Lancet Global Health
Feb 2021 Volume 9 Number 2 e99-e217
https://www.thelancet.com/journals/langlo/issue/current
Comment
What could fair allocation of an efficacious COVID-19 vaccine look like in South Africa?
Keymanthri Moodley, Theresa Rossouw
Lancet Global Health
Feb 2021 Volume 9 Number 2 e99-e217
https://www.thelancet.com/journals/langlo/issue/current
Comment
What could fair allocation of an efficacious COVID-19 vaccine look like in South Africa?
Keymanthri Moodley, Theresa Rossouw
Lancet Global Health
Feb 2021 Volume 9 Number 2 e99-e217
https://www.thelancet.com/journals/langlo/issue/current
Articles
Global Fund contributions to health security in ten countries, 2014–20: mapping synergies between vertical disease programmes and capacities for preventing, detecting, and responding to public health emergencies
Matthew R Boyce, Aurelia Attal-Juncqua, Jessica Lin, Stephanie McKay, Rebecca Katz
Lancet Infectious Diseases
Feb 2021 Volume 21 Number 2 p149-296, e16-e35
https://www.thelancet.com/journals/laninf/issue/current
Editorial
An exceptional vaccination policy in exceptional circumstances
The Lancet Infectious Diseases
Just as hope for relief from the COVID-19 pandemic brightened in December, with emergency use approval for vaccines following phase 3 trials and the start of immunisation programmes, unprecedented numbers of cases, hospital admissions, and deaths have been recorded, including in countries such as Germany and Japan that were previously celebrated for their public health response. Predictions that the northern hemisphere winter would be a difficult time for control of COVID-19, as is typically the case with respiratory viral diseases, have proved to be correct. Emergence in South Africa and the UK of new variants of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) that are substantially more transmissible, has added to concerns that health services will be overwhelmed.
Alarm over the rapid rise in cases led to an unexpected policy twist in the UK on Dec 30: on top of a third national lockdown, the chief medical officers of the four nations of the UK announced that the second dose of vaccines against SARS-CoV-2 should be given at up to 12 weeks after the first dose rather than the recommended interval of 3–4 weeks, on the basis of advice from the Joint Committee on Vaccination and Immunisation (JCVI). While supplies of vaccine remain limited, the JCVI recommends “initially prioritising delivery of the first vaccine dose as this is highly likely to have a greater public health impact in the short term and reduce the number of preventable deaths from COVID-19”. The rationale is that if 95% of people are protected from disease after two doses (as determined in the phase 3 trial of the Pfizer-BioNTech vaccine) and 90% after one dose (JCVI estimate), then 19 of 20 people will be protected by two doses but when given as single doses to 40 people 36 will be protected. The WHO Strategic Advisory Group of Experts (SAGE) on immunisation has advised that the interval between doses of the Pfizer-BioNTech vaccine can be up to 6 weeks in “exceptional epidemiological circumstances”. The Canadian province of British Columbia will extend the vaccine interval to 5 weeks, and other countries are reported to be considering extending the dosing interval while awaiting delivery of more vaccine.
Given that vaccine approval is based on a dosing interval of 3 or 4 week, that efficacy of single doses was not formally tested in randomised trials, and that estimated efficacy of single doses is based on unplanned analyses of few data from these trials, the decision to prioritise the first dose has naturally drawn criticism. The British Medical Association described the decision as “unreasonable and totally unfair”, pointing out the logistical difficulties in rebooking vaccination appointment for the elderly and vulnerable people who have already received their first immunisation. Anthony Fauci, director of the US National Institute of Allergy and Infectious Diseases, said that the US vaccination programme would proceed with the approved two-dose schedule, noting the lack of data on extending the period to the second immunisation.
An important concern is the durability of immunity in the extended dosing interval. Some COVID-19 cases after the first vaccine dose are inevitable among people infected around the time of immunisation—as was observed in clinical trials—because protective immunity takes about 2 weeks to become established. However, if immunity wanes during the interval between doses, post-vaccination infections will be more common, thus reducing the impact of—and public confidence in—the vaccination programme. As John Roberts and colleagues point out, duration of immunity to viral spike protein induced by mRNA vaccines such as the Pfizer-BioNTech and Moderna ones is unknown, and in animal models neutralising antibody production falls at about 28 days from first vaccination. By contrast, these authors note that for viral-vector vaccines (eg, Oxford-AstraZeneca ChAdOx1), there is evidence to support an extended interval between doses. Another unknown is whether potentially suboptimal immunity will foster evolution of vaccine-escape mutants of SARS-CoV-2. In this regard, some reassurance comes from a report that the Pfizer-BioNTech vaccine produces antibodies that neutralise SARS-CoV-2 carrying the N501Y mutation found in the South African and UK variants.
Independent SAGE, a group of UK scientists not known for their support of government COVID-19 policy, has endorsed the decision “to pursue coverage of as high a proportion of the population as possible, as quickly as possible”, noting that it must be part of a comprehensive strategy that includes prospective evaluation of different dosing intervals. Given the urgent need to suppress a rise in cases caused by a new viral variant, we believe that extending the dosing interval is an expedient, short-term decision while there are constraints on vaccine supply.
Lancet Infectious Diseases
Feb 2021 Volume 21 Number 2 p149-296, e16-e35
https://www.thelancet.com/journals/laninf/issue/current
Articles
Safety, tolerability, and immunogenicity of an inactivated SARS-CoV-2 vaccine in healthy adults aged 18–59 years: a randomised, double-blind, placebo-controlled, phase 1/2 clinical trial
Yanjun Zhang, et al
Lancet Infectious Diseases
Feb 2021 Volume 21 Number 2 p149-296, e16-e35
https://www.thelancet.com/journals/laninf/issue/current
Safety, immunogenicity, and efficacy of a Clostridioides difficile toxoid vaccine candidate: a phase 3 multicentre, observer-blind, randomised, controlled trial
Guy de Bruyn, et al
Lancet Infectious Diseases
Feb 2021 Volume 21 Number 2 p149-296, e16-e35
https://www.thelancet.com/journals/laninf/issue/current
Lancet Infectious Diseases
Feb 2021 Volume 21 Number 2 p149-296, e16-e35
https://www.thelancet.com/journals/laninf/issue/current
Safety and immunogenicity of co-administered hookworm vaccine candidates Na-GST-1 and Na-APR-1 in Gabonese adults: a randomised, controlled, double-blind, phase 1 dose-escalation trial
Ayola A Adegnika, et al for the HookVac Consortium
Lancet Infectious Diseases
Feb 2021 Volume 21 Number 2 p149-296, e16-e35
https://www.thelancet.com/journals/laninf/issue/current
Immunogenicity and persistence of trivalent measles, mumps, and rubella vaccines: a systematic review and meta-analysis
Julie Schenk, et al
Lancet Infectious Diseases
Feb 2021 Volume 21 Number 2 p149-296, e16-e35
https://www.thelancet.com/journals/laninf/issue/current
Review
What defines an efficacious COVID-19 vaccine? A review of the challenges assessing the clinical efficacy of vaccines against SARS-CoV-2
Susanne H Hodgson, et al
Lancet Public Health
Feb 2021 Volume 6 Number 2 e82-e135
https://www.thelancet.com/journals/lanpub/issue/current
Editorial
COVID-19—break the cycle of inequality
The Lancet Public Health
Lancet Respiratory Medicine
Feb 2021 Volume 9 Number 2 p117-216, e11-e21
https://www.thelancet.com/journals/lanres/issue/current
Editorial
Realising the potential of SARS-CoV-2 vaccines—a long shot?
The Lancet Respiratory Medicine
… The ability of SARS-CoV-2 vaccines to prevent infection or ongoing transmission remains unclear. The impact of immunisation on hospital admissions will be an important focus as countries aim to keep health-care systems running and protect those most at risk of severe disease in the face of high levels of transmission. Emerging SARS-CoV-2 variants are a cause for concern. New variants include B.1.1.7, which recently emerged in the UK, B.1.351, and P.1. These lineages appear to be more transmissible than previous strains, although there is currently no evidence that they cause more severe disease or compromise the efficacy of available vaccines. Surveillance is needed to detect escape variants at an early stage. New variants also emphasise the ongoing need for public health mitigation strategies and add to the urgency of vaccine rollout across the globe…
Nature
Volume 590 Issue 7844, 4 February 2021
http://www.nature.com/nature/current_issue.html
Article | 02 November 2020
Age-specific mortality and immunity patterns of SARS-CoV-2
The relative risk of COVID-19-associated death for younger individuals (under 65) is consistent across countries and can be used to robustly compare the underlying number of infections in each country.
Megan O’Driscoll, Gabriel Ribeiro Dos Santos[…] & Henrik Salje
Nature Reviews Immunology
Volume 21 Issue 2, February 2021
https://www.nature.com/nri/volumes/21/issues/2
Progress | 18 December 2020
Viral targets for vaccines against COVID-19
As the world races to develop vaccines against SARS-CoV-2, Dai and Gao highlight which viral targets are best to include in a vaccine and how this impacts the induced immune response and, ultimately, the safety and efficacy of a vaccine.
Lianpan Dai & George F. Gao
Nature Reviews Immunology
Volume 21 Issue 2, February 2021
https://www.nature.com/nri/volumes/21/issues/2
Review Article | 22 December 2020
A guide to vaccinology: from basic principles to new developments
This Review, aimed at a broad scientific audience, provides an introductory guide to the history, development and immunological basis of vaccines, immunization and related issues to provide insight into the challenges facing immunologists who are designing the next generation of vaccines.
Andrew J. Pollard & Else M. Bijker
Nature Reviews Drug Discovery
Volume 20 Issue 2, February 2021
https://www.nature.com/nrd/volumes/20/issues/2
Review Article | 22 December 2020
Image-based profiling for drug discovery: due for a machine-learning upgrade?
Image-based profiling is a strategy to mine the rich information in biological images. Carpenter and colleagues discuss how the application of machine learning is renewing interest in image-based profiling for all aspects of the drug discovery process, from understanding disease mechanisms to predicting a drug’s activity or mechanism of action.
Srinivas Niranj Chandrasekaran, Hugo Ceulemans […] & Anne E. Carpenter
New England Journal of Medicine
February 4, 2021 Vol. 384 No. 5
http://www.nejm.org/toc/nejm/medical-journal
Perspective
A Half-Century of Progress in Health: The National Academy of Medicine at 50
Vaccine Innovations — Past and Future
Julie L. Gerberding, M.D., M.P.H., and Barton F. Haynes, M.D.
… Vaccines remain the most effective tool for preventing infectious diseases and improving global health. Remarkable progress has been made with the use of vaccines, including the eradication of smallpox and the control of childhood diseases such as measles, mumps, rubella, and polio. New insights into the functioning of the immune system on a cellular and molecular level have made possible the rapid development of new vaccines. Difficulties facing vaccinologists include predicting the type and timing of the next pandemic; developing vaccines to combat rapidly changing pathogens such as HIV-1, influenza, and multidrug-resistant bacteria; and establishing rapid-response strategies to control emerging and reemerging infectious diseases. The future holds great promise for vaccine-mediated control of global pathogens, but providing affordable access to effective vaccines for everyone who could benefit from them remains an important challenge.
Pediatrics
Vol. 147, Issue 2 1 Feb 2021
https://pediatrics.aappublications.org/
Research Briefs
Parental Vaccine Hesitancy and Vaccination Disparities in a Safety-Net System
Joshua T.B. Williams, John D. Rice, Yingbo Lou, Elizabeth A. Bayliss, Steven G. Federico, Simon J. Hambidge, Sean T. O’Leary
Pediatrics, Feb 2021, 147 (2) e2020010710
PLoS Genetics
https://journals.plos.org/plosgenetics/
(Accessed 6 Feb 2021)
Creating artificial human genomes using generative neural networks
Burak Yelmen, Aurélien Decelle, Linda Ongaro, Davide Marnetto, Corentin Tallec, Francesco Montinaro, Cyril Furtlehner, Luca Pagani, Flora Jay
Research Article | published 04 Feb 2021 PLOS Genetics
https://doi.org/10.1371/journal.pgen.1009303
Abstract
Generative models have shown breakthroughs in a wide spectrum of domains due to recent advancements in machine learning algorithms and increased computational power. Despite these impressive achievements, the ability of generative models to create realistic synthetic data is still under-exploited in genetics and absent from population genetics. Yet a known limitation in the field is the reduced access to many genetic databases due to concerns about violations of individual privacy, although they would provide a rich resource for data mining and integration towards advancing genetic studies. In this study, we demonstrated that deep generative adversarial networks (GANs) and restricted Boltzmann machines (RBMs) can be trained to learn the complex distributions of real genomic datasets and generate novel high-quality artificial genomes (AGs) with none to little privacy loss. We show that our generated AGs replicate characteristics of the source dataset such as allele frequencies, linkage disequilibrium, pairwise haplotype distances and population structure. Moreover, they can also inherit complex features such as signals of selection. To illustrate the promising outcomes of our method, we showed that imputation quality for low frequency alleles can be improved by data augmentation to reference panels with AGs and that the RBM latent space provides a relevant encoding of the data, hence allowing further exploration of the reference dataset and features for solving supervised tasks. Generative models and AGs have the potential to become valuable assets in genetic studies by providing a rich yet compact representation of existing genomes and high-quality, easy-access and anonymous alternatives for private databases.
Author summary
Generative neural networks have been effectively used in many different domains in the last decade, including machine dreamt photo-realistic imagery. In our work, we apply a similar concept to genetic data to automatically learn its structure and, for the first time, produce high quality realistic genomes. These novel genomes are distinct from the original ones used for training the generative networks. We show that artificial genomes, as we name them, retain many complex characteristics of real genomes and the heterogeneous relationships between individuals. They can be used in intricate analyses such as imputation of missing data as we demonstrated. We believe they have a high potential to become alternatives for many genome databases which are not publicly available or require long application procedures or collaborations and remove an important accessibility barrier in genomic research in particular for underrepresented populations.
PLoS One
http://www.plosone.org/
[Accessed 6 Feb 2021]
Knowledge, attitudes, and practices of seasonal influenza vaccination in healthcare workers, Honduras
Zachary J. Madewell, Rafael Chacón-Fuentes, Jorge Jara, Homer Mejía-Santos, Ida-Berenice Molina, Juan Pablo Alvis-Estrada, Maria-Renee Ortiz, Rosa Coello-Licona, Belinda Montejo
Research Article | published 04 Feb 2021 PLOS ONE
https://doi.org/10.1371/journal.pone.0246379
PLoS One
http://www.plosone.org/
[Accessed 6 Feb 2021]
Mental health and psychosocial support strategies in highly contagious emerging disease outbreaks of substantial public concern: A systematic scoping review
Angela M. Kunzler, Jutta Stoffers-Winterling, Marlene Stoll, Alexander L. Mancini, Sophie Lehmann, Manpreet Blessin, Donya Gilan, Isabella Helmreich, Frank Hufert, Klaus Lieb
Research Article | published 03 Feb 2021 PLOS ONE
https://doi.org/10.1371/journal.pone.0244748
PLoS One
http://www.plosone.org/
[Accessed 6 Feb 2021]
Ethics of emerging infectious disease outbreak responses: Using Ebola virus disease as a case study of limited resource allocation
Ariadne A. Nichol, Annick Antierens
Research Article | published 02 Feb 2021 PLOS ONE
https://doi.org/10.1371/journal.pone.0246320
Abstract
Emerging infectious diseases such as Ebola Virus Disease (EVD), Nipah Virus Encephalitis and Lassa fever pose significant epidemic threats. Responses to emerging infectious disease outbreaks frequently occur in resource-constrained regions and under high pressure to quickly contain the outbreak prior to potential spread. As seen in the 2020 EVD outbreaks in the Democratic Republic of Congo and the current COVID-19 pandemic, there is a continued need to evaluate and address the ethical challenges that arise in the high stakes environment of an emerging infectious disease outbreak response. The research presented here provides analysis of the ethical challenges with regard to allocation of limited resources, particularly experimental therapeutics, using the 2013–2016 EVD outbreak in West Africa as a case study. In-depth semi-structured interviews were conducted with senior healthcare personnel (n = 16) from international humanitarian aid organizations intimately engaged in the 2013–2016 EVD outbreak response in West Africa. Interviews were recorded in private setting, transcribed, and iteratively coded using grounded theory methodology. A majority of respondents indicated a clear propensity to adopt an ethical framework of guiding principles for international responses to emerging infectious disease outbreaks. Respondents agreed that prioritization of frontline workers’ access to experimental therapeutics was warranted based on a principle of reciprocity. There was widespread acceptance of adaptive trial designs and greater trial transparency in providing access to experimental therapeutics. Many respondents also emphasized the importance of community engagement in limited resource allocation scheme design and culturally appropriate informed consent procedures. The study results inform a potential ethical framework of guiding principles based on the interview participants’ insights to be adopted by international response organizations and their healthcare workers in the face of allocating limited resources such as experimental therapeutics in future emerging infectious disease outbreaks to ease the moral burden of individual healthcare providers.
Preventive Medicine
Volume 143 February 2021
https://www.sciencedirect.com/journal/preventive-medicine/vol/143/suppl/C
Discussion Abstract only
Core functions, knowledge bases and essential services: A proposed prescription for the evolution of the preventive medicine specialty
Yuri T. Jadotte, Dorothy S. Lane
Article 106286
Preventive Medicine
Volume 143 February 2021
https://www.sciencedirect.com/journal/preventive-medicine/vol/143/suppl/C
Review article Open access
Cognitions and behaviours of general practitioners in France regarding HPV vaccination: A theory-based systematic review
Géraldine Escriva-Boulley, Olena Mandrik, Marie Préau, Rolando Herrero, Patricia Villain
Article 106323
Preventive Medicine
Volume 143 February 2021
https://www.sciencedirect.com/journal/preventive-medicine/vol/143/suppl/C
Research article Abstract only
Low human papillomavirus (HPV) vaccine uptake among men living with human immunodeficiency virus (HIV): Cross-sectional findings from a clinical cohort
Ramandip Grewal, Troy Grennan, Jennifer L. Gillis, Gina Ogilvie, … Ann N. Burchell
Article 106329
Qualitative Health Research
Volume 31 Issue 3, February 2021
http://qhr.sagepub.com/content/current
Research Articles
Acceptance of the HPV Vaccine in a Multiethnic Sample of Latinx Mothers
Ana Cristina Lindsay, Madelyne J. Valdez, Denisse Delgado, Emily Restrepo, Yessica M. Guzmán, Phillip Granberry
First Published January 10, 2021; pp. 472–483
Qualitative Health Research
Volume 31 Issue 3, February 2021
http://qhr.sagepub.com/content/current
Ethics
Ethics Review Boards for Research With Human Participants: Past, Present, and Future
Maya Peled-Raz, Shay S. Tzafrir, Guy Enosh, Yael Efron, Israel (Issi) Doron
First Published November 19, 2020; pp. 590–599
Abstract
The debate around ethics review boards (IRBs) has assumed an increasingly central place in academic practice and discourse. In this article, we summarize a unique workshop (study-group) that convened at the University of Haifa, attended by 27 academics from around the globe, representing nine countries in four continents. The participants presented data and points of view, which served as the basis for an open, interdisciplinary discussion. The group developed a set of recommendations, including working toward a transition from a review system to an advisory and validation system; focusing on respectful research approach to participants, rather than “ethical” research; building a procedure that focuses on feedback, rather than the process itself; recognizing that a unified examination need not necessarily be standardized; and constructing a feedback procedure in which researchers can respond to the review of their research
Revista Panamericana de Salud Pública/Pan American Journal of Public Health (RPSP/PAJPH)
https://www.paho.org/journal/en
5 Feb 2021
Evaluation of the first level of care for tuberculosis control in Buenos Aires, Argentina*
Original research | English |
Science
05 February 2021 Vol 371, Issue 6529
http://www.sciencemag.org/current.dtl
Special Issue: Human Genome at 20
Editorial
A genome to celebrate
By Claire M. Fraser
Science
05 February 2021 Vol 371, Issue 6529
http://www.sciencemag.org/current.dtl
Feature
Genomes arising
By Elizabeth Pennisi
Science05 Feb 2021 : 556-559 Full Access
Africans have begun to study their continent’s rich human diversity—but what comes after current grants end?
Science
05 February 2021 Vol 371, Issue 6529
http://www.sciencemag.org/current.dtl
Letters
Beyond DNA: The rest of the story
By Gregg Duncan, Yongsheng Ji, Marie-Caroline Lefort, Ruty Mehrian-Shai, Prosanta Chakrabarty, Maria Fernanda Torres Jimenez
Science
05 February 2021 Vol 371, Issue 6529
http://www.sciencemag.org/current.dtl
Policy Forum
Complicated legacies: The human genome at 20
By Kathryn Maxson Jones, Robert Cook-Deegan, Charles N. Rotimi, Shawneequa L. Callier, Amy R. Bentley, Hallam Stevens, Kathryn A. Phillips, Jeroen P. Jansen, Christopher F. Weyant, Dorothy E. Roberts, Dina Zielinski, Yaniv Erlich, Nanibaa’ A. Garrison, Stephanie Russo Carroll, Pilar N. Ossorio, Yves Moreau, Maya Wang
Science Translational Medicine
03 February 2021 Vol 13, Issue 579
https://stm.sciencemag.org/
Review
Development and deployment of COVID-19 vaccines for those most vulnerable
By Wayne C. Koff, Theodore Schenkelberg, Tere Williams, Ralph S. Baric, Adrian McDermott, Cheryl M. Cameron, Mark J. Cameron, Matthew B. Friemann, Gabriele Neumann, Yoshihiro Kawaoka, Alyson A. Kelvin, Ted M. Ross, Stacey Schultz-Cherry, Timothy D. Mastro, Frances H. Priddy, Kristine A. Moore, Julia T. Ostrowsky, Michael T. Osterholm, Jaap Goudsmit
Science Translational Medicine03 Feb 2021 Full Access
Ensuring that COVID-19 vaccines are delivered to older populations and those living in low resource settings is essential to end the pandemic.
Abstract
Development of safe and effective COVID-19 vaccines is a global priority and the best hope for ending the COVID-19 pandemic. Remarkably, in less than 1 year, vaccines have been developed and shown to be efficacious and are already being deployed worldwide. Yet, many challenges remain. Immune senescence and comorbidities in aging populations and immune dysregulation in populations living in low-resource settings may impede vaccine effectiveness. Distribution of vaccines among these populations where vaccine access is historically low remains challenging. In this Review, we address these challenges and provide strategies for ensuring that vaccines are developed and deployed for those most vulnerable.
Vaccine
Volume 39, Issue 7 Pages 1025-1172 (12 February 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/7
Discussion Full text access
Development of vaccines for influenza disease: Opportunity costs of the COVID-19 pandemic
Timothy L. Wiemken, Ana Santos Rutschman
Pages 1025-1026
Vaccine
Volume 39, Issue 7 Pages 1025-1172 (12 February 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/7
Correspondence Full text access
Ethical tradeoffs in SARS-CoV-2 vaccine development: Assuring fair availability for low-income countries
David Gurwitz
Vaccine
Volume 39, Issue 7 Pages 1025-1172 (12 February 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/7
Correspondence No access
Ethical trade-offs in vaccine development and distribution—Response to Gurwitz
Annette Rid, Seema K. Shah, Franklin G. Miller, Marion Danis, … Christine Grady
Pages 1028-1029
Vaccine
Volume 39, Issue 7 Pages 1025-1172 (12 February 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/7
Research article Full text access
Predictors of intention to vaccinate against COVID-19: Results of a nationwide survey
Jeanette B. Ruiz, Robert A. Bell
Vaccine
Volume 39, Issue 7 Pages 1025-1172 (12 February 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/7
Research article Open access
Acceptance of the COVID-19 vaccine based on the health belief model: A population-based survey in Hong Kong
Martin C.S. Wong, Eliza L.Y. Wong, Junjie Huang, Annie W.L. Cheung, … Paul K.S. Chan
Vaccines — Open Access Journal
http://www.mdpi.com/journal/vaccines
(Accessed 6 Feb 2021)
Open Access Article
Intention to Receive the COVID-19 Vaccination in China: Application of the Diffusion of Innovations Theory and the Moderating Role of Openness to Experience
by Phoenix Kit-han Mo, Sitong Luo, Suhua Wang, Junfeng Zhao, Guohua Zhang, Lijuan Li, Liping Li, Luyao Xie and Joseph T. F. Lau
Vaccines 2021, 9(2), 129; https://doi.org/10.3390/vaccines9020129 (registering DOI) – 05 Feb 2021
Abstract
COVID-19 has caused a devastating impact on public health and made the development of the COVID-19 vaccination a top priority. Herd immunity through vaccination requires a sufficient number of the population to be vaccinated. Research on factors that promote intention to receive the […]
Vaccines — Open Access Journal
http://www.mdpi.com/journal/vaccines
(Accessed 6 Feb 2021)
Open Access Viewpoint
Why It Is Important to Develop an Effective and Safe Pediatric COVID-19 Vaccine
by Nicola Principi and Susanna Esposito
Vaccines 2021, 9(2), 127; https://doi.org/10.3390/vaccines9020127 – 05 Feb 2021
Abstract
The need to cope with the medical, social, and economic storm due to the new coronavirus 2019 (COVID-19) pandemic as quickly as possible has led to the very rapid development of a huge number of vaccines. All these vaccines have been mainly developed […]
Vaccines — Open Access Journal
http://www.mdpi.com/journal/vaccines
(Accessed 6 Feb 2021)
Open Access Article
COVID-19 Vaccine Acceptance among Health Care Workers in the United States
by ahul Shekhar, Abu Baker Sheikh, Shubhra Upadhyay, Mriganka Singh, Saket Kottewar, Hamza Mir,
Eileen Barrett and Suman Pal
Vaccines 2021, 9(2), 119; https://doi.org/10.3390/vaccines9020119 – 03 Feb 2021
Abstract
Background: Acceptance of the COVID-19 vaccine will play a major role in combating the pandemic. Healthcare workers (HCWs) are among the first group to receive vaccination, so it is important to consider their attitudes about COVID-19 vaccination to better address barriers to widespread […]
Vaccines — Open Access Journal
http://www.mdpi.com/journal/vaccines
(Accessed 6 Feb 2021)
Open Access Review
Ethics of Vaccination in Childhood—A Framework Based on the Four Principles of Biomedical Ethics
by Meta Rus and Urh Groselj
Vaccines 2021, 9(2), 113; https://doi.org/10.3390/vaccines9020113 – 02 Feb 2021
Abstract
Although vaccination is recognised as the top public health achievement of the twentieth century, unequivocal consensus about its beneficence does not exist among the general population. In countries with well-established immunisation programmes, vaccines are “victims of their own success”, because low incidences of diseases now prevented with vaccines diminished the experience of their historical burdens. Increasing number of vaccine-hesitant people in recent years threatens, or even effectively disables, herd immunity levels of the population and results in outbreaks of previously already controlled diseases. We aimed to apply a framework for ethical analysis of vaccination in childhood based on the four principles of biomedical ethics (respect for autonomy, nonmaleficence, beneficence and justice) to provide a comprehensive and applicable model on how to address the ethical aspects of vaccination at both individual and societal levels. We suggest finding an “ethical equilibrium”, which means that the degree of respect for parents’ autonomy is not constant, but variable; it shall depend on the level of established herd immunity and it is specific for every society. When the moral obligation of individuals to contribute to herd immunity is not fulfilled, mandatory vaccination policies are ethically justified, because states bear responsibility to protect herd immunity as a common good.
Vaccines — Open Access Journal
http://www.mdpi.com/journal/vaccines
(Accessed 6 Feb 2021)
Open Access Communication
The Model of “Informed Refusal” for Vaccination: How to Fight against Anti-Vaccinationist Misinformation without Disregarding the Principle of Self-Determination
by Stefano D’Errico, Emanuela Turillazzi, Martina Zanon, Rocco Valerio Viola, Paola Frati and Vittorio Fineschi
Vaccines 2021, 9(2), 110; https://doi.org/10.3390/vaccines9020110 – 01 Feb 2021
Abstract
Vaccines are arguably a public health success story as well as an incredibly cost-effective medical resource. Despite this, worldwide concerns about their safety are growing, with the risk of increased morbidity and mortality in vaccine-preventable diseases because of vaccine refusal. The global political
Media/Policy Watch
This watch section is intended to alert readers to substantive news, analysis and opinion from the general media and selected think tanks and similar organizations on vaccines, immunization, global public health and related themes. Media Watch is not intended to be exhaustive, but indicative of themes and issues CVEP is actively tracking. This section will grow from an initial base of newspapers, magazines and blog sources, and is segregated from Journal Watch above which scans the peer-reviewed journal ecology.
We acknowledge the Western/Northern bias in this initial selection of titles and invite suggestions for expanded coverage. We are conservative in our outlook in adding news sources which largely report on primary content we are already covering above. Many electronic media sources have tiered, fee-based subscription models for access. We will provide full-text where content is published without restriction, but most publications require registration and some subscription level.
The Atlantic
http://www.theatlantic.com/magazine/
Accessed 6 Feb 2021
[No new, unique, relevant content]
BBC
http://www.bbc.co.uk/
Accessed 6 Feb 2021
[No new, unique, relevant content]
The Economist
http://www.economist.com/
Accessed 6 Feb 2021
[No new, unique, relevant content]
Financial Times
https://www.ft.com/
Accessed 6 Feb 2021
Covid-19 vaccines
Oxford/AstraZeneca jab fails to prevent mild and moderate Covid from S African strain, study shows
Impact on hospitalisations and deaths caused by variant not yet determined, according to preliminary findings
February 6, 2021
The Oxford/AstraZeneca Covid-19 vaccine does not appear to offer protection against mild and moderate disease caused by the viral variant first identified in South Africa, according to a study due to be published on Monday. Although none of the more than 2,000 mainly healthy and young patients in the study died or was hospitalised, the findings, which have not yet been peer reviewed, could complicate the race to roll out vaccines as new strains emerge. In both the human trials and tests on the blood of those vaccinated, the jab showed significantly reduced efficacy against the 501Y.V2 viral variant, which is dominant in South Africa, according to the randomised, double-blind study seen by the Financial Times…
Coronavirus Business Update
Global vaccinations surpass new infections
February 3, 2021
Top of Form
Bottom of Form
Forbes
http://www.forbes.com/
Accessed 6 Feb 2021
[No new, unique, relevant content]
Foreign Affairs
http://www.foreignaffairs.com/
Accessed 6 Feb 2021
[No new, unique, relevant content]
Foreign Policy
http://foreignpolicy.com/
Accessed 6 Feb 2021
[No new, unique, relevant content]
The Guardian
http://www.guardiannews.com/
Accessed 6 Feb 2021
[No new, unique, relevant content]
New Yorker
http://www.newyorker.com/
Accessed 6 Feb 2021
[No new, unique, relevant content]
New York Times
http://www.nytimes.com/
Accessed 6 Feb 2021
U.S.
A New Front in the Anti-Vaccine Fight Emerges in California
For months, far-right activists have rallied against masks and lockdowns imposed during the coronavirus pandemic. Now some protesters have shifted their focus to the Covid-19 vaccine.
By Manny Fernandez 6 Feb 2021
Real Estate
Can My Building Mandate Vaccinations for Workers and Staff?
There will be plenty of legal hurdles, but some buildings are beginning to think about a future when mandatory vaccines are possible.
By Ronda Kaysen 6 Feb 2021
Technology
Russian Campaign Promotes Homegrown Vaccine and Undercuts Rivals
Russian news outlets with connections to the Kremlin ran disingenuous Spanish-language stories targeting vaccines made in the United States and Europe, researchers said.
By Sheera Frenkel, Maria Abi-Habib and Julian E. Barnes Feb. 5
Middle East
Israel’s Vaccination Results Point a Way Out of Virus Pandemic
Covid cases fell dramatically and quickly among people who were vaccinated, Israeli studies found. It’s the strongest evidence yet that a robust vaccination program can tame the pandemic.
By Isabel Kershner and Carl Zimmer 5 Feb 2021
Washington Post
https://www.washingtonpost.com/
Accessed 6 Feb 2021
With Carnival scrapped, Rio’s Sambadrome hosts vaccinations
· Feb 6, 2021
Think Tanks et al
Brookings
http://www.brookings.edu/
Accessed 6 Feb 2021
TechTank
Five ways that health information exchange can help with vaccination efforts
Niam Yaraghi and Peter L. Levin Monday, February 1, 2021
Center for Global Development [to 6 Feb 2021]
http://www.cgdev.org/page/press-center
February 4, 2021
Five Factors that Drive Successful Large-Scale Vaccine Distribution
Five key operational levers and choices determine how fast countries are able to roll out the COVID-19 vaccine. Ideally, someone at the national level makes these decisions, but this hasn’t always been easy in multi-level government systems.
Prashant Yadav
Chatham House [to 6 Feb 2021]
https://www.chathamhouse.org/
Accessed 6 Feb 2021
[No new relevant content]
CSIS
https://www.csis.org/
Accessed 6 Feb 2021
Upcoming Event
Online Event: A Global Approach to Covid-19 Vaccination
February 10, 2021
Transcript
Online Event: The State of Immunization Under Covid-19
February 3, 2021
Council on Foreign Relations
http://www.cfr.org/
Accessed 6 Feb 2021
February 5, 2021
Pharmaceuticals and Vaccines
A Guide to Global COVID-19 Vaccine Efforts
Only a year after the new coronavirus emerged, the first vaccines to protect against it are being administered. But production challenges, vaccine nationalism, and new virus strains are all presentin…
Backgrounder by Claire Felter
Kaiser Family Foundation
https://www.kff.org/search/?post_type=press-release
Accessed 6 Feb 2021
February 3, 2021 News Release
Data as of February 1 on State Vaccinations by Race/Ethnicity
Updated analysis of state-reported data as of February 1, 2021 on COVID-19 vaccinations, cases, and deaths by race/ethnicity is now available. Based on nearly half of states reporting vaccination data by race/ethnicity, Black and Hispanic people continue to receive smaller shares of vaccinations compared to their shares of cases and…
February 3, 2021 News Release
Vaccine Monitor: Where People are Getting Information About COVID-19 Vaccinations
Additional Data Available Through Online Vaccine Monitor Dashboard As vaccination efforts ramp up across the country, the KFF COVID-19 Vaccine Monitor examines where the public is getting its relevant information. Large shares of the public report that they are getting at least a fair amount of vaccine information from television…
World Economic Forum [to 6 Feb 2021]
https://agenda.weforum.org/news/
Media
[No new relevant content]
Vaccines and Global Health: The Week in Review is a weekly digest summarizing news, events, announcements, peer-reviewed articles and research in the global vaccine ethics and policy space. Content is aggregated from key governmental, NGO, international organization and industry sources, key peer-reviewed journals, and other media channels. This summary proceeds from the broad base of themes and issues monitored by the Center for Vaccine Ethics & Policy in its work: it is not intended to be exhaustive in its coverage. You are viewing the blog version of our weekly digest, typically comprised of between 30 and 40 posts below all dated with the current issue date
.– Request an Email Summary: Vaccines and Global Health : The Week in Review is published as a single email summary, scheduled for release each Saturday evening before midnight (EDT in the U.S.). If you would like to receive the email version, please send your request to david.r.curry@centerforvaccineethicsandpolicy.org.
– pdf version: A pdf of the current issue is available here:
– blog edition: comprised of the approx. 35+ entries posted below.
– Twitter: Readers can also follow developments on twitter: @vaxethicspolicy.
.
– Links: We endeavor to test each link as we incorporate it into any post, but recognize that some links may become “stale” as publications and websites reorganize content over time. We apologize in advance for any links that may not be operative. We believe the contextual information in a given post should allow retrieval, but please contact us as above for assistance if necessary.
Support this knowledge-sharing service: Your financial support helps us cover our costs and to address a current shortfall in our annual operating budget. Click here to donate and thank you in advance for your contribution.
.
David R. Curry, MS
Executive Director
Center for Vaccine Ethics and Policy
Milestones :: Perspectives :: Research
::::::
WHO – 148th session of the Executive Board EB148
18-26 January 2021
:: Webcast
WHO Director-General’s closing remarks at 148th session of the Executive Board
26 January 2021
…You have addressed an enormous range of issues, reflecting the huge scope of health challenges we face, and the huge scope of expectations the world has of WHO.
Your agenda has spanned many issues related to the “triple billion” targets, including oral health, social determinants of health, patient safety, diabetes, NCDs, disability, HIV, hepatitis and sexually transmitted infections, eye care, antimicrobial resistance, immunization, medical devices, substandard and falsified medical products, local production, health innovation and intellectual property – and more.
You have also had important discussions about strengthening WHO to better support countries in addressing this massive scope of issues – the programme budget, sustainable financing, sexual exploitation and abuse, and more.
All of this is happening under the shadow of the greatest health crisis of our time.
This Saturday marks one year since I declared a public health emergency of international concern over the emergence of the novel coronavirus. At the time, there were fewer than 100 cases – to be exact, 98 cases – and no deaths reported outside China.
This week, we expect to reach 100 million reported cases, and more than 2 million people have lost their lives.
When this Board met a year ago, I said the world had a “window of opportunity” to prevent widespread transmission of this new virus. Some heeded that call; some did not. Now, vaccines are giving us another window of opportunity to bring the pandemic under control. We must not squander it.
As we speak, rich countries are rolling out vaccines, while the world’s least-developed countries watch and wait. With every day that passes, the divide grows larger between the world’s haves and have nots.
In my opening remarks last Monday, I said the world faced a catastrophic moral failure if it doesn’t walk the talk on vaccine equity.
A new study published yesterday by the International Chamber of Commerce Research Foundation confirms that it would also be an economic failure.
The study finds that vaccine nationalism could cost the global economy up to US$9.2 trillion, and almost half of that – US$4.5 trillion – would be incurred in the wealthiest economies.
By contrast, the financing gap for the Access to COVID-19 Tools Accelerator this year is US$26 billion. If fully funded, the ACT Accelerator would return up to US$166 for every dollar invested.
In our press conference yesterday, the Secretary General of the International Chamber of Commerce called the funding gap for the ACT Accelerator a “rounding error” – and he’s right – compared to the stimulus packages that have been rolled out in many countries.
Vaccine nationalism might serve short-term political goals. But it’s in every nation’s own interests to support vaccine equity.
I leave you with the challenge I set at the beginning of the week: together, we must ensure that vaccination of health workers and older people is underway in all countries within the first 100 days of this year. We have 74 days left. Time is short, and the stakes could not be higher. Every moment counts…
::::::
Presentation to EB148 on ACT Accelerator & COVAX

Presentation to E]148: COVID-19 situation and strategic response
[Selected Slides]

Presentation to EB: Strengthening WHO’s Global Emergency Preparedness and Response
[Selected Slides]

Coronavirus [COVID-19] – WHO
Public Health Emergency of International Concern (PHEIC)
https://www.who.int/emergencies/diseases/novel-coronavirus-2019
Weekly Epidemiological and Operational updates
Last update: 23 January 2021
Confirmed cases :: 101 561 219 [week ago: 96 877 399] [two weeks ago: 92 506 811
Confirmed deaths :: 2 196 944 [week ago: 2 098 879] [two weeks ago: 2 001 773]
Countries, areas or territories with cases :: 223
Weekly epidemiological update -27 January 2021
Overview
Globally, 4.1 million new cases were reported in the past week, a decline of 15% from last week, and the number of new deaths reported was just under 96 000, comparable to the previous week. This brings the cumulative numbers to over 98.2 million reported cases and over 2.1 million deaths globally since the start of the pandemic
Special Focus: Update on SARS-CoV-2 variants of concern
[1] Variant VOC 202012/01, lineage B.1.1.7: Since our last update on 19 January, variant VOC 202012/01 has been detected in ten additional countries. As of 25 January, a total of 70 countries across all six WHO regions have reported either imported cases or community transmission of this variant (Figure 3). Local transmission has been reported in several other European countries…
[2] Variant 501Y.V2, lineage B.1.351: Since the last update on 19 January, 501Y.V2 has been reported from eight additional countries– now totalling 31 countries across five of the six WHO regions (Figure 4). In South Africa, where this variant was initially identified, new weekly cases increased from early November 2020, peaking in early January 2021. In the past two weeks, a decreasing trend has been observed…
[3] Variant P.1, lineage B.1.1.28: Since our last update, variant P.1 has been reported in six additional countries. To date, this variant is reported in eight countries (Figure 5). In Brazil, where the variant was initially identified in addition to detection in a group of travellers from Brazil to Japan, the number of new weekly cases in the past two weeks are reported at higher levels compared to that of September to November 2020, and new weekly deaths have increased since early November 2020. The highest weekly cases since the start of the pandemic was reported in the week commencing 11 January 2021. Based on the preliminary investigations conducted in Manaus, Amazonas State, there has been an increase in the proportion of cases sequenced as variant P.1, from 52.2% (35/67) in December 2020 to 85.4% (41/48) in January 2021, highlighting ongoing local transmission of this variant and, given the mutations documented, raising similar concerns for potential increases in transmissibility or propensity for re-infection (10). Further studies are needed to assess if there are changes in transmissibility, severity or antibody neutralizing activity as a result of these new variants…
The emergence of new variants has underscored the importance for everyone, including those previously infected or vaccinated, to strictly adhere to public health and social measures. They also highlight the importance of increasing diagnostic capacity and systematic sequencing of SARS-CoV-2 where capacity allows, as well as the timely sharing of sequence data internationally. Systematic sequencing should be considered for a subset of incoming travellers, as well as community-based samples to ascertain the existence and extent of local transmission. Virus sequencing should be performed in all breakthrough disease following vaccination, in addition to population-based vaccine effectiveness studies. Global surveillance on virus evolution should continue to inform adjustments to public health and social measures
::::::
Interim recommendations for use of the Moderna mRNA-1273 vaccine against COVID-19
25 January 2021 | COVID-19: Critical preparedness, readiness and response
Overview
This interim guidance has been developed on the basis of the advice issued by the Strategic Advisory Group of Experts on Immunization (SAGE) at its extraordinary meeting on 21 January 2021.
PDF: https://apps.who.int/iris/rest/bitstreams/1328356/retrieve
Milestones :: Perspectives :: Research
UNICEF COVID-19 Vaccine Market Dashboard :: Agreements Table Accessed 30 Jan 2021
An overview of information collected from publicly announced bilateral and multilateral supply agreement [Agreements view from 2021-01-16 to date]
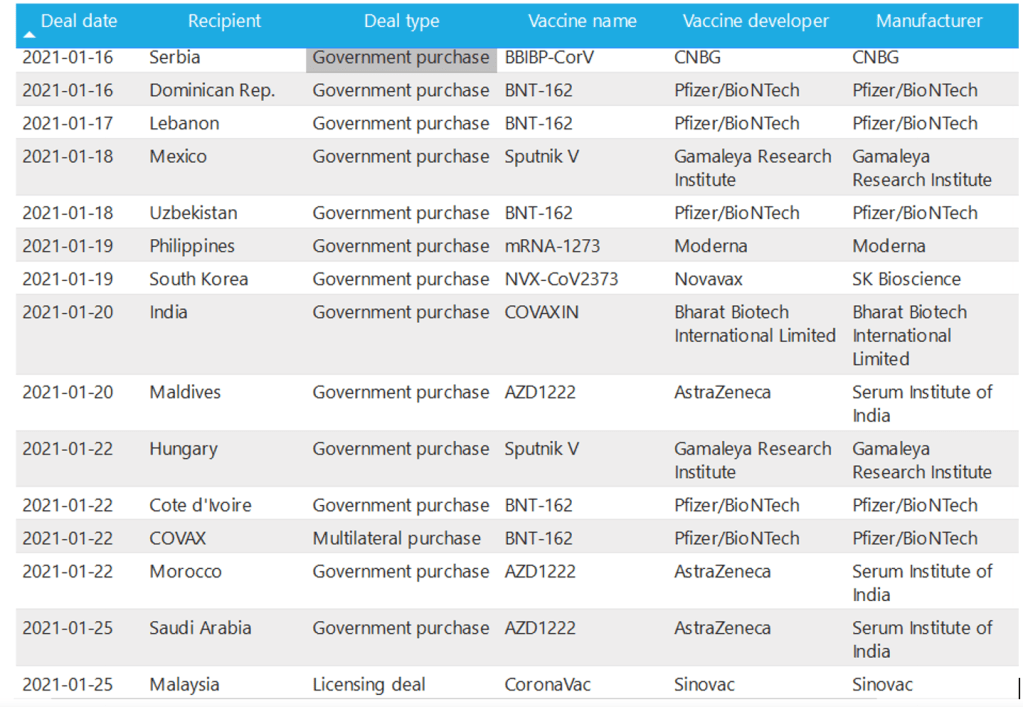
Milestones :: Perspectives :: Research
Vaccine Developer Announcements
COVID-19 Vaccine AstraZeneca authorised for use in the EU
29 January 2021
AstraZeneca’s COVID-19 vaccine has been granted a conditional marketing authorisation (CMA) in the European Union (EU) for active immunisation to prevent COVID-19 caused by SARS-CoV-2, in individuals 18 years of age and older.
Following review of the application, the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency based its positive opinion on data from a rolling review of trial data from the primary analysis of the Phase III programme led by the University of Oxford. Additional safety and efficacy data for the vaccine will continue to accumulate from ongoing clinical trials and is expected to be published in the coming weeks.
The CHMP recommends two doses of COVID-19 Vaccine AstraZeneca, formerly AZD1222, to be administered at a four- to 12-week interval in people aged 18 years and older. This dosing regimen was shown in clinical trials to be safe and effective in preventing symptomatic COVID-19, with no severe cases and no hospitalisations more than 14 days after the second dose.
AstraZeneca is working with the EU following the approval of a CMA for active immunisation to begin across member states…
Johnson & Johnson Announces Single-Shot Janssen COVID-19 Vaccine Candidate Met Primary Endpoints in Interim Analysis of its Phase 3 ENSEMBLE Trial
:: Vaccine Candidate 72% Effective in the US and 66% Effective Overall at Preventing Moderate to Severe COVID-19, 28 Days after Vaccination
:: 85% Effective Overall in Preventing Severe Disease and Demonstrated Complete Protection Against COVID-19 related Hospitalization and Death as of Day 28
:: Protection Against Severe Disease Across Geographies, Ages, and Multiple Virus Variants, including the SARS-CoV-2 Variant from the B.1.351 Lineage¹ Observed in South Africa
:: Single-shot compatible with standard vaccine distribution channels provides important tool in pandemic setting
Jan 29, 2021
Novartis signs initial agreement to provide manufacturing capacity for Pfizer-BioNTech COVID-19 vaccine
Jan 29, 2021
:: First of multiple agreements being explored by Novartis to leverage manufacturing capacity and capabilities to address pandemic, supporting global supply of COVID-19 vaccine
:: Initial agreement signed with BioNTech to use Novartis manufacturing plant in Stein, Switzerland, to support production of mRNA COVID-19 vaccine
::Production to commence in the second quarter for estimated delivery commencing in the third quarter 2021
Merck Discontinues Development of SARS-CoV-2/COVID-19 Vaccine Candidates; Continues Development of Two Investigational Therapeutic Candidates
January 25, 2021
Merck (NYSE: MRK), known as MSD outside the United States and Canada, today announced that the company is discontinuing development of its SARS-CoV-2/COVID-19 vaccine candidates, V590 and V591, and plans to focus its SARS-CoV-2/COVID-19 research strategy and production capabilities on advancing two therapeutic candidates, MK-4482 and MK-7110. This decision follows Merck’s review of findings from Phase 1 clinical studies for the vaccines. In these studies, both V590 and V591 were generally well tolerated, but the immune responses were inferior to those seen following natural infection and those reported for other SARS-CoV-2/COVID-19 vaccines…