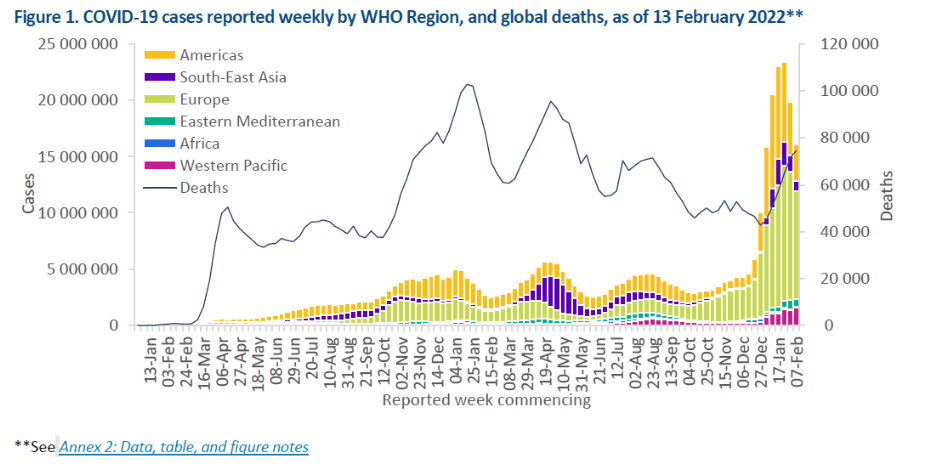
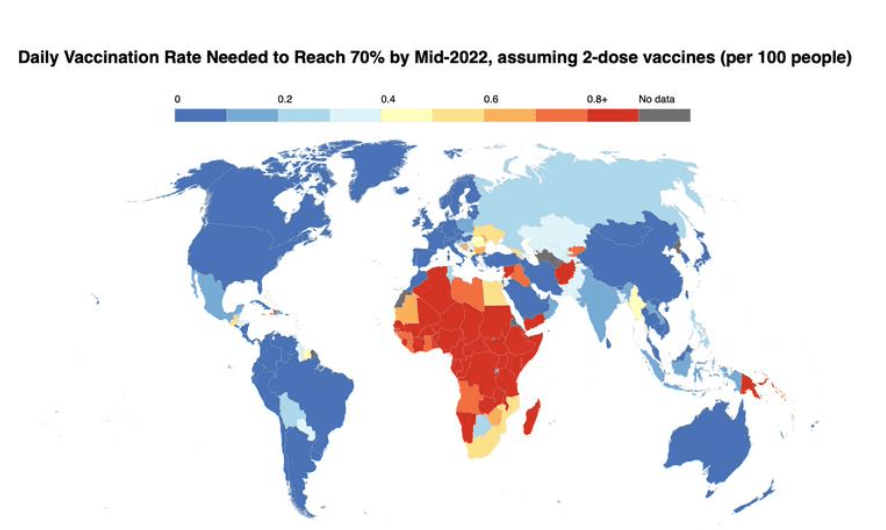
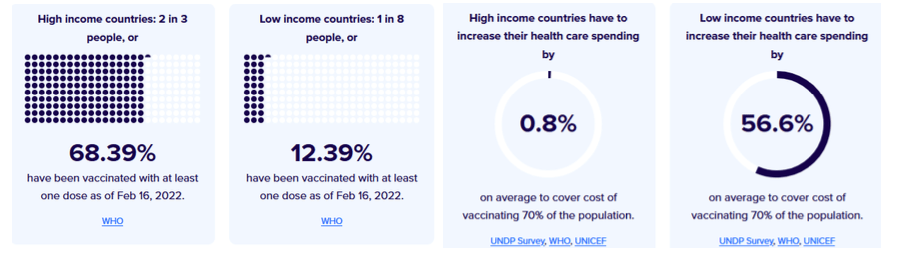
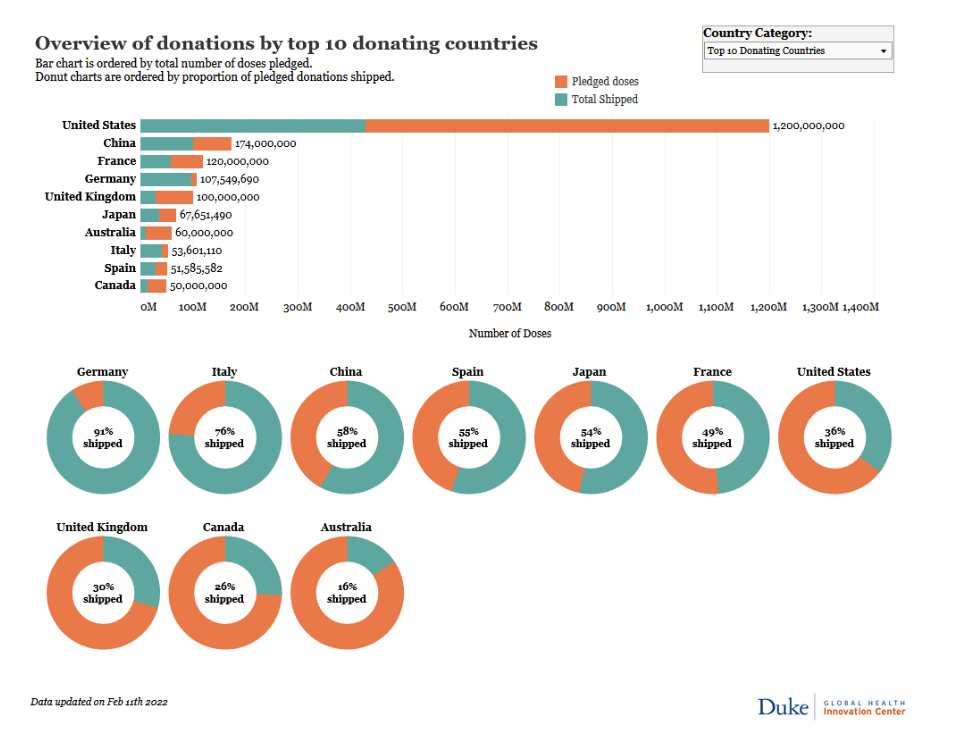
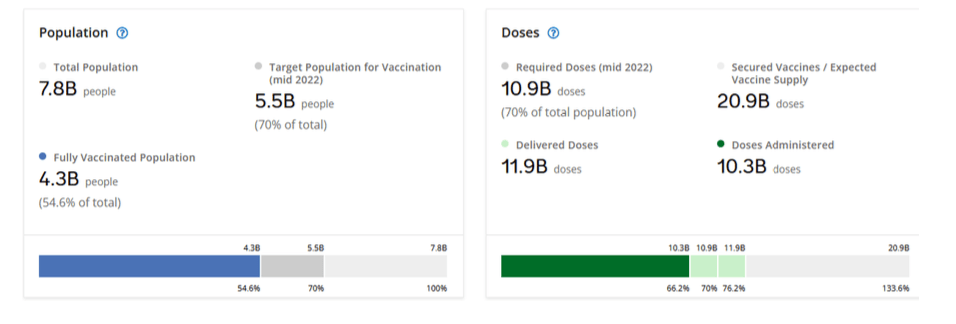
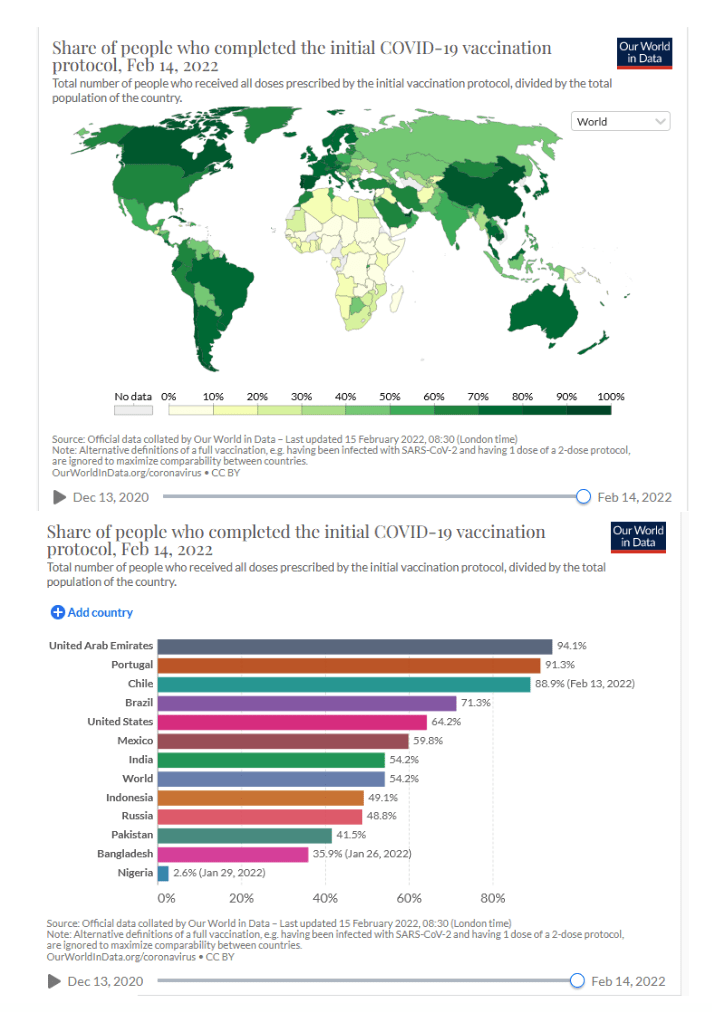
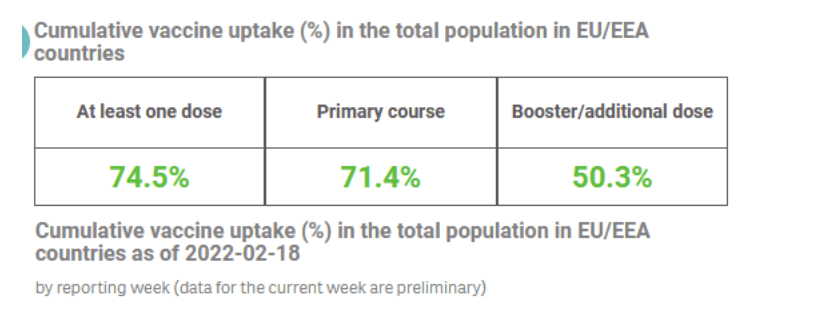
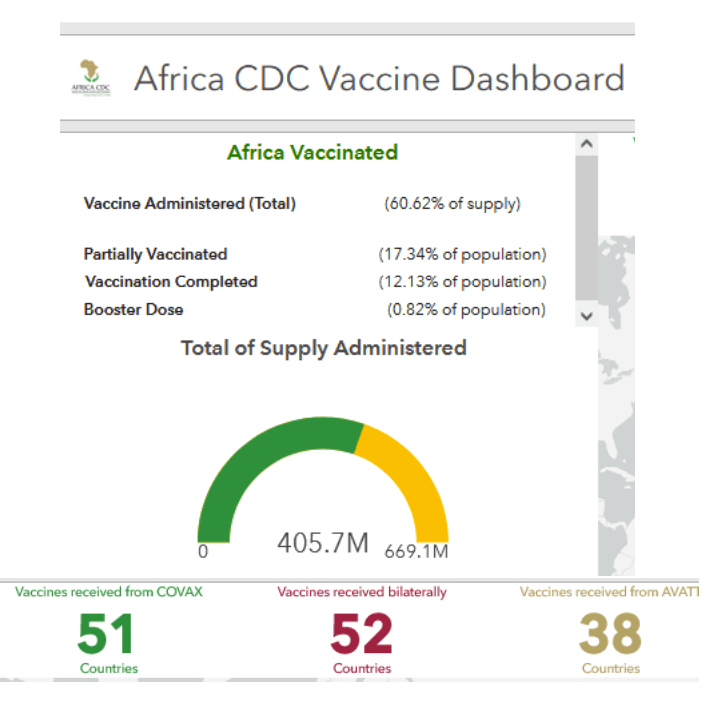
Pre-Print Servers
Gates Open Research
https://gatesopenresearch.org/browse/articles
[Accessed 26 Feb 2022]
Research Article metrics
Revised
Do differentiated service delivery models for HIV treatment in sub-Saharan Africa save money? Synthesis of evidence from field studies conducted in sub-Saharan Africa in 2017-2019 [version 2; peer review: 1 approved, 1 approved with reservations]
Sydney Rosen, Brooke Nichols, Teresa Guthrie, Mariet Benade, Salome Kuchukhidze, Lawrence Long
Peer Reviewers Ingrid Eshun-Wilson; Michael J. Vinikoor
Funder
NIH
LATEST VERSION PUBLISHED 25 Feb 2022
medRxiv
https://www.medrxiv.org/content/about-medrxiv
medRxiv is a free online archive and distribution server for complete but unpublished manuscripts (preprints) in the medical, clinical, and related health sciences. Preprints are preliminary reports of work that have not been certified by peer review. They should not be relied on to guide clinical practice or health-related behavior and should not be reported in news media as established information. medRxiv is for the distribution of preprints – complete but unpublished manuscripts – that describe human health research conducted, analyzed, and interpreted according to scientific principles…
Waning Effectiveness of the Third Dose of the BNT162b2 mRNA COVID-19 Vaccine
Tal Patalon, Yaki Saciuk, Asaf Peretz, Galit Perez, Yoav Lurie, Yasmin Maor, Sivan Gazit
medRxiv 2022.02.25.22271494; doi: https://doi.org/10.1101/2022.02.25.22271494
The effect of job strain and worksite social support on reported side effects of COVID-19 vaccine: a prospective study of employees in Japan
Natsu Sasaki, Reiko Kuroda, Kanami Tsuno, Kotaro Imamura, Norito Kawakami
medRxiv 2022.02.24.22271484; doi: https://doi.org/10.1101/2022.02.24.22271484
Booster protection against Omicron infection in a highly vaccinated cohort
Caroline Tai, Lisa L Maragakis, Sarah Connelly, John DiFiori, Leroy Sims, Eleanor Adams, Deverick Anderson, Michael Merson, David Ho, Yonatan Grad, Christina DeFilippo Mack
medRxiv 2022.02.24.22271347; doi: https://doi.org/10.1101/2022.02.24.22271347
Rapid genome surveillance of SARS-CoV-2 and study of risk factors using shipping container laboratories and portable DNA sequencing technology
SARA FARAHI BILOOEI, Dejana Jovicevic, Arash Iranzadeh, Cynthia Mpofu, Ivan Muscat, Anthony Thomas, Helene Steiner, Thomas Meany
medRxiv 2022.02.25.22271277; doi: https://doi.org/10.1101/2022.02.25.22271277
Public Health Impact of the Pfizer-BioNTech COVID-19 vaccine (BNT162b2) in the first year of rollout in the United States
Manuela Di Fusco, Kinga Marczell, Kristen A Deger, Mary M Moran, Timothy L Wiemken, Alejandro Cane, Solene de Boisvilliers, Jingyan Yang, Shailja Vaghela, Julie Roiz
medRxiv 2022.02.24.22271478; doi: https://doi.org/10.1101/2022.02.24.22271478
At least three doses of leading vaccines essential for neutralisation of SARS-CoV-2 Omicron variant
Nagendrakumar B Singanallur, Petrus Jansen van Vuren, Alexander James McAuley, Matthew P Bruce, Michael J Kuiper, Stella May Gwini, Shane Riddell, Sarah Goldie, Trevor W Drew, Kim Rebecca Blasdell, Mary Tachedjian, Shruthi Mangalaganesh, Simran Chahal, Leon Caly, Julian D Druce, Jennifer A Juno, Stephen Kent, Adam K Wheatley, Seshadri S Vasan
medRxiv 2022.02.20.22271237; doi: https://doi.org/10.1101/2022.02.20.22271237
Effectiveness of the third dose of BNT162b2 vaccine on neutralizing Omicron variant in the Japanese population
Hitoshi Kawasuji, Yoshitomo Morinaga, Hideki Tani, Yumiko Saga, Makito Kaneda, Yushi Murai, Akitoshi Ueno, Yuki Miyajima, Yasutaka Fukui, Kentaro Nagaoka, Chikako Ono, Yoshiharu Matsuura, Hideki Niimi, Yoshihiro Yamamoto
medRxiv 2022.02.23.22271433; doi: https://doi.org/10.1101/2022.02.23.22271433
Bibliometric study of preclinical, clinical, and public health systematic reviews and meta-analyses in Ethiopia: systematically mapping publication outputs, authors collaboration networks, trending research topics, and influential articles
Tesfa Dejenie Habtewold, Nigussie Tadesse Sharew, Aklilu Endalamaw, Henok Mulugeta, Getenet Dessie, Nigus Gebremedhin Asefa, Getachew Mulu Kassa, Wubet Alebachew Bayih, Mulugeta Molla Birhanu, Andreas A. Teferra, Abera Kenay Tura, Balewgize Sileshi Tegegne, Sisay Mulugeta Alemu
medRxiv 2022.02.24.22271416; doi: https://doi.org/10.1101/2022.02.24.22271416
The impact of COVID passport mandates on the number of cases of and hospitalizations with COVID-19 in the UK: a difference-in-differences analysis
Kim López-Güell, Albert Prats-Uribe, Martí Catalá, Clara Prats, Jotun Hein, DANIEL PRIETO-ALHAMBRA
medRxiv 2022.02.24.22271325; doi: https://doi.org/10.1101/2022.02.24.22271325
Dynamics of the Delta and Omicron variants of SARS-CoV-2 in the United States: the battle of supremacy in the presence of vaccination, mask usage and antiviral treatment
Calistus N. Ngonghala, Hemaho B. Taboe, Abba B. Gumel
medRxiv 2022.02.23.22271394; doi: https://doi.org/10.1101/2022.02.23.22271394
When mistrust in the government and scientists reinforce social inequalities in vaccination against Covid-19
Nathalie Bajos, Alexis Spire, Léna Silberzan, Antoine Sireyjol, Florence Jusot, Laurence Meyer, Jeanna-Eve Franck, Josiane Warszawski, the EpiCov study group
medRxiv 2022.02.23.22271397; doi: https://doi.org/10.1101/2022.02.23.22271397
Estimating the impact of implementation and timing of COVID-19 vaccination programme in Brazil: a counterfactual analysis
Leonardo Souto Ferreira, Flavia Maria Darcie Marquitti, Rafael Lopes Paixão da Silva, Marcelo Eduardo Borges, Marcelo Ferreira da Costa Gomes, Oswaldo Gonçruz Cruz, Roberto André Kraenkel, Renato Mendes Coutinho, Paulo Inácio Prado, Leonardo Soares Bastos
medRxiv 2021.12.24.21268384; doi: https://doi.org/10.1101/2021.12.24.21268384
SARS-CoV-2 seroprevalence, vaccination, and hesitancy in agricultural workers in Guatemala
Diva M. Calvimontes, Lyndsay Krisher, Alex Cruz-Aguilar, Daniel Pilloni-Alessio, Luis E. Crisostomo-Cal, Edgar A. Castañeda-Sosa, Jaime Butler-Dawson, Daniel Olson, Lee S. Newman, Edwin J. Asturias
medRxiv 2022.02.22.22270907; doi: https://doi.org/10.1101/2022.02.22.22270907
An inactivated SARS-CoV-2 vaccine is safe and induces humoral and cellular immunity against virus variants in healthy children and adolescents in Chile
Jorge A Soto, Felipe Melo-González, Cristián Gutierrez-Vera, Bárbara M Schultz, Roslye V Berríos-Rojas, Daniela Rivera-Pérez, Alejandro Piña-Iturbe, Guillermo Hoppe-Elsholz, Luisa F Duarte, Yaneisi Vázquez, Daniela Moreno-Tapia, Mariana Ríos, Pablo A Palacios, Richard Garcia-Betancourt, Álvaro Santibañez, Constanza Mendez, Benjamín Diethelm-Varela, Patricio Astudillo, Mario Calvo, Antonio Cárdenas, Marcela González, Macarena Goldsack, Valentina Gutiérrez, Marcela Potin, Andrea Schilling, Lorena I Tapia, Loreto Twele, Rodolfo Villena, Alba Grifoni, Alessandro Sette, Daniela Weiskopf, Rodrigo A Fasce, Jorge Fernández, Judith Mora, Eugenio Ramírez, Aracelly Gaete-Argel, Mónica Acevedo, Fernando Valiente-Echeverría, Ricardo Soto-Rifo, Angello Retamal-Díaz, Nathalia Muñoz-Jofré, PedCoronaVac03CL Study Group, Xing Meng, Qianqian Xin, Eduardo Alarcón-Bustamante, José V González-Aramundiz, Nicole Le Corre, María Javiera Álvarez, Pablo A González, Katia Abarca, Cecilia Perret, Leandro J Carreño, Alexis M Kalergis, Susan M Bueno
medRxiv 2022.02.15.22270973; doi: https://doi.org/10.1101/2022.02.15.22270973
Community-based Case Studies of Vaccine Hesitancy and the COVID-19 Response in South Africa; The VaxScenes Study
Charles Shey Wiysonge, Nancy Coulson, Nirvana Pillay, Sara Cooper, Candice Groenewald, Zaynab Essack, Saahier Parker, Gregory Houston, Jane Simmonds, Anelisa Jaca, Muyunda Mutemwa, Patrick DMC Katoto, Heidi van Rooyen
medRxiv 2022.02.21.22271272; doi: https://doi.org/10.1101/2022.02.21.22271272
Anti-SARS-CoV-2 IgG antibodies after recovery from COVID-19 or vaccination in Libyan population: comparison of four vaccines
Fawzi Ebrahim, Salah Tabal, Yosra Lamami, Inas M Alhudiri, Salah Edin El Meshri, Samira M Al Dwigen, Ramadan Arfa, Asma Alboeshi, Hafsa A. Alemam, Fauzia Abuhtna, Rabeeah Altrhouni, Mohamed B Milad, Nada A Elgriw, Mahmoud A Ruaua, Zakarya Abusrewil, Warda Harroush, Mwada Jallul, Fouziyah S Ali, Farag Eltaeb, Adam Elzaghied
medRxiv 2022.02.18.22271130; doi: https://doi.org/10.1101/2022.02.18.22271130
Wellcome Open Research [to 26 Feb 2022]
https://wellcomeopenresearch.org/browse/articles
[Accessed 26 Feb 2022]
Wellcome Open Research provides all Wellcome researchers with a place to rapidly publish any results they think are worth sharing. All articles benefit from rapid publication, transparent peer review and editorial guidance on making all source data openly available.
Research Article metrics
Revised
Revealing the extent of the first wave of the COVID-19 pandemic in Kenya based on serological and PCR-test data [version 2; peer review: 1 approved, 1 approved with reservations]
John Ojal, Samuel P. C. Brand, Vincent Were, Emelda A. Okiro, Ivy K. Kombe, Caroline Mburu, Rabia Aziza, Morris Ogero, Ambrose Agweyu, George M. Warimwe, Sophie Uyoga, Ifedayo M. O. Adetifa, J. Anthony G. Scott, Edward Otieno, Lynette I. Ochola-Oyier, Charles N. Agoti, Kadondi Kasera, Patrick Amoth, Mercy Mwangangi, Rashid Aman, Wangari Ng’ang’a, Benjamin Tsofa, Philip Bejon, Edwine Barasa, Matt J. Keeling, D. James Nokes
Peer Reviewers Mark Kimathi; Amy Wesolowski
Funders
Wellcome Trust
National Institute for Health Research
Medical Research Council
Foreign, Commonwealth and Development Office
LATEST VERSION PUBLISHED 25 Feb 2022
Research Article metrics
Revised
Individual-level variations in malaria susceptibility and acquisition of clinical protection [version 3; peer review: 1 approved, 1 approved with reservations]
John Joseph Valletta, John W.G. Addy, Adam J. Reid, Francis M. Ndungu, Yaw Bediako, Jedida Mwacharo, Khadija Said, Jennifer Musyoki, Joyce Mwongeli Ngoi, Joshua Wambua, Edward Otieno, Matt Berriman, Philip Bejon, Kevin Marsh, Jean Langhorne, Chris I. Newbold, Mario Recker
Peer Reviewers Lucy C. Okell; Wendy Prudhomme-O’Meara and Christine Markwalter
Funders
Wellcome Trust
Cancer Research UK
Medical Research Council
Crick Africa Network
LATEST VERSION PUBLISHED 25 Feb 2022