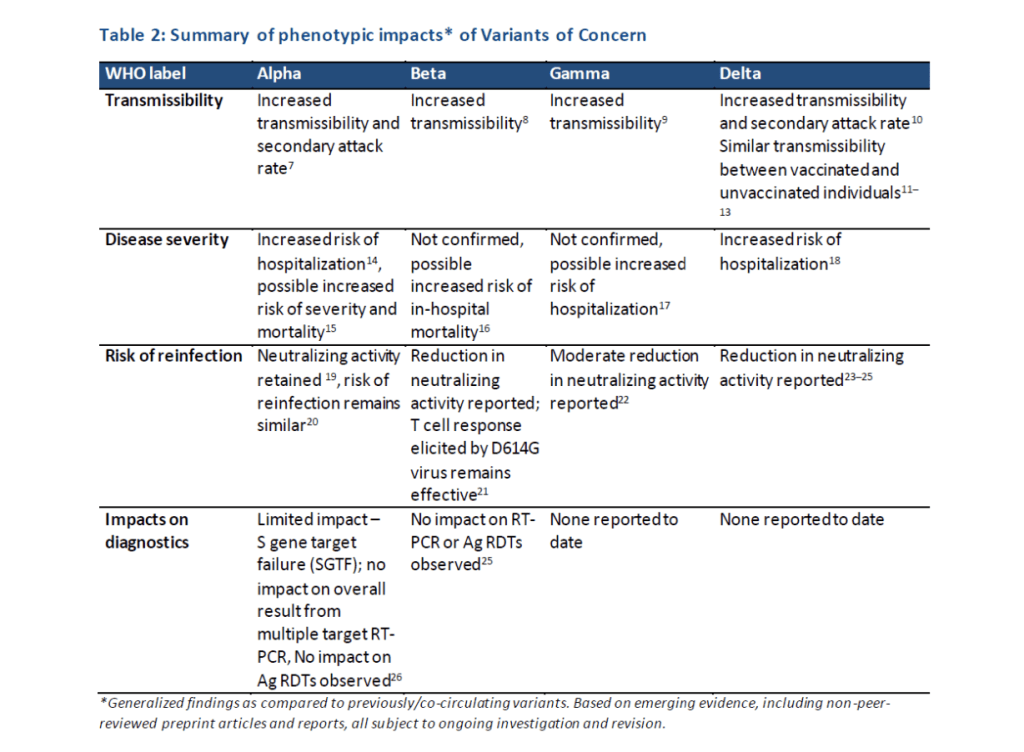
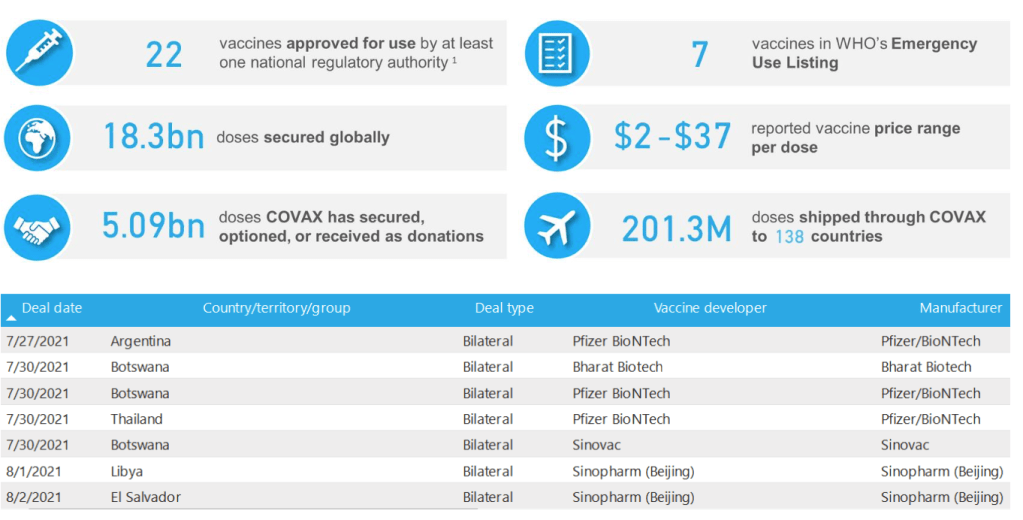
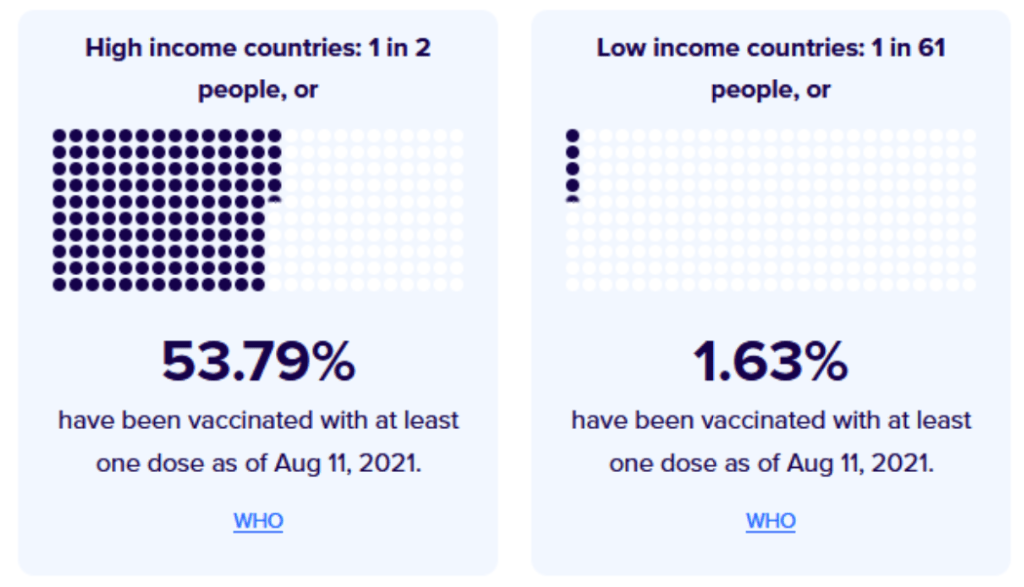
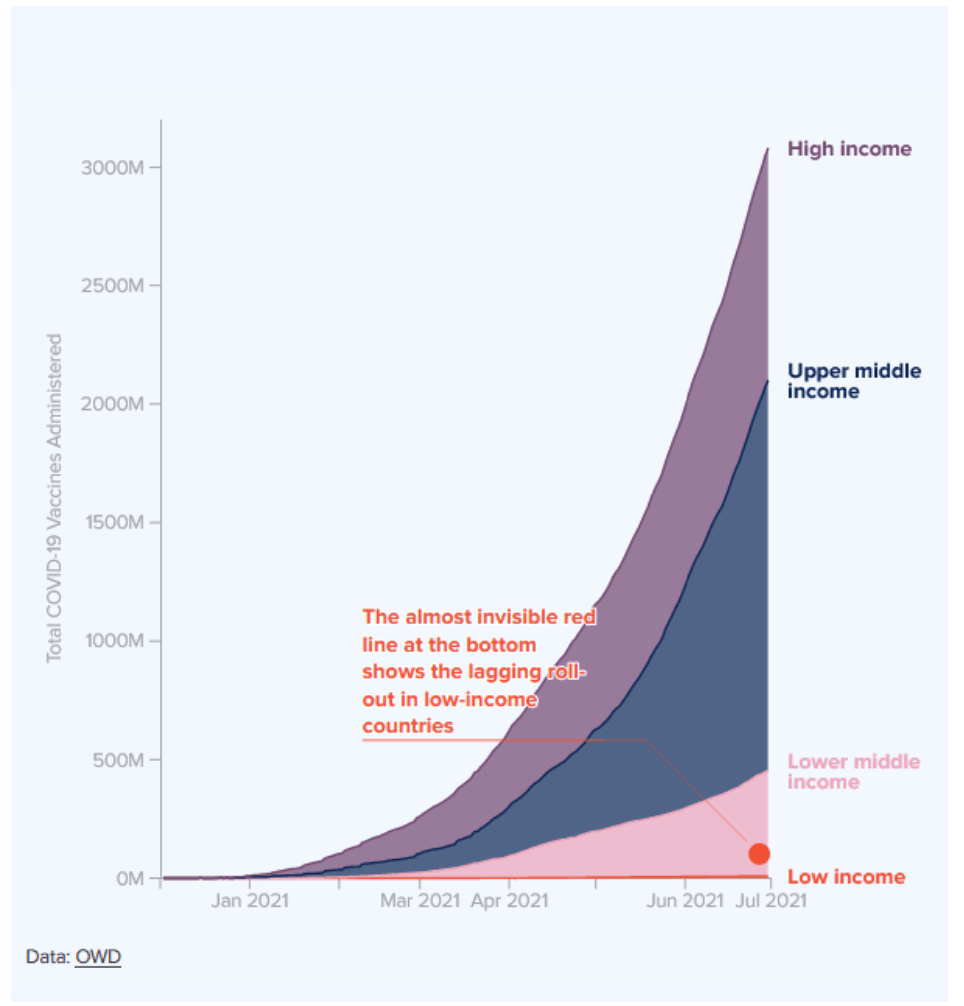
Milestones :: Perspectives :: Research
WHO’s Solidarity clinical trial enters a new phase with three new candidate drugs
11 August 2021 News release
Solidarity PLUS trial for promising drugs will roll out in 52 countries, an unprecedented global collaboration for COVID-19 R&D
The World Health Organization (WHO) has announced the next phase in its Solidarity trial: Solidarity PLUS will enroll hospitalized patients to test three new drugs in hospitalized COVID-19 patients.
These therapies – artesunate, imatinib and infliximab – were selected by an independent expert panel for their potential in reducing the risk of death in hospitalized COVID-19 patients. They are already used for other indications: artesunate is used for severe malaria, imatinib for certain cancers, and infliximab for diseases of the immune system such as Crohn’s Disease and rheumatoid arthritis.
These drugs were donated for the trial by their manufacturers.
“Finding more effective and accessible therapeutics for COVID-19 patients remains a critical need, and WHO is proud to lead this global effort,” said Dr Tedros Adhanom Ghebreyesus, WHO Director-General. “I would like to thank the participating governments, pharmaceutical companies, hospitals, clinicians and patients, who have come together to do this in true global solidarity.”
The Solidarity PLUS trial is a platform trial that represents the largest global collaboration among WHO Member States. It involves thousands of researchers in over 600 hospitals in 52 countries, 16 more countries than the first phase of trials. This allows the trial to assess multiple treatments at the same time using a single protocol, recruiting thousands of patients to generate robust estimates on the effect a drug may have on mortality–even moderate effects.
::::::
Interim statement on COVID-19 vaccine booster doses
10 August 2021 Statement
WHO, with support of the Strategic Advisory Group of Experts (SAGE) on Immunization and its COVID-19 Vaccines Working Group, is reviewing the emerging evidence on the need for and timing of an additional vaccine dose (booster dose 1) for the currently available COVID-19 vaccines which have received Emergency Use Listing (EUL). SAGE is continuously reviewing the literature and has reached out to vaccine manufacturers, the research community and Member States to obtain the most complete and recent data on the issue.
Rationale for booster doses
There are several reasons why COVID-19 vaccine booster doses may be needed: (i) waning protection against infection or disease, in particular severe disease, over time (i.e., waning immunity), (ii) reduced protection against variant(s) of concern (VOC), or (iii) inadequate protection from the currently recommended primary series for some risk groups for which evidence from the Phase 3 clinical trials may have been lacking. The rationale for booster doses may differ by vaccine product, epidemiological setting, risk group, and vaccine coverage rates…
…Conclusions
In the context of ongoing global vaccine supply constraints, administration of booster doses will exacerbate inequities by driving up demand and consuming scarce supply while priority populations in some countries, or subnational settings, have not yet received a primary vaccination series. The focus for the time being remains on increasing global vaccination coverage with the primary series (either one or two doses for current EUL vaccines).
Introducing booster doses should be firmly evidence-driven and targeted to the population groups in greatest need. The rationale for implementing booster doses should be guided by evidence on waning vaccine effectiveness, in particular a decline in protection against severe disease in the general population or in high risk populations, or due to a circulating VOC. To date, the evidence remains limited and inconclusive on any widespread need for booster doses following a primary vaccination series. WHO is carefully monitoring the situation and will continue to work closely with countries to obtain the data required for policy recommendations.
::::::
Interim statement on dose-sparing strategies for COVID-19 vaccines (fractionated vaccine doses)
10 August 2021 Statement
WHO, with support of the Strategic Advisory Group of Experts (SAGE) on Immunization and its COVID-19 Vaccines Working Group, is reviewing the role of fractionating doses as a dose-sparing strategy in light of global vaccine supply constraints. SAGE is continuously reviewing the literature and has reached out to vaccine manufacturers and the research community for available information.
Evidence for fractionated doses
All current COVID-19 vaccines have undergone dose-finding studies in their clinical development. The potential for dose-reduction may depend on the individual vaccine and its platform technology (e.g., mRNA, vectored or inactivated virus). Safety, immunogenicity and programmatic feasibility of fractionating doses has been shown and implemented for various, hitherto well characterized vaccines (e.g., polio, rabies, and yellow fever vaccines) (1, 2). Intradermal administration may enable reduction of dose volume, but intradermal application may also change the immunogenicity, safety and increase the reactogenicity profile of the vaccines. Scaling up intradermal administration at a global level would also have major programmatic challenges that would require substantial investments in training and logistics to address.
Reducing the amount of vaccine given (e.g., 1/2, 1/3 or 1/5) could theoretically be considered with various options: fractionated doses for the priming schedule, or fractionated doses for any booster doses should booster doses prove to be needed in the future.
However, policy recommendations for reducing doses should only be made after an extensive evidence review in terms of immunogenicity and safety. Emergency use listing for all COVID-19 vaccines for which SAGE has issued policy recommendations has been based on the evidence derived from Phase 3 trials using the full dose. Additional clinical studies would therefore be needed to inform policy…
…Conclusion
While SAGE acknowledges the potential public health benefits of dose-sparing strategies to increase vaccine supply and accelerate population-level vaccination coverage, and possibly also a reduction in reactogenicity, SAGE considers there is currently insufficient evidence to recommend the use of fractional doses. Any use of a fractional dose at this point in time constitutes an off-label use of the vaccine. SAGE encourages research in the area, with a particular emphasis on research into using fractionated doses as potential boosters and fractional doses in children and adolescents. Programmatic and operational considerations should be considered from the start.
::::::
Interim statement on heterologous priming for COVID-19 vaccines
10 August 2021 Statement
WHO with support of the Strategic Advisory Group of Experts (SAGE) on Immunization and its COVID-19 Vaccines Working Group is reviewing the emerging evidence on the use of heterologous priming schedules (also known as mix and match schedules). In a heterologous priming schedule, the second dose uses a different vaccine product than the first dose. By contrast, heterologous boosting refers to the administration of a vaccine from a different vaccine platform from the vaccine that was used to complete the primary vaccine series. The interim statement here pertains only to heterologous priming and not heterologous boosting.
Rationale for Heterologous Priming
The most common reason for considering a heterologous COVID-19 vaccine as second priming dose is lack of availability of the same vaccine in settings with limited vaccine supply or unpredictable supply. Interchangeability of vaccine products would therefore allow for added programmatic flexibility. There are other reasons to investigate the utility of heterologous priming such as reducing reactogenicity, increasing immunogenicity and enhancing vaccine effectiveness. Heterologous priming should only be instituted if supportive evidence is available.
Current state of knowledge
SAGE continuously reviews new data on heterologous priming vaccine schedules. Guidance is available in some of the product specific interim recommendations (to date, for mRNA vaccines, i.e. BNT162b2 or mRNA-1273, and ChAdOx1-S [recombinant] vaccines).
In general, for COVID-19 vaccines listed for emergency use by WHO with a 2-dose primary series schedule, WHO recommends that the same vaccine product should be used for both doses. If different COVID-19 vaccine products are inadvertently administered in the two doses, no additional doses of either vaccine are recommended. At present, mix and match schedules constitute off-label use of respective vaccines and as such should only be used if benefits outweigh the risks such as in situations of interrupted vaccine supply.
Studies to date of immune responses after a first dose of ChAdOx1-S [recombinant] products followed by an mRNA vaccine (i.e., BNT162b2 or mRNA-1273) show higher neutralising antibody levels and higher T cell-mediated immune responses in comparison with two doses of ChAdOx1-S [recombinant] products and similar levels to those of two mRNA vaccines (1). The sequence of the first dose being ChAdOx1-S [recombinant] followed by the mRNA vaccine as second dose was more immunogenic than a first dose mRNA vaccine followed by ChAdOx1-S [recombinant] vaccine.
While these studies are encouraging, they require cautious interpretation given the limited sample sizes and lack of follow up, especially related to safety data, and the uncertain relevance of immunological readouts in relation to clinical impact. The first results on short-term vaccine effectiveness (VE) against infection following a heterologous schedule have become available from Denmark showing an effectiveness of 88% (95% CI 83-92%) when combining the ChAdOx1 and an mRNA vaccine, similar to the VE of two doses of an mRNA vaccine, in a population-wide register-based study when the Alpha variant was dominant (2). More observational data on safety and effectiveness will be forthcoming.
While there are currently no data for heterologous priming with other vaccine products, a large number of clinical studies of various vaccine combinations and schedules are currently ongoing. SAGE will review these data as they become available and update the recommendations accordingly.