::::::
Organization Announcements
Editor’s Note:
Careful readers will note that the number and range of organizations now monitored in our Announcements section below has grown as the impacts of the pandemic have spread across global economies, supply chains and programmatic activity of multilateral agencies and INGOs.
Paul G. Allen Frontiers Group [to 28 Aug 2021]
https://alleninstitute.org/what-we-do/frontiers-group/news-press/
News
No new digest content identified.
BARDA – U.S. Department of HHS [to 28 Aug 2021]
https://www.phe.gov/about/barda/Pages/default.aspx
News
No new digest content identified.
BMGF – Gates Foundation [to 28 Aug 2021]
https://www.gatesfoundation.org/ideas/media-center
Press Releases and Statements
No new digest content identified.
Bill & Melinda Gates Medical Research Institute [to 28 Aug 2021]
https://www.gatesmri.org/
The Bill & Melinda Gates Medical Research Institute is a non-profit biotech organization. Our mission is to develop products to fight malaria, tuberculosis, and diarrheal diseases—three major causes of mortality, poverty, and inequality in developing countries. The world has unprecedented scientific tools at its disposal; now is the time to use them to save the lives of the world’s poorest people
No new digest content identified.
CARB-X [to 28 Aug 2021]
https://carb-x.org/
News
No new digest content identified.
Center for Vaccine Ethics and Policy – GE2P2 Global Foundation [to 28 Aug 2021]
https://centerforvaccineethicsandpolicy.net/
News/Analysis/Statements
:: Past weekly editions and posting of all segments of Vaccines and Global Health: The Week in Review are available here.
:: [NEW] Informed Consent: A Monthly Review – August 2021 is now posted here
CEPI – Coalition for Epidemic Preparedness Innovations [to 28 Aug 2021]
http://cepi.net/
Latest News
Zalgen Awarded CEPI Contract to Provide Test Kits for Largest Ever Lassa Fever Study in West Africa
The study, Enable, seeks to better understand the rate, location, and spread of Lassa virus across the region, while also informing the location and implementation of future late-stage trials of Lassa vaccines.
25 Aug 2021
CEPI partners, IVI and BBIL, launch global Chikungunya vaccine Phase II/III trial in Costa Rica
IVI is leading the Global Chikungunya vaccine Clinical Development Program consortium in partnership with Bharat Biotech, and with support from CEPI and Ind-CEPI.
24 Aug 2021
CEPI welcomes US FDA approval of Pfizer/BioNTech’s COVID-19 vaccine
Statement from the CEO of CEPI, Dr. Richard Hatchett.
24 Aug 2021
DARPA – Defense Advanced Research Projects Agency [to 28 Aug 2021
https://www.darpa.mil/news
News
8/27/2021
Taking Guess Work out of Vaccine Development
Military service members rely on effective vaccination for the prevention of communicable disease and to guard against biothreat exposure, but current vaccine development is expensive, time-consuming and prone to failure. Many current vaccines do not provide effective protection over long periods of time, and there are multiple pathogens that lack prophylactic options. Immune response to vaccines is unpredictable, transient and ineffective, largely due to a lack of understanding of the complex mechanisms of action underlying immune memory. The Assessing Immune Memory (AIM) program seeks to develop a platform capability to predict immune memory informed by a systems-level view of the host response to vaccination and its mechanisms.
8/25/2021
Delivering Next-Gen Biomanufacturing Capability
Slow production methods and reliance on a global supply chain currently limit DoD access to critical proteins such as medical countermeasures (MCMs) (i.e. antibodies, vaccines, and clotting factors), diagnostic components, and key enzymes for producing nucleic acids. State-of-the-art protein-based MCM production calls for massive centralized infrastructure and complex pipelines that require lengthy cellular engineering, sufficient growth of cells, and intensive purification and quality control.
Duke Global Health Innovation Center [to 28 Aug 2021]
https://dukeghic.org/
Our Blog
No new digest content identified.
EDCTP [to 28 Aug 2021]
http://www.edctp.org/
The European & Developing Countries Clinical Trials Partnership (EDCTP) aims to accelerate the development of new or improved drugs, vaccines, microbicides and diagnostics against HIV/AIDS, tuberculosis and malaria as well as other poverty-related and neglected infectious diseases in sub-Saharan Africa, with a focus on phase II and III clinical trials
No new digest content identified.
Emory Vaccine Center [to 28 Aug 2021]
http://www.vaccines.emory.edu/
Vaccine Center News
No new digest content identified.
European Vaccine Initiative [to 28 Aug 2021]
http://www.euvaccine.eu/
Latest News
No new digest content identified.
Fondation Merieux [to 28 Aug 2021]
http://www.fondation-merieux.org/
News, Events
No new digest content identified.
Gavi [to 28 Aug 2021]
https://www.gavi.org/
News Releases
26 August 2021
First Danish doses delivered through COVAX arrive in Algeria
As part of its pledge to deliver 1 million doses of COVID-19 vaccines, the first doses donated by Denmark have been delivered to Algeria
GHIT Fund [to 28 Aug 2021]
https://www.ghitfund.org/newsroom/press
GHIT was set up in 212 with the aim of developing new tools to tackle infectious diseases that
No new digest content identified.
Global Fund [to 28 Aug 2021]
https://www.theglobalfund.org/en/news/
News & Stories
No new digest content identified.
Global Research Collaboration for Infectious Disease Preparedness [GloPID-R] [to 28 Aug 2021]
https://www.glopid-r.org/news/
News
The power of tracking COVID-19 research funding in real time
25/08/2021
In a Wellcome Open Research blog post, Dr. Alice Norton explains how the COVID-19 Project Tracker and associated Living Mapping Review were developed and why they are so valuable for funders.
The living mapping review, created jointly by UKCDR and GloPID-R, is a powerful tool for global funders to inform their ongoing research funding decisions. With analyses updated every three months, it allows them identify funding gaps and trends and avoid duplication.
As of the 1st July 2021 publication on Wellcome Open, the analysis contained 10,608 projects, funded by 201 funders, conducted in 142 countries and representing an investment of at least $4.7 billion.
Hilleman Laboratories [to 28 Aug 2021]
http://www.hillemanlabs.org/
Website reports “under maintenance” at inquiry
Human Vaccines Project [to 28 Aug 2021]
http://www.humanvaccinesproject.org/
News
No new digest content identified.
IAVI [to 28 Aug 2021]
https://www.iavi.org/newsroom
Latest News
No new digest content identified.
International Coalition of Medicines Regulatory Authorities [ICMRA]
http://www.icmra.info/drupal/en/news
Selected Statements, Press Releases, Research
No new digest content identified.
ICRC [to 28 Aug 2021]
https://www.icrc.org/en/whats-new
Selected News Releases, Statements, Reports
No new digest content identified.
IFFIm
http://www.iffim.org/
Press Releases/Announcements
No new digest content identified.
IFRC [to 28 Aug 2021]
http://media.ifrc.org/ifrc/news/press-releases/
Selected Press Releases, Announcements
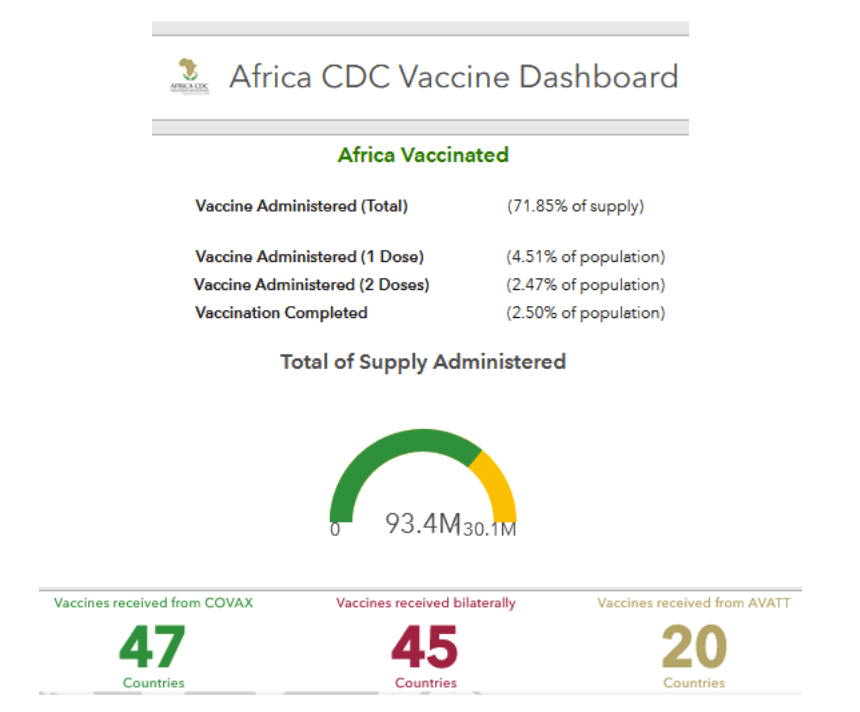
Africa
Africa CDC and IFRC ramp up COVID-19 response in Africa
Addis Ababa, 25 August 2021 – The Africa Centres for Disease Control and Prevention (Africa CDC) and the International Federation of Red Cross and Red Crescent Societies (IFRC) today launched a new collaboration to strengthen community resilience and response to public health emergencies at community level. The two institutions have signed a Memorandum of Understanding to ramp up pandemic response—including testing support to countries; community mobilization; advocacy and scaling up of contact tracing. In addition to COVID-19, the collaboration includes other areas of public health.
Africa CDC and IFRC will strengthen investments in locally-led action—for prevention and response purposes—while working with governments to ensure they intensify efforts to roll out the COVID-19 vaccination. Additionally, Africa CDC and IFRC will scale up advocacy against vaccine wastage.
This new initiative comes at a time Africa continues to face major vaccine shortages, amid a high level of community transmission in countries such as Botswana, Burundi, Eswatini, Cabo Verde, Namibia, Seychelles, South Africa, Zambia and Zimbabwe…
Institut Pasteur [to 28 Aug 2021]
https://www.pasteur.fr/en/press-area
Press Documents
Press Info
26.08.2021
COVID-19: Will telecommuting strategies stop the virus from circulating?
How can we best organise on-site workplace and school attendance periods and remote work to slow the circulation of…
IOM / International Organization for Migration [to 28 Aug 2021]
http://www.iom.int/press-room/press-releases
News
26 Aug 2021
USD 24 Million Urgently Needed for Acute Humanitarian Needs in Afghanistan
News
23 Aug 2021
IOM Appeals for USD 15 Million for Critical Needs of Families Affected by Haiti Earthquake
ISC / International Science Council [to 28 Aug 2021]
ISC is a non-governmental organization with a unique global membership that brings together 40 international scientific Unions and Associations and over 140 national and regional scientific organizations including Academies and Research Councils.
https://council.science/current/
News
No new digest content identified.
IVAC [to 28 Aug 2021]
https://www.jhsph.edu/research/centers-and-institutes/ivac/index.html
Updates; Events
No new digest content identified.
IVI [to 28 Aug 2021]
http://www.ivi.int/
Selected IVI News, Announcements, Events
IVI and BBIL launch global Chikungunya vaccine Phase II/III trial in Costa Rica
IVI is leading the Global Chikungunya vaccine Clinical Development Program (GCCDP) consortium in partnership with Bharat Biotech International Ltd. and with support from the Coalition for Epidemic Preparedness Innovations (CEPI) and Ind-CEPI.
August 24, 2021 – SEOUL, Republic of Korea – The International Vaccine Institute (IVI) announced today the first participant received Bharat Biotech International Ltd’s (BBIL) Chikungunya vaccine candidate (BBV87) in a Phase II/III clinical trial in Costa Rica, marking the start of a multi-country study led by IVI in partnership with BBIL and funded by the Coalition for Epidemic Preparedness Innovations (CEPI) with support from the Ind-CEPI mission of the Department of Biotechnology, India.
IVI is advancing clinical development of BBV87 through a Phase II/III randomized, controlled trial to evaluate the safety and immunogenicity of a 2-dose regimen of BBV87 Chikungunya vaccine in healthy adults at 9 clinical trial sites across 5 countries with endemic Chikungunya. In addition to the trial at Clinica San Agustin in Costa Rica, trials are expected to begin in Panama and Colombia by September 2021 and in Thailand and Guatemala soon after…
Johns Hopkins Center for Health Security [to 28 Aug 2021]
https://www.centerforhealthsecurity.org/news/center-news/
Center News
No new digest content identified.
MSF/Médecins Sans Frontières [to 28 Aug 2021]
http://www.msf.org/
Latest [Selected Announcements]
South Sudan
Hepatitis E on the rise among poor sanitary conditions in Bentiu camp
Press Release 25 Aug 2021
National Academy of Medicine – USA [to 28 Aug 2021]
https://nam.edu/programs/
Selected News/Programs
No new digest content identified.
National Academy of Sciences – USA [to 28 Aug 2021]
http://www.nasonline.org/news-and-multimedia/
News
No new digest content identified.
National Vaccine Program Office – U.S. HHS [to 28 Aug 2021]
https://www.hhs.gov/vaccines/about/index.html
Upcoming Meetings/Latest Updates
No new digest content identified.
NIH [to 28 Aug 2021]
http://www.nih.gov/news-events/news-releases
News Releases
NIH launches study of extra COVID-19 vaccine dose in people with autoimmune disease
Trial also will test pausing immunosuppressive medication to improve antibody response.
August 27, 2021 — The National Institutes of Health has begun a clinical trial to assess the antibody response to an extra dose of an authorized or approved COVID-19 vaccine in people with autoimmune disease who did not respond to an original COVID-19 vaccine regimen. The trial also will investigate whether pausing immunosuppressive therapy for autoimmune disease improves the antibody response to an extra dose of a COVID-19 vaccine in this population. The Phase 2 trial is sponsored and funded by the National Institute of Allergy and Infectious Diseases (NIAID), part of NIH, and is being conducted by the NIAID-funded Autoimmunity Centers of Excellence…
PATH [to 28 Aug 2021]
https://www.path.org/media-center/
Press Releases
PATH welcomes remarkable results from clinical trial combining RTS,S/AS01 malaria vaccine with Seasonal Malaria Chemoprevention
Seattle, WA, August 25, 2021 — Today, the results of a three-year Phase 3 malaria vaccine study conducted in Burkina Faso and Mali by the London School of Hygiene and Tropical Medicine (LSHTM), Institut de Recherche en Sciences de la Santé, and the Malaria Research and Training Centre, University of Bamako, were published. The study found that not only was the impact of seasonal vaccination with RTS,S/AS01E (RTS,S) comparable to that of seasonal malaria chemoprevention (SMC), but that combining the two interventions reduced malaria episodes and deaths in children by about 70 percent compared to either intervention alone…
Sabin Vaccine Institute [to 28 Aug 2021]
http://www.sabin.org/updates/pressreleases
Statements and Press Releases
No new digest content identified.
UNAIDS [to 28 Aug 2021]
http://www.unaids.org/en
Selected Press Releases/Reports/Statements
24 August 2021
Key population-led social enterprises awarded UNAIDS Solidarity Fund grants begin implementation
23 August 2021
The humanitarian activist supporting Venezuelan migrants living with HIV in Brazil
UNHCR Office of the United Nations High Commissioner for Refugees [to 28 Aug 2021]
http://www.unhcr.org/en-us/media-centre.htmlS
Selected News Releases, Announcements
No new digest content identified.
UNICEF [to 28 Aug 2021]
https://www.unicef.org/media/press-releases
Press Releases, News Notes, Statements [Selected]
Press release
08/24/2021
Poverty, conflict and displacement leave children at risk from renewed Bubonic plague outbreaks in Ituri region of eastern DRC
The centuries-old illness, easily treatable with antibiotics, is making a comeback through a combination of poverty and continuing insecurity
Statement
08/22/2021
Amidst Kabul airport blockage, WHO and UNICEF call for assistance to deliver critical health supplies to Afghanistan
Joint statement by WHO Regional Director for Eastern and Mediterranean Region, Dr. Ahmed Al-Mandhari, and UNICEF Regional Director for South Asia, George Laryea-Adjei
Unitaid [to 28 Aug 2021]
https://unitaid.org/
Featured News
No new digest content identified.
Vaccine Equity Initiative [to 28 Aug 2021]
https://vaccineequitycooperative.org/news/
News
No new digest content identified.
Vaccination Acceptance & Demand Initiative [Sabin) [to 28 Aug 2021]
https://www.vaccineacceptance.org/
Announcements
No new digest content identified.
Vaccine Confidence Project [to 28 Aug 2021]
http://www.vaccineconfidence.org/
News, Research and Reports
Coronavirus global impact
Launched April 2, 2020 and recurring every 3 days, Premise Data is utilizing its global network of Contributors to assess economic, social, and health sentiment surrounding the coronavirus (COVID-19).
Vaccine Education Center – Children’s Hospital of Philadelphia [to 28 Aug 2021]
http://www.chop.edu/centers-programs/vaccine-education-center
News
No new digest content identified.
Wellcome Trust [to 28 Aug 2021]
https://wellcome.ac.uk/news
News and reports
Opinion
No new digest content identified.
The Wistar Institute [to 28 Aug 2021]
https://www.wistar.org/news/press-releases
Press Releases
Aug. 25, 2021
Wistar Scientists Unveil Widespread Tumor Suppression Mechanism That Stops Cancer Progression by Interfering With Cancer Cell Metabolism
Parkin tumor suppressor blocks tumor growth and invasion by inhibiting metabolic reprogramming and mitochondrial function.
WFPHA: World Federation of Public Health Associations [to 28 Aug 2021]
https://www.wfpha.org/
Latest News
No new digest content identified.
World Bank [to 28 Aug 2021]
http://www.worldbank.org/en/news/all
Selected News, Announcements
Joint Statement of the Multilateral Leaders Taskforce on Scaling COVID-19 Tools
A Crisis of Vaccine Inequity
Date: August 27, 2021 Type: Statement
[See COVID above for full text]
World Customs Organization – WCO [to 28 Aug 2021]
http://www.wcoomd.org/
Latest News – Selected Items
No new digest content identified.
World Organisation for Animal Health (OIE) [to 28 Aug 2021]
https://www.oie.int/en/for-the-media/press-releases/2021/
Press Releases, Statements
No new digest content identified.
WTO – World Trade Organisation [to 28 Aug 2021]
http://www.wto.org/english/news_e/news_e.htm
WTO News and Events
No new digest content identified.
::::::
ARM [Alliance for Regenerative Medicine] [to 28 Aug 2021]
Press Releases – Alliance for Regenerative Medicine (alliancerm.org)
Selected Press Releases
No new digest content identified.
BIO [to 28 Aug 2021]
https://www.bio.org/press-releases
Press Releases, Letters, Testimony, Comments [Selected]
No new digest content identified.
DCVMN – Developing Country Vaccine Manufacturers Network [to 28 Aug 2021]
http://www.dcvmn.org/
News; Upcoming events
No new digest content identified.
ICBA – International Council of Biotechnology Associations [to 28 Aug 2021]
https://internationalbiotech.org/news/
News
No new digest content identified.
IFPMA [to 28 Aug 2021]
http://www.ifpma.org/resources/news-releases/
Selected Press Releases, Statements, Publications
IFPMA Statement at the 11th APEC High-level Meeting on Health and the Economy – Session
24 August 2021 – The biopharmaceutical industry is delivering. We have several safe and highly effective vaccines. We estimate that we are on track to produce more than 11 billion doses by the end of the year. This is a massive scale up from zero to billions in a matter of months and would be enough to fulfill the promise of fair and equitable access to vaccines.
Critically, however, COVID-19 vaccines are not equally reaching priority populations worldwide. Since May, the industry has been calling for five steps to urgently advance COVID-19 vaccine equity – top of the list is dose sharing, and we are very pleased that political leaders have responded to the call. But more needs to be done. We know that, just in the US and the EU, there could be 1 billion doses in surplus by the end of the year. We have it in our gasp to make a difference; and get the vaccines to those who need them – wherever they live.
A key lesson of equitable access for future pandemics is that we can only leverage the full potential of multilateral effort such as COVAX if we manage to lock funding to secure doses earlier in the process.
Our solutions need to be sustainable. We should avoid quick fixes that respond to political pressure. For example, when we talk about increasing manufacturing capacity, we need to ensure its sustainability and guard against creating more problems than solutions.
There is a misconception that waiving intellectual property (IP) rights would lead to increased manufacturing capacity and solve the vaccine inequality we are witnessing. There is no such evidence. The challenge of vaccines production is manufacturing infrastructure, lack of skilled workers, trade restriction, and shortages of raw materials, not IP.
To scale up manufacturing, technology transfer is already happening. To date, there are 157 such deals, out of over 230 voluntary collaborations. Many of these were forged in the early days of the pandemic…
IFPMA Statement at the 71st session of the WHO Regional Committee for Africa: Special Event
24 August 2021
… IFPMA also supports the objectives of increasing local production of vaccines on the African continent, and we recognize the importance of voluntary technology transfer collaborations to meet this goal.
IFPMA and member companies will continue to work with countries to administer all the doses allocated to them and limit the risk of dose wastage. Likewise, IFPMA will continue to work on countering misinformation, misunderstanding, and disinformation on vaccines, which have led to vaccine hesitancy and the tragic destruction of unused vaccines on the continent.
IFPMA welcomes WHO AFRO’s commitment to work with the private sector as a partner; to address policies that enable access to vaccines and treatments for those in need, such as review of national stockpiling; and to foster an environment that encourages innovation, research, and development. The industry supports Universal Health Coverage (UHC) through collaborations and partnerships with governments, NGOs, and private sector entities to strengthen healthcare systems, ensure the quality and integrity of the pharmaceutical supply chain, train healthcare workers, and explore innovative approaches to financing UHC…
International Generic and Biosimilar Medicines Association [IGBA]
https://www.igbamedicines.org/
News
No new digest content identified.
International Alliance of Patients’ Organizations – IAPO [to 28 Aug 2021]
https://www.iapo.org.uk/news/topic/6
Press and media [Selected]
APPIS examines capability building gaps of Patient Organizations
Monday, 23 August 2021
The Asia Pacific Patient innovation Summit has published a whitepaper, which aims to serve as a blueprint to help patient organizations improve their approach to sustainable funding, digitalization, and multi-stakeholder partnerships.
PDF: https://www.iapo.org.uk/sites/default/files/files/The%20Asia%20Pacific%20Patient%20Innovation%20Summit%20Whitepaper.pdf
PhRMA [to 28 Aug 2021]
http://www.phrma.org/
Latest News [Selected]
Driving further innovation is critical to global vaccine equity
August 24, 2021 [Text bolding from original]
Since the start of the pandemic, biopharmaceutical companies have worked around the clock to research, develop and manufacture treatments and vaccines to fight COVID-19…
Thankfully, scientists and researchers have continued their tireless work to find solutions. In addition to the more than 100 candidate vaccines currently in clinical development and another 184 in pre-clinical development, scientists are:
Studying how well current vaccines can protect against new COVID-19 variants, and how vaccines might be updated to address these emerging variants. Several companies are researching vaccines that protect against multiple strains and looking into the impact of tailored booster shots in defending against new COVID-19 variants.
Exploring ways to improve dosage formulations and more convenient storage methods, as well as options for co-administration of vaccines. Pfizer and BioNTech, for instance, are currently exploring a freeze-dried COVID-19 vaccine that would allow for the vaccine to be stored at 2-8 °C—addressing vaccine storage requirements that are not always available in some parts of the world. Moderna is exploring the efficacy of boosters that contain lower dose levels, which could help provide more doses to communities worldwide.
Conducting clinical trials in populations that have not yet been cleared to receive vaccines, such as children and those with certain conditions. Vaccine manufacturers are currently studying the safety and efficacy of their COVID-19 vaccines for children—an important step in ensuring all populations are able to receive protection against the virus and serious disease.
Researching innovative treatment approaches. To date, the Food and Drug Administration (FDA) has granted emergency use authorization for six COVID-19 treatments and fully approved one antiviral therapy. And scientists around the world currently have nearly 600 different therapies in clinical trials that could potentially allow clinicians to more effectively treat COVID-19 and ultimately improve patient outcomes…Blog Post
Vaccine Approval Important Step Forward Against Pandemic, Reminder of What’s at Stake in Drug Pricing Debate
August 23, 2021
The FDA’s approval of Pfizer-BioNTech’s mRNA vaccine marks a significant step forward in our work to end COVID-19. But ironically, while we celebrate this major step forward, members of Congress are meeting to advance policies that could inhibit our industry’s ability to find future groundbreaking treatments and cures.
Press release