa, with a focus on phase II and III clinical trials
News
No new digest content identified.
Emory Vaccine Center [to 23 Apr 2022]
http://www.vaccines.emory.edu/
Vaccine Center News
No new digest content identified.
European Vaccine Initiative [to 23 Apr 2022]
http://www.euvaccine.eu/
Latest News, Events
No new digest content identified.
Evidence Aid [to 23 Apr 2022]
Evidence Aid aims to save lives and livelihoods in disasters by providing decision-makers with the best available evidence and by championing its use.
http://www.evidenceaid.org/
New Resources
No new digest content identified.
Fondation Merieux [to 23 Apr 2022]
http://www.fondation-merieux.org/
News, Events
Mérieux Foundation & University of Geneva co-organized event
22nd Advanced Vaccinology Course (ADVAC)
May 9 – 20, 2022 – Les Pensières Center for Global Health, Veyrier-du-Lac (France)
Gavi [to 23 Apr 2022]
https://www.gavi.org/
News Releases
No new digest content identified.
GHIT Fund [to 23 Apr 2022]
https://www.ghitfund.org/newsroom/press
Press Releases
No new digest content identified.
Global Fund [to 23 Apr 2022]
https://www.theglobalfund.org/en/news/
News & Stories
No new digest content identified.
Global Research Collaboration for Infectious Disease Preparedness [GloPID-R] [to 23 Apr 2022]
https://www.glopid-r.org/news/
News
No new digest content identified
Hilleman Laboratories [to 23 Apr 2022]
http://www.hilleman-labs.org/
News & Insights
No new digest content identified
HHMI – Howard Hughes Medical Institute [to 23 Apr 2022]
https://www.hhmi.org/news
Press Room
No new digest content identified.
Human Vaccines Project [to 23 Apr 2022]
http://www.humanvaccinesproject.org/
News
No new digest content identified.
IAVI [to 23 Apr 2022]
https://www.iavi.org/
Latest News
No new digest content identified.
ICRC [to 23 Apr 2022]
https://www.icrc.org/en/whats-new
What’s New [Selected]
ICRC Moscow Delegation: in brief
The delegation is one of our key humanitarian diplomacy hubs around the world, building dialogue with the Russian authorities, academic and expert circles, and other influential people and organizations in order to support our mission to protect and as
18-04-2022 | Publication
International Coalition of Medicines Regulatory Authorities [ICMRA]
http://www.icmra.info/drupal/en/news
Selected Statements, Press Releases, Research
No new digest content identified.
IFFIm
http://www.iffim.org/
Press Releases/Announcements
No new digest content identified.
IFRC [to 23 Apr 2022]
http://media.ifrc.org/ifrc/news/press-releases/
Press Releases [Selected]
Americas: IFRC urges governments to save migrants’ lives at High Level Regional Meeting on Migration
20/04/2022 | Press release
IFRC urges governments to save migrants’ lives, ensure their access to essential services and social inclusion mechanisms, and scale up support to persons at risk of displacement related to disasters and the climate crisis…
Institut Pasteur [to 23 Apr 2022]
https://www.pasteur.fr/en/press-area
Press Documents
Press release
20.04.2022
When severe infection causes long-term mood disorders: A promising avenue to prevent mental illness following a transient infection
The brain is able to detect and regulate localized or systemic inflammation by using two communication pathways.
IOM / International Organization for Migration [to 23 Apr 2022]
http://www.iom.int/press-room/press-releases
News – Selected
News
22 Apr 2022
Almost 1 Million War Displaced to Benefit from IOM Ukraine Shelter Rehabilitation Programme
News
21 Apr 2022
One in Six People Internally Displaced in Ukraine
News
19 Apr 2022
Ukraine: Remarks by IOM Director General, António Vitorino, at the UN Security Council
ISC / International Science Council [to 23 Apr 2022]
https://council.science/current/
ISC is a non-governmental organization with a unique global membership that brings together 40 international scientific Unions and Associations and over 140 national and regional scientific organizations including Academies and Research Councils.
News Blogs Podcasts Events
News Press releases
International science organizations unite in support of at-risk, displaced and refugee scientists
20.04.2022
Join the advocacy campaign to support at-risk, displaced and refugee scientists by signing the Science in Exile Declaration.
IVAC [to 23 Apr 2022]
https://www.jhsph.edu/research/centers-and-institutes/ivac/index.html
Updates; Events
World Immunization Week 2022: Webinars
April 2022
During World Immunization Week 2022 the International Vaccine Access Center, along with global immunization and indigenous health partners, will host a three webinars. Registration is free, and we encourage all to sign up and save these dates on their calendars. Enabling Equitable Access to COVID-19 Vaccines for Indigenous Populations When: April 26, 2022 from 6:00 […]
IVI [to 23 Apr 2022]
http://www.ivi.int/
IVI News & Announcements
International Vaccine Institute appoints two members to its Board of Trustees representing Ecuador and Rwanda
April 20, 2022 – SEOUL, Republic of Korea – The International Vaccine Institute (IVI), an international organization with the mission to discover, develop and deliver safe, effective and affordable vaccines for global health, announced today that Dr. Ximena Garzón Villalba, Minister of Public Health of Ecuador, and Dr. Leon Mutesa, Professor of Human Genetics at the University of Rwanda, will join its Board of Trustees (BOT), representing Ecuador and Rwanda, respectively, two member states of IVI. Their terms will commence at the next BOT meeting scheduled for May 19-20, 2022, in Stockholm, Sweden…
Johns Hopkins Center for Health Security [to 23 Apr 2022]
https://www.centerforhealthsecurity.org/news/center-news/
Center News
No new digest content identified.
MSF/Médecins Sans Frontières [to 23 Apr 2022]
http://www.msf.org/
Latest [Selected Announcements]
No new digest content identified.
National Academy of Medicine – USA [to 23 Apr 2022]
https://nam.edu/programs/
News [Selected]
NAM Statement on the Importance of Ensuring a Systems-Based Approach to Patient Safety and Quality
April 19, 2022
The recent case of a former nurse who was found guilty of negligent homicide after the death of a patient due to a medication error prompts the need to underscore the recommendations from To Err is Human: Building a Safer Health System. A core finding of To Err is Human is that medical errors are most often caused by failures […]
New National Academy of Medicine Consensus Study Launched on Improving Cross-Sectoral, Coordinated, and Equitable Governance of Emerging Technologies in Health and Medicine
April 15, 2022
An ad hoc committee under the auspices of the National Academies of Sciences, Engineering, and Medicine has been convened to develop a cross-sectoral governance framework for considering the potential benefits and risks that emerging science, technology, and innovation in health and medicine can bring to society. The framework will translate key ethics principles into the […]
National Academy of Sciences – USA [to 23 Apr 2022]
http://www.nasonline.org/news-and-multimedia/
News
American Chemical Society Joins U.S. National Academy of Sciences in Effort to Support Researchers Forced to Flee Ukraine
April 20, 2022
With a 500000 donation the American Chemical Society has joined the U.S. National Academy of Sciences in an effort to help researchers who are being forced to flee Ukraine because of Russia’s invasion.
National Vaccine Program Office – U.S. HHS [to 23 Apr 2022]
https://www.hhs.gov/vaccines/about/index.html
Upcoming Meetings/Latest Updates
No new digest content identified.
NIH [to 23 Apr 2022]
http://www.nih.gov/
News Releases
Virtual Meeting and Listening Session on Oversight Framework for Research Involving Enhanced Potential Pandemic Pathogens
April 21, 2022 — Input from this listening session will help inform the deliberations and policy recommendations of the National Science Advisory Board for Biosecurity related to its current charge.
OECD [to 23 Apr 2022]
http://www.oecd.org/newsroom/publicationsdocuments/bydate/
Newsroom/Library
No new digest content identified.
PATH [to 23 Apr 2022]
https://www.path.org/media-center/
Press Releases
PATH announces Dr. Fredrick Were as Vice Chair of the Board of Directors
April 21, 2022 by PATH
PATH is pleased to announce Dr. Fredrick Namenya Were, MD, MMed, PhD as Vice Chair of the board of directors. Dr. Were brings a unique mix of policy, research, and clinical experience to the leadership role. He has served on the board since March 2021 and is a member of the Governance, Impact, and Responsibility Committee…
PATH welcomes US$ 5 million grant to expand malaria vaccine access in Ghana, Kenya, and Malawi
April 19, 2022 by PATH
Seattle, April 19, 2022—PATH today welcomed a grant of close to US$ 5 million to enable expanded introduction of the RTS,S/AS01 (RTS,S) malaria vaccine through the pilot program currently underway in areas of Ghana, Kenya, and Malawi. The grant would support use of RTS,S in pilot areas that have not yet received the vaccine.
The grant to PATH was recommended by GiveWell, a US-based non-profit, for funding by Open Philanthropy, also based in the United States. The award will provide each pilot country with funds to plan for and implement the expansion of malaria vaccination through the end of 2023, the formal end of the pilot program. The grant also will allow PATH and the World Health Organization (WHO) to provide technical assistance to the countries. The vaccine doses for the expansion into the (currently) non-vaccinating pilot areas are part of GSK’s donation of up to 10 million doses for use in the pilot program…
Sabin Vaccine Institute [to 23 Apr 2022]
http://www.sabin.org/updates/pressreleases
Statements and Press Releases
No new digest content identified.
UNAIDS [to 23 Apr 2022]
http://www.unaids.org/en
Selected Press Releases/Reports/Statements
22 April 2022
Sustaining HIV services for people who use drugs in Odesa
21 April 2022
Welcoming Ukrainian refugees in Germany: big test for all
20 April 2022
Social entrepreneurship—a tool of self-empowerment for the LGBTI community
UNHCR Office of the United Nations High Commissioner for Refugees [to 23 Apr 2022]
http://www.unhcr.org/en-us/media-centre.htmlS
Selected News Releases, Announcements
No new digest content identified.
UNICEF [to 23 Apr 2022]
https://www.unicef.org/media/press-releases
Press Releases, News Notes, Statements [Selected]
Press release
04/22/2022
At least 10 million children face severe drought in the Horn of Africa – UNICEF
https://www.unicef.org/press-releases/least-10-million-children-face-severe-drought-horn-africa-unicef
NAIROBI, 22 April 2022 – The number of children facing severe drought conditions across the Horn of Africa has increased by more than 40 per cent in the space of two months, warns UNICEF. Between February and April, the number of children facing the impact of drought including acute hunger, malnutrition and thirst increased from 7.25 million to…
Press release
04/21/2022
Child immunization programme reaches Rohingya refugee children on Bhasan Char island
https://www.unicef.org/press-releases/child-immunization-programme-reaches-rohingya-refugee-children-bhasan-char-island
DHAKA, 21 April 2022 – This week, Rohingya refugee children on Bhasan Char in Bangladesh are receiving vaccinations through a routine childhood immunization programme. UNICEF provides technical and financial assistance to the Government of Bangladesh in support of childhood immunizations on Bhasan Char. While Rohingya refugee children have…
[See Perspectives above for detail]
Press release
04/20/2022
Lebanon’s crisis threatening children’s health
https://www.unicef.org/press-releases/lebanons-crisis-threatening-childrens-health
BEIRUT, 20 April 2022 – The ripple effects of the global economic situation – with heightened prices and increased inflation – are exacerbating Lebanon’s calamitous crisis, with grave consequences for children’s health, UNICEF said in a report released to coincide with World Immunization Week (WIW). This will cause more disruptions in the health…
Statement
04/19/2022
Statement by UNICEF Executive Director Catherine Russell on attacks against schools in Kabul, Afghanistan
https://www.unicef.org/press-releases/statement-unicef-executive-director-catherine-russell-attacks-against-schools-kabul
NEW YORK, 19 April 2022 – “I am horrified by the brutal attacks targeting school children today in western Kabul, Afghanistan. The coordinated blasts occurred at the Mumtaz Tuition Center and Abdul Raheem Shaheed High School. “Reports are still being verified, but at least nine children are believed to have been killed and more than fifty injured…
Press release
04/18/2022
Sana’a authorities in Yemen sign action plan with UN to prevent grave violations against children
https://www.unicef.org/press-releases/sanaa-authorities-yemen-sign-action-plan-un-prevent-grave-violations-against
SANA’A, 18 April 2022 – Today Sana’a authorities signed an Action Plan with the United Nations to protect children and prevent grave violations against them in the context of the armed conflict in Yemen. The Action Plan commits the Sana’a authorities and their forces to comply with the prohibition of the recruitment and use of all children in…
Unitaid [to 23 Apr 2022]
https://unitaid.org/
Featured News
22 April 2022
World Malaria Day: Unitaid drives forward equitable access to innovation to reduce the burden of malaria
Vaccine Equity Cooperative [nee Initiative] [to 23 Apr 2022]
https://vaccineequitycooperative.org/news/
News
No new digest content identified.
Vaccination Acceptance & Demand Initiative [Sabin) [to 23 Apr 2022]
https://www.vaccineacceptance.org/
Announcements, Blog
Blog
Keeping Vaccination in the Conversation
April 18, 2022
by: Meredith Dockery, Nick Boehman, Abigail Quinn
Vaccine Confidence Project [to 23 Apr 2022]
http://www.vaccineconfidence.org/
News, Research and Reports
No new digest content identified.
Vaccine Education Center – Children’s Hospital of Philadelphia [to 23 Apr 2022]
http://www.chop.edu/centers-programs/vaccine-education-center
News
No new digest content identified.
Wellcome Trust [to 23 Apr 2022]
https://wellcome.ac.uk/news
News. Opinion, Reports
No new digest content identified.
The Wistar Institute [to 23 Apr 2022]
https://www.wistar.org/news/press-releases
Press Releases
No new digest content identified.
WFPHA: World Federation of Public Health Associations [to 23 Apr 2022]
https://www.wfpha.org/
Latest News – Blog
No new digest content identified.
World Bank [to 23 Apr 2022]
http://www.worldbank.org/en/news/all
Selected News, Announcements
EVENT: Scaling up Vaccine Deployment
As global vaccine supplies increase, a key challenge is to ensure that supplies quickly get to where they are needed and, once they are there, countries have the capabilities and resources to get vaccines…
Date: April 28, 2022 Type: Feature Story
World Customs Organization – WCO [to 23 Apr 2022]
http://www.wcoomd.org/
Latest News
No new digest content identified.
World Organisation for Animal Health (OIE) [to 23 Apr 2022]
https://www.oie.int/en/media/news/
Press Releases, Statements
Prevention is better than a cure: delivering quality vaccines to stop the spread of animal diseases
20 April 2022
WTO – World Trade Organisation [to 23 Apr 2022]
http://www.wto.org/english/news_e/news_e.htm
WTO News and Events [Selected]
Multilateralism “must be reimagined and fit for purpose,” DG tells Brazilian diplomats
18 April 2022
Brazil, and in particular its powerhouse agri-food sector, has benefited from open markets provided by the multilateral trading system, and it can benefit even more with a revitalized WTO, Director-General Ngozi Okonjo-Iweala told young diplomats and other guests at a lecture at Instituto Rio Branco, Brazil’s diplomatic academy, in Brasilia on 18 April. She stressed that multilateralism “must be reimagined and made fit for purpose to serve 21st century challenges”. The lecture opened a two-day visit for talks with government officials, the agriculture industry, parliamentarians and businesspeople. Her remarks are below.
Speech
::::::
ARM [Alliance for Regenerative Medicine] [to 23 Apr 2022]
https://alliancerm.org/press-releases/
Selected Press Releases
No new digest content identified.
BIO [to 23 Apr 2022]
https://www.bio.org/press-releases
Press Releases, Letters, Testimony, Comments [Selected]
No new digest content identified.
DCVMN – Developing Country Vaccine Manufacturers Network [to 23 Apr 2022]
http://www.dcvmn.org/
News; Upcoming events
No new digest content identified.
ICBA – International Council of Biotechnology Associations [to 23 Apr 2022]
https://internationalbiotech.org/news/
News
No new digest content identified.
IFPMA [to 23 Apr 2022]
http://www.ifpma.org/resources/news-releases/
Selected Press Releases, Statements, Publications
Joint Guidance on Virtual and Hybrid International Medical Congresses
13 April 2022
Joint Guidance on Virtual and Hybrid International Medical Congresses by the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA), the European Federation of Pharmaceutical Industries and Associations (EFPIA) and the Pharmaceutical Research and Manufacturers of America (PhRMA)
:: Joint Guidance on Virtual and Hybrid International Medical Congresses (April 2022)
:: Joint Guidance on Virtual and Hybrid International Medical Congresses (April 2022) Q&A
International Generic and Biosimilar Medicines Association [IGBA]
https://www.igbamedicines.org/
News
No new digest content identified.
International Alliance of Patients’ Organizations – IAPO [to 23 Apr 2022]
https://www.iapo.org.uk/news/topic/6
Press and media [Selected]
No new digest content identified.
PhRMA [to 23 Apr 2022]
http://www.phrma.org/
Latest News [Selected]
No new digest content identified.