Milestones :: Perspectives :: Research
Editor’s Note:
These Gavi announcements represent critical milestones in COVID response and, as such, we present their full text.
Gavi Board strengthens commitment to reaching the most vulnerable through routine immunisation and COVAX
:: Gavi Board makes key strategic decisions to support Gavi’s 5.0 strategy of leaving no child behind through immunisation; doubles down on ambitious targets for COVAX
:: New market-shaping strategy, alliances with civil society among measures aimed at reducing the number of children receiving no doses of vaccine, by 25% globally – by 2025
:: US$ 775 million in COVAX delivery funding approved for lower-income economies that are eligible for support through the Gavi COVAX Advance Market Commitment (AMC)
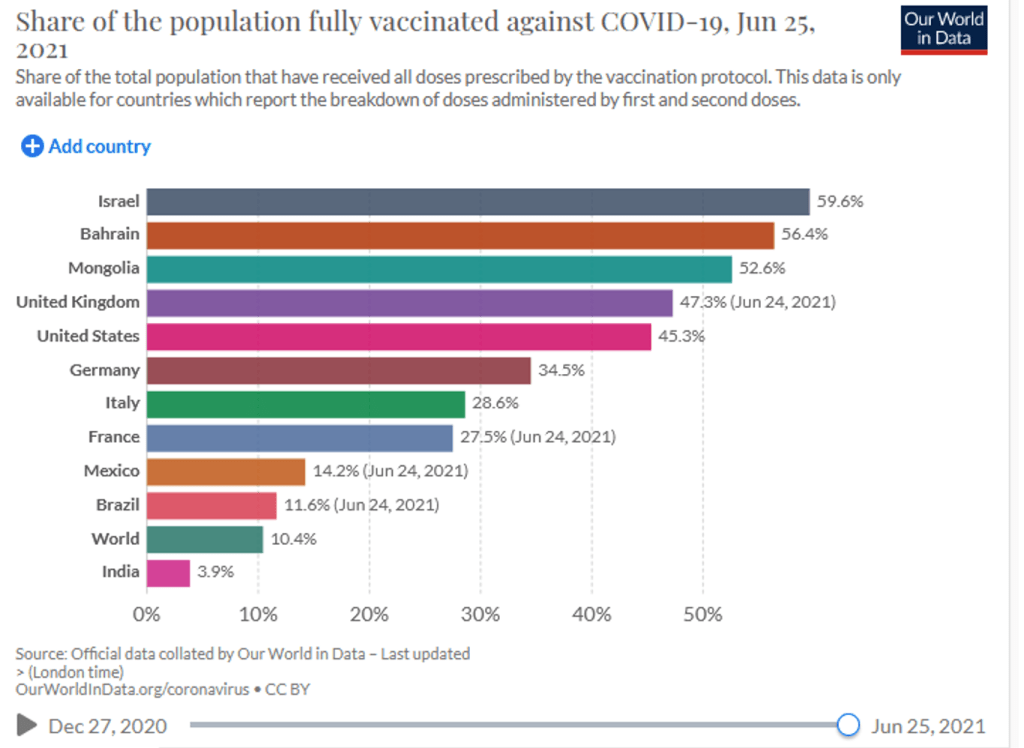
:: José Manuel Barroso: “Protecting the most vulnerable and enabling access to the lifesaving power of vaccines has always been at the heart of Gavi’s mission. As Africa, with the lowest vaccine coverage in the world, and other regions face a renewed surge of the virus, the global community must step up now to help COVAX to meet its ambitious equitable access goals to support these countries.”
Geneva, 25 June 2021 – The Board of Gavi, the Vaccine Alliance has made several decisions to strengthen the organisation’s commitment to protecting the most vulnerable everywhere – through routine immunisation, access to COVID-19 vaccines via the Gavi-hosted COVAX Facility and support for outbreak preparedness and response activities.
In addition to taking decisions relating to Gavi’s strategic direction, the Board united in condemning today’s gross inequity in vaccine delivery and committed itself to redoubling efforts to close the gap.
“Protecting the most vulnerable and enabling equitable access to lifesaving vaccines has always been at the heart of Gavi’s mission,” said José Manuel Barroso, Chair of the Gavi Board. “As Africa, with the lowest vaccine coverage in the world, and other regions face a renewed surge of the virus, we must use all our resourcefulness and innovation to help those most at risk: the global community must step up now to help COVAX to meet its ambitious equitable goals to support these countries.”
Supporting COVAX to deliver
Earlier this week COVAX, the world’s first global procurement facility for pandemic vaccines, released an updated supply forecast demonstrating that, thanks to new agreements with manufacturers and dose-sharing by higher income economies, supplies were expected to reach two billion doses in January 2022 – only a short time later than previous forecasts, despite severe on-going supply delays. According to the forecast, COVAX’s goal of delivering 1.8 billion doses to lower-income economies eligible for donor-supported doses via the Gavi COVAX Advance Market Commitment (COVAX AMC) would be reached in the first quarter of 2022.
In order to help prepare countries for this rapid scale up, the Gavi Board approved new funding of US$ 775 million to support the delivery of COVAX-funded doses in lower-income economies and humanitarian zones over the next two years. Gavi is co-lead of COVAX and hosts the Office of the COVAX Facility. The funding comes in addition to a previously Board-approved envelope of US$ 150 million in delivery support.
The Board also approved an evolution in the model of participation for self-financing participants (SFPs) of the COVAX Facility. Starting in 2022, the model will enable SFPs that rely on the Facility to access doses to continue procuring vaccines through COVAX under revised terms and conditions. The move, based on lessons learned over the past year, will enable simplified operations and reduce financial risks to Gavi and COVAX.
“As the only global solution designed around equitable access to COVID-19 vaccines, we need to continue to adapt the COVAX model as the pandemic evolves,” said Dr Seth Berkley, CEO of Gavi. “The additional delivery support funding will help make sure that COVAX-funded doses are rapidly and efficiently rolled out in lower-income economies. But it will not help us end the acute phase of pandemic alone: to do this, we need governments, manufacturers and multilateral development banks to truly get behind COVAX and get us doses now so we can scale up protecting lives and do so quickly.”
Reaching the most vulnerable with routine immunisation
Many decisions at the two-day board meeting concerned Gavi’s new 5.0 strategy.
Guiding the Alliance’s mission over the period 2021–2025, Gavi 5.0’s goal is to “leave no one behind with immunisation”, with a core focus on reaching “zero-dose” children and missed communities, with equity as the organising principle.
The following decisions are aimed at providing a critical framework for achieving this goal – building on Gavi’s existing strengths, as well as forming new partnerships and evolving key policies:
:: A measurement framework for the work of the Alliance in the next five years. In line with its 5.0 overarching objective of leaving no one behind, Gavi will aim to reduce the number of zero-dose children globally by 25% in 2021–2025. Other indicators include immunising an additional 300 million children and preventing 7-8 million future deaths through Gavi’s support for routine immunisation programmes and vaccination campaigns, while unlocking US$ 80-100 billion in economic benefits.
:: A 2021-2025 Market Shaping Strategy that will foster a sustainable, competitive future supplier base by encouraging healthy demand through a more coordinated and country-centric approach. The strategy will also focus on enabling vaccine product innovations that best meet country needs and that directly contribute to the Gavi 5.0 goals of increasing equitable vaccine coverage and reaching zero-dose children and missed communities.
:: The Civil Society and Community Engagement Approach to ensure effective management, enhanced capacity and efficient funding of Gavi’s engagement with civil society organisations (CSOs). Beyond their comparative advantage in reaching zero-dose children and missed communities, CSOs have key roles to play in helping to ensure immunisation is maintained, restored and strengthened in the context of COVID-19, and that vaccines are introduced in an equitable way.
:: An initial investment of up to US$ 5 million for the Vaccine Innovation Prioritisation Strategy (VIPS) for costs related to short-term (2021–2023) activities to provide guidance for and incentivise development of three innovation priorities, including clarifying the potential demand, defining investment cases and understanding the need for and design of push and pull funding mechanisms, as well as generating evidence of programmatic impact.
:: Yellow Fever diagnostic capacity strengthening initially to create the first commercial test kits for Yellow Fever, which has been accomplished, and support for costs related to the procurement and distribution of laboratory reagents, supplies and equipment which has been extended until the end of 2022. The costs remain within the initially approved envelope of US$ 8.2 million authorised for the 2019–2021 period.
:: An approach to strengthen fiduciary risk assurance and financial management of Gavi grants in the 5.0 strategic period, with associated investment of US$ 139 million for 2022–2025. As Gavi funding will increasingly be channelled through government systems, the investments will be used to help build sustainable financial management capacity of government systems, as well as ensure the continued use of fiduciary risk mitigation mechanisms.
Encompassing both COVAX and Gavi 5.0 is Gavi’s risk appetite statement, which defines the criteria and trade-off considerations that guide the Alliance’s appetite for risk at the mission, strategy and organisational level. Against the backdrop of the ongoing COVID-19 pandemic, the Board approved an approach where the Alliance will seek to find the right balance between successfully delivering on the COVAX mission, where Gavi plays a central role, while minimising risks to Gavi’s core resources and programmes, acknowledging the reality that a higher risk appetite is needed in a pandemic setting.
Key departures and appointments
The Board approved a one-year exceptional contract extension for Gavi CEO Dr Seth Berkley to guarantee stable and strong leadership of the Alliance in this critical time and ensure successful implementation of COVAX and Gavi 5.0. The current CEO’s 3rd term will now end in August 2023 and succession planning will be led by Gavi Board Chair.
The Board also welcomed new Board members including Awa Marie Coll Seck, Naguib Kheraj and Bounfeng Phoummalaysith.
In addition, the Board thanked Stephen Zinser and Muhammad Ali Pate as their terms as Board members concluded. Stephen Zinser has been a Gavi Board member since 2014 and has guided Gavi’s transition through two strategic periods. Muhammad Ali Pate began his term as a Board member in July 2019 lending his counsel and extensive experience to support the work of the Alliance. Mr Pate has also been an invaluable member of Market Sensitive Decisions Committee (MSDC) since October 2019.
Finally, the Gavi Board gave a warm send-off to Bill Roedy. Mr Roedy joined Gavi in 2010 as its first envoy. He was appointed to the Board in 2015 and has been Board Vice Chair in 2018–2019. Mr Roedy is recognised as a global health leader who has used his extensive network and experience to champion both Gavi and immunisation.
::::::
What does COVAX’s latest supply forecast tell us?
23 June 2021 – by Aurélia Nguyen, Managing Director, Office of the COVAX Facility
COVAX’s latest supply forecast is now published. According to this latest assessment, the vaccines pillar of the ACT-Accelerator will have 1.9 billion doses available for supply by the end of 2021 and, thanks to a number of recently signed advance purchase agreements, now has signed deals in place for a total of 2.8 billion doses in 2021 and 2022, with a further 1.3 billion secured should it need them. This is due in large part to the pledges made by donors to the Gavi COVAX AMC.
The forecast is encouraging given the supply disruptions COVAX and countries with bilateral deals are currently experiencing, as well as the general challenges manufacturers are experiencing as they ramp up supply at historic speed and scale. But what does it mean in the context of COVAX’s stated goal of ending the acute phase of the pandemic? Unsurprisingly, given that it represents the most ambitious roll-out of vaccines in history, the numbers in the document provide some important insights.
Short term supply concerns persist
The first observation is that, despite remaining more or less on target for its original year-end goal to make 2 billion doses available to participants, COVAX deliveries will continue to be very lean through July and August. This is a result of a number of well-documented factors, including the diversion of production at the Serum Institute of India (SII), an important early supplier, to meet acute domestic needs, as well as challenges scaling up deliveries across AstraZeneca’s global manufacturing network and fulfilling requirements to enable delivery of the Pfizer vaccine – COVAX’s other two currently active suppliers.
Dose sharing is becoming an important immediate-term source of doses
In response to COVAX’s call to countries with excess supply to help alleviate short term supply shortages by sharing doses, a number of donors have now stepped forward. According to the forecast, 515 million doses have been pledged, and they should start becoming available in significant numbers very soon, with 80 million expected by the end of July and a total of 115 million by the end of August. Apart from the period between September and October when 100 million dose donations are expected to come online, supply from countries’ pledges are anticipated to be relatively evenly paced through the year, with around 40-50 million donated doses available each month until the end of December.
Absorptive capacity is a very real concern
The reason why COVAX has been urging donors to share doses in the third quarter rather than later in the year is to avoid the possibility of COVAX participants’ health systems becoming too stretched by the volume of deliveries later in the year. COVAX’s partners work closely with countries in the Facility, especially the 92 lower-income economies eligible for support through the Gavi COVAX AMC, to ensure conditions are in place to enable mass vaccination. From September onwards, the forecast projects that a number in the region of 400 million doses will become available every month so it’s critical that sufficient funding is unlocked, where necessary, to help countries prepare for this.
COVAX will keep on doing deals
COVAX has currently signed deals for 2.8 billion doses. This is more than enough to fulfil our goal of delivering 1.8 billion doses to AMC economies as well as supplying the approximately 480 million doses ordered by self-financing economies. A key reason COVAX has signed deals and options for more doses is to allow participants that want to receive more doses to procure them through cost-sharing; taking advantage of COVAX’s pricing and access, plus other advantages such as its no-fault compensation scheme, to raise protection rates in their countries further. COVAX’s goal from the beginning has also been to build a portfolio of 10-12 vaccines to ensure suitability for all contexts and to mitigate risks around over-reliance on specific sources in case there are issues related to failure at the R&D stage, regulatory hurdles or supply challenges. Gavi, on behalf of the Facility, also negotiates key flexibilities such as options and potential access to future variant-adapted vaccines to ensure the Facility portfolio can continue to be tailored to participants’ evolving needs. To this end, expect further advance purchase agreements to be announced in the coming weeks and months.
Mind the gap (between availability and deliveries)
One important distinction between the supply forecast and COVAX’s delivery tracker, which is hosted by COVAX’s key delivery partner UNICEF, is that the forecast tracks the availability of doses at the manufacturer. With a number of technical, regulatory and legal tasks to be overcome before doses can be released, there is always a delay between doses becoming available and being shipped and delivered to countries. Although COVAX partners work closely with countries, manufacturers and other stakeholders to minimise this gap, the estimated timings shown here reflect the availability of doses at the manufacturer and not in country.
The Supply Forecast will continue to evolve
There are a number of other caveats worth noting in the forecast too, all of which relate to the scale and number of moving pieces across COVAX’s unprecedented end-to-end solution. The timing of regulatory approval, for example, can be volatile, especially since it is common that more than one manufacturing site is involved in the supply chain. Likewise local licensing or export controls can and have had an impact on supply schedules. For these reasons as well as the fact that more secured doses are expected to be added to its portfolio, COVAX will aim to keep this forecast updated as regularly as possible through 2021 and beyond. Watch this space