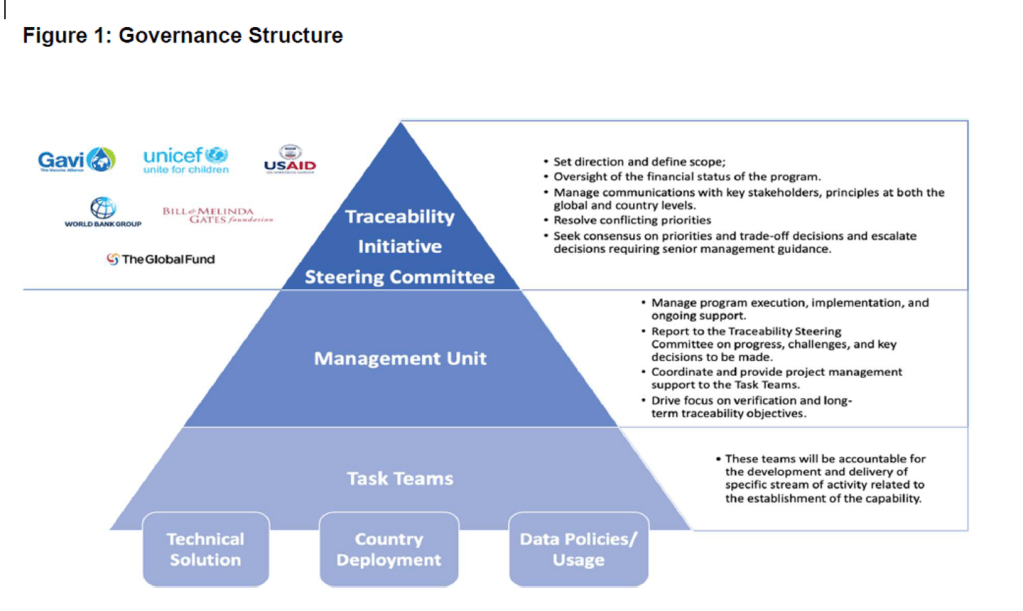
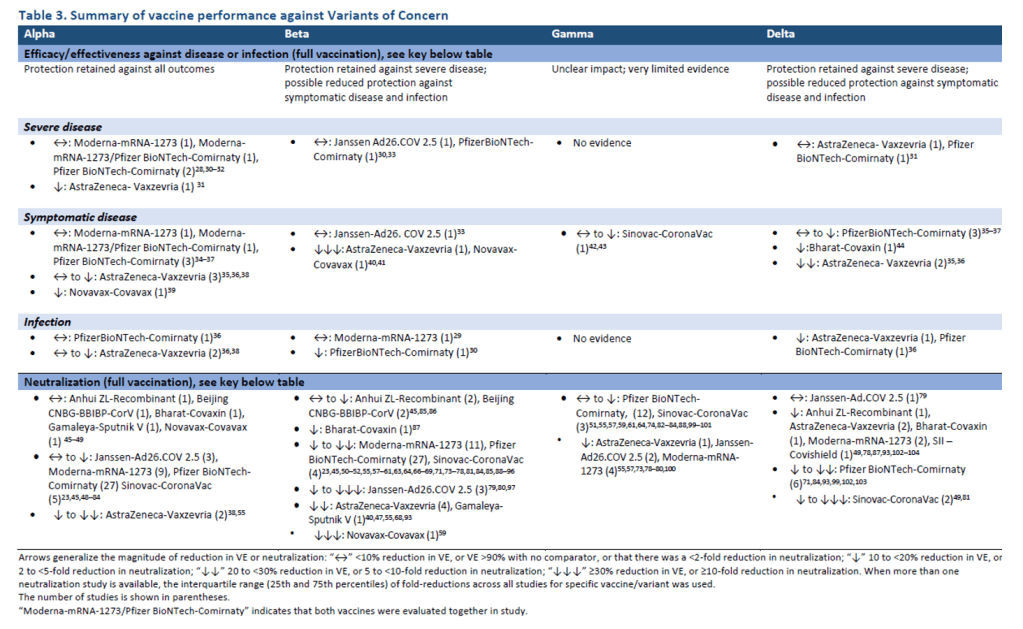
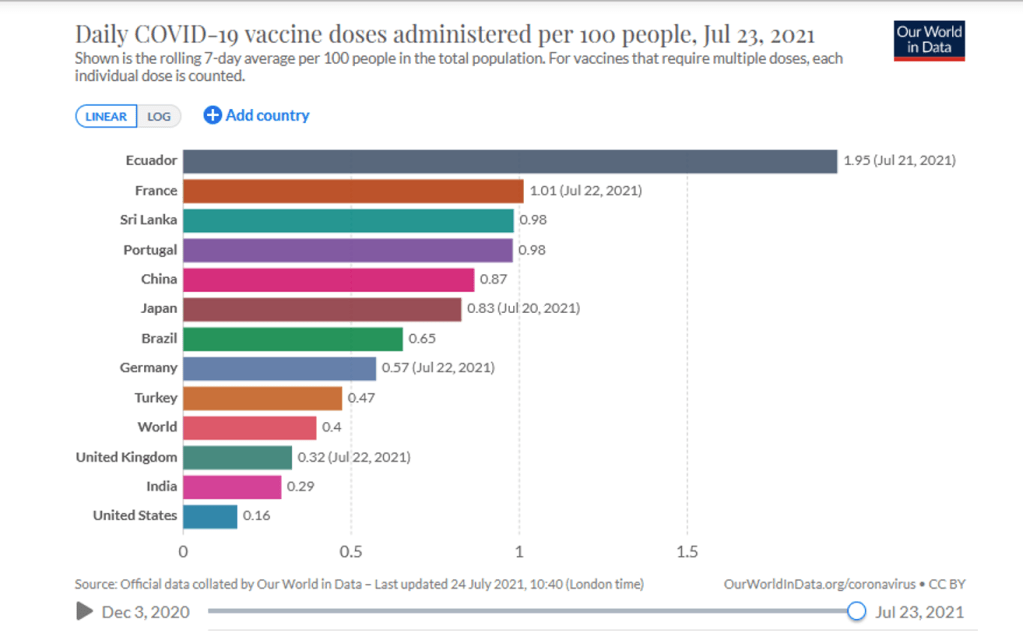
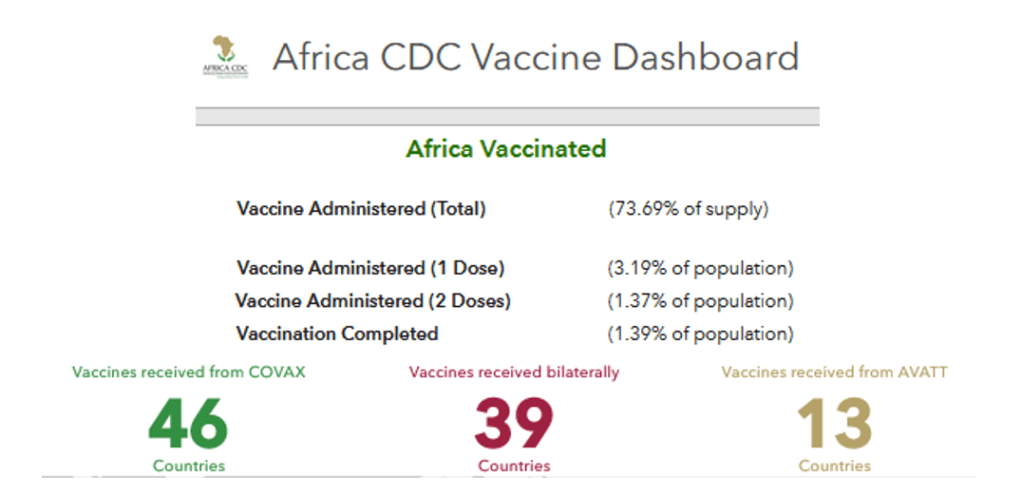
Organization Announcements
Editor’s Note:
Careful readers will note that the number and range of organizations now monitored in our Announcements section below has grown as the impacts of the pandemic have spread across global economies, supply chains and programmatic activity of multilateral agencies and INGOs.
Paul G. Allen Frontiers Group [to 24 Jul 2021]
https://alleninstitute.org/what-we-do/frontiers-group/news-press/
News
No new digest content identified.
BARDA – U.S. Department of HHS [to 24 Jul 2021]
https://www.phe.gov/about/barda/Pages/default.aspx
News
No new digest content identified.
BMGF – Gates Foundation [to 24 Jul 2021]
https://www.gatesfoundation.org/ideas/media-center
Press Releases and Statements
No new digest content identified.
Bill & Melinda Gates Medical Research Institute [to 24 Jul 2021]
https://www.gatesmri.org/
The Bill & Melinda Gates Medical Research Institute is a non-profit biotech organization. Our mission is to develop products to fight malaria, tuberculosis, and diarrheal diseases—three major causes of mortality, poverty, and inequality in developing countries. The world has unprecedented scientific tools at its disposal; now is the time to use them to save the lives of the world’s poorest people
No new digest content identified.
CARB-X [to 24 Jul 2021]
https://carb-x.org/
News
No new digest content identified.
Center for Vaccine Ethics and Policy – GE2P2 Global Foundation [to 24 Jul 2021]
https://centerforvaccineethicsandpolicy.net/
News/Analysis/Statements
:: Past weekly editions and posting of all segments of Vaccines and Global Health: The Week in Review are available here.
:: [NEW] Informed Consent: A Monthly Review – July 2021 is now posted here
CEPI – Coalition for Epidemic Preparedness Innovations [to 24 Jul 2021]
http://cepi.net/
Latest News
CEPI partners with Shanghai Zerun Biotech to develop COVID-19 variant vaccine
CEPI will provide US$13.1 million to advance development of both COVID-19 prototype and variant vaccine candidates.
COVAX 21 Jul 2021
CEPI and IVI collaborate on clinical research to expand access to COVID-19 vaccines in Africa
New programme of clinical research in Mozambique aims to support the expanded use of the BBIBP-CorV vaccine in Africa.
COVID-19 20 Jul 2021
CIOMS – COUNCIL FOR INTERNATIONAL ORGANIZATIONS OF MEDICAL SCIENCES [to 24 Jul 2021]
https://cioms.ch/
News; Publications
No new digest content identified.
DARPA – Defense Advanced Research Projects Agency [to 24 Jul 2021
https://www.darpa.mil/news
News
No new digest content identified.
Duke Global Health Innovation Center [to 24 Jul 2021]
https://dukeghic.org/
How can LMIC countries prepare to receive and efficiently administer large numbers of COVID-19 vaccine doses?
Melissa Slogan July 16, 2021
EDCTP [to 24 Jul 2021]
http://www.edctp.org/
The European & Developing Countries Clinical Trials Partnership (EDCTP) aims to accelerate the development of new or improved drugs, vaccines, microbicides and diagnostics against HIV/AIDS, tuberculosis and malaria as well as other poverty-related and neglected infectious diseases in sub-Saharan Africa, with a focus on phase II and III clinical trials
No new digest content identified.
Emory Vaccine Center [to 24 Jul 2021]
http://www.vaccines.emory.edu/
Vaccine Center News
No new digest content identified.
European Vaccine Initiative [to 24 Jul 2021]
http://www.euvaccine.eu/
Latest News
No new digest content identified
FDA [to 24 Jul 2021]
https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/default.htm
Press Announcements
July 23, 2021 – Coronavirus (COVID-19) Update: July 23, 2021
:: A new FDA Voices, Global Regulators Envision Paradigm Shift Toward Inclusion of Pregnant and Breastfeeding Women in Clinical Research for Medicines and Vaccines, describes efforts being made to address inadequacies in research through a paradigm shift in the approach to studying medical products in pregnant and breastfeeding women.
July 20, 2021 – Coronavirus (COVID-19) Update: July 20, 2021
:: This week, at the FDA SBIA REdI Annual Conference, FDA leaders reflected on the FDA’s use of Emergency Use Authorizations (EUAs) and other resources in making drug, device and biological products available to support the public health response to the COVID-19 pandemic. Day 1External Link Disclaimer and Day 2External Link Disclaimer recordings are now available on YouTube. The conference c
ontinues through Friday, with additional sessionsExternal Link Disclaimer on devices and biologics.
:: Last week, the FDA held a stakeholder call to discuss COVID-19 vaccines, including preliminary reports of Guillain-Barré Syndrome following Janssen COVID-19 vaccination. HereExternal Link Disclaimer is an excerpt from that call where Dr. Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research discussed the importance of getting vaccinated.
:: An FDA Consumer Update, Learn More About COVID-19 Vaccines From the FDA, is now available in five additional languages: Spanish, Chinese, Korean, Tagalog, and Vietnamese. See the evidence for each COVID-19 vaccine and the reasoning behind the FDA’s Emergency Use Authorizations (EUAs).
Fondation Merieux [to 24 Jul 2021]
http://www.fondation-merieux.org/
News, Events
No new digest content identified.
Gavi [to 24 Jul 2021]
https://www.gavi.org/
News Releases
23 July 2021
First doses donated by Japan to COVAX reach lower-income countries
GHIT Fund [to 24 Jul 2021]
https://www.ghitfund.org/newsroom/press
GHIT was set up in 212 with the aim of developing new tools to tackle infectious diseases that
No new digest content identified.
Global Fund [to 24 Jul 2021]
https://www.theglobalfund.org/en/news/
News & Stories
No new digest content identified.
Global Research Collaboration for Infectious Disease Preparedness [GloPID-R] [to 24 Jul 2021]
https://www.glopid-r.org/news/
News
No new digest content identified.
Hilleman Laboratories [to 24 Jul 2021]
http://www.hillemanlabs.org/
Website reports “under maintenance” at inquiry
Human Vaccines Project [to 24 Jul 2021]
http://www.humanvaccinesproject.org/
News
News webpage not responding at inquiry
IAVI [to 24 Jul 2021]
https://www.iavi.org/newsroom
Latest News
No new digest content identified.
International Coalition of Medicines Regulatory Authorities [ICMRA]
http://www.icmra.info/drupal/en/news
Selected Statements, Press Releases, Research
No new digest content identified.
ICRC [to 24 Jul 2021]
https://www.icrc.org/en/whats-new
Selected News Releases, Statements, Reports
Afghanistan: Civilians paying steep price of surge in violence and COVID-19
ICRC activities in Afghanistan, one of the the deadliest places in the world to be a civilian, and women and children make up nearly half of all civilian casualties.
21-07-2021 | News release
International Generic and Biosimilar Medicines Association [IGBA]
https://www.igbamedicines.org/
News
No new digest content identified.
IFFIm
http://www.iffim.org/
Press Releases/Announcements
No new digest content identified.
IFRC [to 24 Jul 2021]
http://media.ifrc.org/ifrc/news/press-releases/
Selected Press Releases, Announcements
No new digest content identified.
Institut Pasteur [to 24 Jul 2021]
https://www.pasteur.fr/en/press-area
Press Documents
Press release 16.07.2021
ComCor study: analysis of the effectiveness of messenger RNA vaccines against the SARS-CoV-2 Alpha and Beta variants in France
Over the past six months, the World Health Organization has categorized four SARS-CoV-2 variants as being “of concern”…
IOM / International Organization for Migration [to 24 Jul 2021]
http://www.iom.int/press-room/press-releases
News
No new digest content identified.
IRC International Rescue Committee [to 24 Jul 2021]
http://www.rescue.org/press-release-index
Media highlights [Selected]
No new digest content identified.
IVAC [to 24 Jul 2021]
https://www.jhsph.edu/research/centers-and-institutes/ivac/index.html
Updates; Events
Webinar: Preventing the Next Pandemic: Vaccine Diplomacy, Surveillance and COVID Vaccines
When: Jul 30, 2021 at 11:00 AM Eastern Time
Description: Join Dr. Peter Hotez for a 60-minute webinar hosted by the International Vaccine Access Center to discuss topics ranging from vaccine diplomacy, the antivaccine movement, safety surveillance, and a new COVID vaccine.
IVI [to 24 Jul 2021]
http://www.ivi.int/
Selected IVI News, Announcements, Events
CEPI and IVI collaborate on clinical research to expand access to COVID-19 vaccines in Africa
July 20, 2021; Oslo, Norway and Seoul, Republic of Korea: The Coalition for Epidemic Preparedness Innovations (CEPI) and the International Vaccine Institute (IVI) today announced a new programme of clinical research which aims to expand access to COVID-19 vaccines in Africa. CEPI will provide funding of up to $12.7m to the Expanding Access and Delivery of COVID-19 Vaccines in Africa (ECOVA) consortium led by IVI to carry out clinical trials of Sinopharm’s BBIBP-CorV vaccine which will support the expanded use of the vaccine on the African continent.
Up to 170 million doses of BBIBP-CorV will be distributed by the COVAX Facility under an agreement announced on 12 July 2021, and the vaccine is already being deployed in over 50 countries around the world. However there have been no clinical trials of BBIBP-CorV in African populations or against the variants of concern circulating in southern Africa. The ECOVA research programme aims to generate crucial data to inform the use of the vaccine in Africa, and potentially expand use of the vaccine to HIV-infected individuals. All results generated by ECOVA will be made available open source to inform policy makers and regulatory authorities’ recommendations on the use of BBIBP-CorV in national immunization programmes…
The ECOVA consortium is led by IVI, working in partnership with Mozambique’s Instituto Nacional de Saúde (INS), the International Centre for Diarrhoeal Disease Research, Bangladesh (icddr,b), the University of Heidelberg, Germany (UH), Harvard University, USA, and the University of Antananarivo in Madagascar (UA)…
JEE Alliance [to 24 Jul 2021]
https://www.jeealliance.org/
Selected News and Events
No new digest content identified.
Johns Hopkins Center for Health Security [to 24 Jul 2021]
https://www.centerforhealthsecurity.org/news/center-news/
Center News
No new digest content identified.
MSF/Médecins Sans Frontières [to 24 Jul 2021]
http://www.msf.org/
Latest [Selected Announcements]
South Africa
Supporting overstretched health centres and vulnerable communities in the aftermath of the violence
Project Update 23 Jul 2021
Mediterranean migration
MSF requests the revocation of the Geo Barents’ detention
Open Letter 23 Jul 2021
Central African Republic
Repeated attacks on medical care leave people vulnerable to disease and death
Press Release 19 Jul 2021
National Academy of Medicine – USA [to 24 Jul 2021]
https://nam.edu/programs/
Selected News/Programs
Discussion Paper Offers Guidance on Identifying Credible Sources of Health Information in Social Media
July 21, 2021
WASHINGTON — A new individually authored discussion paper in NAM Perspectives — the digital periodical of the National Academy of Medicine — outlines principles and attributes that social media platforms can use to identify potentially credible sources of health information online.
National Academy of Sciences – USA [to 24 Jul 2021]
http://www.nasonline.org/news-and-multimedia/
News
As Surgeon General Urges ‘Whole-of-Society’ Effort to Fight Health Misinformation the Work of the National Academies Helps Foster an Evidence-Based Information Environment
July 14, 2021
The U.S. surgeon general has released a public advisory calling misinformation a ‘serious threat to public health.’ The National Academies have been addressing misinformation in health and science on multiple fronts and are taking steps to help cultivate a fact- and evidence-based information environment.
National Vaccine Program Office – U.S. HHS [to 24 Jul 2021]
https://www.hhs.gov/vaccines/about/index.html
Upcoming Meetings/Latest Updates
No new digest content identified.
NIH [to 24 Jul 2021]
http://www.nih.gov/news-events/news-releases
News Releases
Tuesday, July 20, 2021
More than 1.5 million children lost a primary or secondary caregiver due to the COVID-19 pandemic
Addressing the impact of caregiver deaths critical for pediatric mental health, authors note.
PATH [to 24 Jul 2021]
https://www.path.org/media-center/
Press Releases
No new digest content identified.
Sabin Vaccine Institute [to 24 Jul 2021]
http://www.sabin.org/updates/pressreleases
Statements and Press Releases
No new digest content identified.
UNAIDS [to 24 Jul 2021]
http://www.unaids.org/en
Selected Press Releases/Reports/Statements
23 July 2021
2021 ECOSOC resolution on the Joint Programme adopted by consensus
22 July 2021
UNAIDS saddened by the death of Cyriaque Yapo Ako
22 July 2021
Changing the landscape of community engagement in the HIV response in Indonesia
21 July 2021
An HIV vaccine: who needs it?
21 July 2021
2025 AIDS targets: the next generation of goals for the global AIDS response
19 July 2021
Opening session of the IAS Conference on HIV Science
19 July 2021
KFF/UNAIDS analysis finds that while donor government spending on HIV increased in 2020, future funding is uncertain with COVID-19 challenges
UNHCR Office of the United Nations High Commissioner for Refugees [to 24 Jul 2021]
http://www.unhcr.org/en-us/media-centre.htmlS
Selected News Releases, Announcements
Record numbers forced to flee ongoing violence in Burkina Faso
23 Jul 2021
UNICEF [to 24 Jul 2021]
https://www.unicef.org/media/press-releases
Press Releases, News Notes, Statements [Selected]
Statement 07/19/2021
Statement by UNICEF Executive Director Henrietta Fore on children deprived of parental care due to COVID-19
NEW YORK, 19 July 2021 – “As the official COVID-19 death toll around the world passed 4 million earlier this month, UNICEF is increasingly concerned for children left without one or both parents.
“As with all crises and health pandemics, the most vulnerable children are at increased risk of losing parental care – due to death, severe illness or financial hardship. This in turn increases their risk of being placed in unsuitable alternative care.
“While it’s too early to estimate the number of children orphaned or abandoned as a result of the pandemic, a spike in deaths in some countries means many children already vulnerable to the impacts of COVID-19 face further emotional distress and protection concerns…
Unitaid [to 24 Jul 2021]
https://unitaid.org/
Featured News
No new digest content identified.
Vaccine Equity Initiative [to 24 Jul 2021]
https://vaccineequitycooperative.org/news/
News
No new digest content identified.
Vaccination Acceptance & Demand Initiative [Sabin) [to 24 Jul 2021]
https://www.vaccineacceptance.org/
Announcements
No new digest content identified.
Vaccine Confidence Project [to 24 Jul 2021]
http://www.vaccineconfidence.org/
News, Research and Reports
Coronavirus global impact
Launched April 2, 2020 and recurring every 3 days, Premise Data is utilizing its global network of Contributors to assess economic, social, and health sentiment surrounding the coronavirus (COVID-19).
Vaccine Education Center – Children’s Hospital of Philadelphia [to 24 Jul 2021]
http://www.chop.edu/centers-programs/vaccine-education-center
News
No new digest content identified.
Wellcome Trust [to 24 Jul 2021]
https://wellcome.ac.uk/news
News and reports
No new digest content identified.
The Wistar Institute [to 24 Jul 2021]
https://www.wistar.org/news/press-releases
Press Releases
Jul. 20, 2021
Wistar Scientists Discovered Fundamental and Widespread Gene Expression Control Mechanism that Potentially Creates Proteins at Distinct Locations in the Cell, with Implications in Immunity and Cancers
Through alternative polyadenylation, RNA molecules are directed to sites of protein synthesis to facilitate production of certain proteins where they are most needed.
WFPHA: World Federation of Public Health Associations [to 24 Jul 2021]
https://www.wfpha.org/
Latest News
No new digest content identified.
World Bank [to 24 Jul 2021]
http://www.worldbank.org/en/news/all
Selected News, Announcements
World Bank Group’s $157 Billion Pandemic Surge Is Largest Crisis Response in Its History
In response to COVID-19 severely damaging the lives and livelihoods of millions of people in developing countries, the World Bank Group deployed over $157 billion to fight the pandemic’s health, economic, and social impacts over the last 15 months (April 1, 2020 – June 30, 2021). This is the largest crisis response of any such period in the Bank Group’s history and represents an increase of more than 60% over the 15-month period prior to the pandemic. Bank Group commitments and mobilizations in fiscal year 2021 (FY21) alone (July 1, 2020 – June 30, 2021) amounted to almost $110 billion (or $84 billion excluding mobilization, short-term financing, and recipient-executed trust funds).
Since the start of the pandemic, the Bank Group supported countries to address the health emergency, strengthen health systems, protect the poor and vulnerable, support businesses, create jobs and jump start a green, resilient, and inclusive recovery.
Following last year’s COVID-related economic deterioration, the global economy is expected to expand 5.6% in 2021. Thus far, the recovery is uneven and many of the world’s poorest countries are being left behind. While about 90% of advanced economies are expected to regain their pre-pandemic per capita income levels by 2022, only about one-third of emerging market and developing economies are projected to do the same. In 2020, global extreme poverty rose for the first time in over 20 years, with nearly 100 million people pushed into extreme poverty.
Date: July 19, 2021 Type: Press Release
Health Emergency Preparedness and Response (HEPR) Umbrella Program
Supporting better health emergency preparedness and more effective responses
Given the urgency for stronger global health security and the need to help developing countries get better prepared for disease outbreaks, the World Bank Group established the new Health Emergency Preparedness and Response (HEPR) Umbrella Program to support eligible countries and territories to improve their capacities to prepare for, prevent, respond and mitigate the impact of epidemics on people.
The HEPR Umbrella Program provides financing to low-income countries and to countries with low health emergency preparedness and response capabilities. The program was set up earlier this year to support low-income countries and countries with struggling health emergency responses in building resilience and increasing investments in health preparedness – including their immediate COVID-19 response. It is an important tool in the World Bank’s pandemic response and longer-term health preparedness in countries that need support and their most vulnerable communities, including refugees.
Financial support is also available for countries that are unable to access regular for World Bank financing because they are in arrears with their payments to the International Development Association (IDA), and to Jordan and Lebanon for supporting Syrian refugees. Looking ahead, the program seeks to support more countries to meet critical needs.
Date: July 19, 2021 Type: Brief
World Customs Organization – WCO [to 24 Jul 2021]
http://www.wcoomd.org/
Latest News – Selected Items
23 July 2021
The role of Customs presented in a WHO-WTO High-Level Dialogue on expanding COVID-19 vaccine manufacturing
20 July 2021
The WCO Environment Programme engages with 11 Customs administrations in the field of illegal wildlife trade
World Organisation for Animal Health (OIE) [to 24 Jul 2021]
https://www.oie.int/en/for-the-media/press-releases/2021/
Press Releases, Statements
Editorial
Responding to emergencies through preparedness and resilience
19 July 2021
The COVID-19 pandemic has impacted almost all walks of life and it has required a multisectoral and interdisciplinary approach to respond to this global emergency.
Monique Éloit, Director General, World Organisation for Animal Health (OIE)
WTO – World Trade Organisation [to 24 Jul 2021]
http://www.wto.org/english/news_e/news_e.htm
WTO News and Events
WHO-WTO dialogue steps up efforts for increased COVID-19 vaccine production and equitable access
21 July 2021
The World Health Organization (WHO) and the WTO on 21 July hosted a High-Level Dialogue on “Expanding COVID-19 vaccine manufacture to promote equitable access”, with the participation of senior policymakers, heads of multilateral agencies, vaccine manufacturers, development finance institutions, global health initiatives and public health activists.
High-Level Dialogue on “Expanding COVID-19 vaccine manufacture to promote equitable access”
21 July 2021
Remarks by DG Okonjo-Iweala
TRIPS Council agrees to continue discussions on IP response to COVID-19
20 July 2021
At a meeting of the Council for Trade-Related Aspects of Intellectual Property Rights (TRIPS) on 20 July 2021, WTO members agreed to continue consideration of the proposal for a temporary waiver of certain TRIPS obligations in response to COVID-19 and other related proposals. Members approved a status report which they tasked the chair to deliver at the General Council at its next meeting on 27-28 July.
WTO publishes list of bottlenecks and facilitating measures on critical COVID-19 products
20 July 2021
The WTO Secretariat has issued an indicative list of trade-related bottlenecks and trade-facilitating measures on critical products to combat COVID-19, including inputs used in vaccine manufacturing, vaccine distribution and approval, therapeutics and pharmaceuticals, diagnostics and medical devices. The list is being released in advance of a High-Level Dialogue by the World Health Organization (WHO) and the WTO on 21 July on expanding COVID-19 vaccines manufacturing to promote equitable access.
::::::
ARM [Alliance for Regenerative Medicine] [to 24 Jul 2021]
Press Releases – Alliance for Regenerative Medicine (alliancerm.org)
Selected Press Releases
No new digest content identified.
BIO [to 24 Jul 2021]
https://www.bio.org/press-releases
Press Releases, Letters, Testimony, Comments [Selected]
No new digest content identified.
DCVMN – Developing Country Vaccine Manufacturers Network [to 24 Jul 2021]
http://www.dcvmn.org/
News; Upcoming events
No new digest content identified.
ICBA – International Council of Biotechnology Associations [to 24 Jul 2021]
https://internationalbiotech.org/news/
News
No new digest content identified.
IFPMA [to 24 Jul 2021]
http://www.ifpma.org/resources/news-releases/
Selected Press Releases, Statements, Publications
IFPMA Statement at WTO-WHO High Level Dialogue “Expanding COVID-19 Vaccine Manufacture to Promote Equitable Access”
21 July 2021
[See COVID above for detail]
International Alliance of Patients’ Organizations – IAPO [to 24 Jul 2021]
https://www.iapo.org.uk/news/topic/6
Press and media [Selected]
Patient empowerment can unlock the benefits of self-care
Thursday, 22 July 2021
Self-care activities are an essential component of patient-centered health care systems. The World Health Organization defines self-care as “the ability of individuals, families, and communities to promote health, prevent disease, maintain health, and to cope with illness and disability with or without the support of a healthcare provider.”
PhRMA [to 24 Jul 2021]
http://www.phrma.org/
Latest News [Selected]
Dose sharing as a part of the COVID-19 global vaccine equity strategy
July 20, 2021
Dose sharing is one of the strategies helping to accelerate work to broaden global access to COVID-19 vaccines…
…With millions of doses either shared or making their way to nations in need, we are already seeing the benefits of dose sharing as a strategy to promote COVID-19 vaccine equity. Leaders should focus on this and other meaningful solutions to ensure more shots are administered to more people.
Rather than pursuing misguided proposals to waive international commitments to protect intellectual property (IP) for COVID-19 innovations, industry supports practical strategies like dose sharing that better serve low- and middle-income nations while also upholding the IP ecosystem that made COVID-19 innovation possible.
Blog Post