The Lancet
Sep 25, 2021 Volume 398 Number 10306 p1105-1194
https://www.thelancet.com/journals/lancet/issue/current
Comment
Global youth advocating for COVID-19 vaccines for all
Neema Kaseje, David Walcott
The Lancet
Sep 25, 2021 Volume 398 Number 10306 p1105-1194
https://www.thelancet.com/journals/lancet/issue/current
Comment
Global youth advocating for COVID-19 vaccines for all
Neema Kaseje, David Walcott
The Lancet
Sep 25, 2021 Volume 398 Number 10306 p1105-1194
https://www.thelancet.com/journals/lancet/issue/current
Articles
Cerebral venous thrombosis after vaccination against COVID-19 in the UK: a multicentre cohort study
Richard J Perry, et al.on behalf of the CVT After Immunisation Against COVID-19 (CAIAC) collaborators
The Milbank Quarterly
A Multidisciplinary Journal of Population Health and Health Policy
Volume 99, Issue 3 Pages: 601-852 September 2021
https://onlinelibrary.wiley.com/toc/14680009/current
Perspectives
Artificial Intelligence and Liability in Medicine: Balancing Safety and Innovation
GEORGE MALIHA, SARA GERKE, I. GLENN COHEN, RAVI B. PARIKH
Pages: 629-647
First Published: 06 April 2021
Nature
Volume 597 Issue 7877, 23 September 2021
https://www.nature.com/nature/volumes/597/issues/7875
Editorial | 22 September 2021
Greece used AI to curb COVID: what other nations can learn
Governments are hungry to deploy big data in health emergencies. Scientists must help to lay the legal, ethical and logistical groundwork.
Nature
Volume 597 Issue 7877, 23 September 2021
https://www.nature.com/nature/volumes/597/issues/7875
Article | 15 September 2021
Resurgence of Ebola virus in 2021 in Guinea suggests a new paradigm for outbreaks
The viral lineage responsible for the February 2021 outbreak of Ebola virus disease in Guinea is nested within a clade that predominantly consists of genomes sampled during the 2013–2016 epidemic, suggesting that the virus might have re-emerged after a long period of latency within a previously infected individual.
Alpha Kabinet Keita, Fara R. Koundouno, N’. Faly Magassouba
Nature Human Behaviour
Volume 5 Issue 9, September 2021
https://www.nature.com/nathumbehav/volumes/5/issues/9
Editorial | 23 August 2021
Our commitment to care in handling sensitive science
Science is not separate from societal issues. The production and impact of research are fundamentally social processes, and these too warrant scrutiny.
Nature Human Behaviour
Volume 5 Issue 9, September 2021
https://www.nature.com/nathumbehav/volumes/5/issues/9
News & Views | 23 August 2021
The value of consent for biobanking
Biobanks facilitate large-scale tests of hypotheses that may advance health, but whether biobanking participants adequately comprehend the potential uses of their data should concern researchers and the public. Consent matters because it provides a singular safeguard and a participatory mechanism to influence science’s production of new forms of power.
Elizabeth Bromley, Dmitry Khodyakov
Nature Human Behaviour
Volume 5 Issue 9, September 2021
https://www.nature.com/nathumbehav/volumes/5/issues/9
Article | 03 August 2021
A worldwide assessment of changes in adherence to COVID-19 protective behaviours and hypothesized pandemic fatigue
In the context of the COVID-19 pandemic, Petherick, Goldszmidt et al. show that, from March to December 2020, adherence to physical distancing fell worldwide, while mask-wearing adherence increased.
Anna Petherick, Rafael Goldszmidt, Andrew Wood
New England Journal of Medicine
September 23, 2021 Vol. 385 No. 13
http://www.nejm.org/toc/nejm/medical-journal
Perspective
Public Health Law after Covid-19 M.M. Mello and W.E. Parmet
New England Journal of Medicine
September 23, 2021 Vol. 385 No. 13
http://www.nejm.org/toc/nejm/medical-journal
Original Article
Safety and Efficacy of NVX-CoV2373 Covid-19 Vaccine
List of authors.
Paul T. Heath, F.R.C.P.C.H., et al. for the 2019nCoV-302 Study Group*
Conclusions
A two-dose regimen of the NVX-CoV2373 vaccine administered to adult participants conferred 89.7% protection against SARS-CoV-2 infection and showed high efficacy against the B.1.1.7 variant. (Funded by Novavax; EudraCT number, 2020-004123-16. opens in new tab.)
New England Journal of Medicine
September 23, 2021 Vol. 385 No. 13
http://www.nejm.org/toc/nejm/medical-journal
Original Article
Subcutaneous REGEN-COV Antibody Combination to Prevent Covid-19
Meagan P. O’Brien, M.D., et al for the Covid-19 Phase 3 Prevention Trial Team*
New England Journal of Medicine
September 23, 2021 Vol. 385 No. 13
http://www.nejm.org/toc/nejm/medical-journal
Sounding Board
Controlled Human Infection Model — Fast Track to HCV Vaccine? T.J. Liang, J.J. Feld, A.L. Cox, and C.M. Rice
PLoS Neglected Tropical Diseases
http://www.plosntds.org/
Research Article
Human leishmaniasis vaccines: Use cases, target population and potential global demand
Stefano Malvolti, Melissa Malhame, Carsten F. Mantel, Epke A. Le Rutte, Paul M. Kaye
| published 21 Sep 2021 PLOS Neglected Tropical Diseases
https://doi.org/10.1371/journal.pntd.0009742
PLoS One
http://www.plosone.org/
[Accessed 25 Sep 2021]
Use of medicinal plants for COVID-19 prevention and respiratory symptom treatment during the pandemic in Cusco, Peru: A cross-sectional survey
Magaly Villena-Tejada, Ingrid Vera-Ferchau, Anahí Cardona-Rivero, Rina Zamalloa-Cornejo, Maritza Quispe-Florez, Zany Frisancho-Triveño, Rosario C. Abarca-Meléndez, Susan G. Alvarez-Sucari, Christian R. Mejia, Jaime A. Yañez
Research Article | published 22 Sep 2021 PLOS ONE
https://doi.org/10.1371/journal.pone.0257165
PLoS One
http://www.plosone.org/
[Accessed 25 Sep 2021]
“Hot-spotting” to improve vaccine allocation by harnessing digital contact tracing technology: An application of percolation theory
Mark D. Penney, Yigit Yargic, Lee Smolin, Edward W. Thommes, Madhur Anand, Chris T. Bauch
Research Article | published 22 Sep 2021 PLOS ONE
https://doi.org/10.1371/journal.pone.0256889
PLoS One
http://www.plosone.org/
[Accessed 25 Sep 2021]
Knowledge of mothers regarding children’s vaccinations in Cyprus: A cross-sectional study
Maria Kyprianidou, Eleana Tzira, Petros Galanis, Konstantinos Giannakou
Research Article | published 20 Sep 2021 PLOS ONE
https://doi.org/10.1371/journal.pone.0257590
PLoS One
http://www.plosone.org/
[Accessed 25 Sep 2021]
Knowledge and preventive actions toward COVID-19, vaccination intent, and health literacy among educators in Japan: An online survey
Yasue Fukuda, Shuji Ando, Koji Fukuda
Research Article | published 20 Sep 2021 PLOS ONE
https://doi.org/10.1371/journal.pone.0257552
Research Ethics
Volume 17 Issue 4, October 2021
http://journals.sagepub.com/toc/reab/current
Original Article: Empirical
Towards an Ọmọlúàbí code of research ethics: Applying a situated, participant-centred virtue ethics framework to fieldwork with disadvantaged populations in diverse cultural settings
Bukola Oyinloye
First Published April 16, 2021; pp. 401–422
Research Ethics
Volume 17 Issue 4, October 2021
http://journals.sagepub.com/toc/reab/current
Original Article: Non-Empirical
A critical self-reflexive account of a privileged researcher in a complicated setting: Kakuma refugee camp
Neil Bilotta
First Published July 31, 2021; pp. 435–447
Research Ethics
Volume 17 Issue 4, October 2021
http://journals.sagepub.com/toc/reab/current
Original Article: Empirical
Negotiating the practicalities of informed consent in the field with children and young people: learning from social science researchers
Gina Sherwood, Sarah Parsons
First Published May 9, 2021; pp. 448–463
Revista Panamericana de Salud Pública/Pan American Journal of Public Health (RPSP/PAJPH)
https://www.paho.org/journal/en
24 Sep 2021
Sustainable Development Goals: impact of lack of financial protection in health in Latin American and Caribbean countries
Original research | Spanish |
Revista Panamericana de Salud Pública/Pan American Journal of Public Health (RPSP/PAJPH)
https://www.paho.org/journal/en
Selected Articles
24 Sep 2021
Virtual and in-person accompaniment of hospitalized patients during the COVID-19 pandemic in Colombia
Original research | Spanish |
Risk Management and Healthcare Policy
https://www.dovepress.com/risk-management-and-healthcare-policy-archive56
[Accessed 25 Sep 2021]
Original Research
Predictors of Intention to Get a COVID-19 Vaccine of Health Science Students: A Cross-Sectional Study
Nguyen VT, Nguyen MQ, Le NT, Nguyen TNH, Huynh G
Risk Management and Healthcare Policy 2021, 14:4023-4030
Published Date: 24 September 2021
Science
Volume 373| Issue 6562| 24 Sep 2021
https://www.science.org/toc/science/current
Special issue – Human Genomics
Introduction to Special Issue
The human genome
BY Laura M. Zahn
23 Sep 2021: 1458-1459
In the two decades since the publication of the human genome, advances in genetics have raced forward. Progress in human genomics has produced insights applicable to treating cancer, identifying drug targets, and diagnosing disease—especially in infants. However, despite these steps forward, we still lack the ability to predict how an individual’s inherited genome contributes to aging, complex disease, and even some monogenic diseases. Furthermore, de novo mutations have increasingly been proposed to affect disease onset and progression.
Although some hoped that having the human genome in hand would fuel a sprint to medical miracles, progress in this field is more accurately characterized as an ongoing relay race of contributions from genomic studies. We now have a better understanding of our history from ancient DNA sequencing, which, along with modern human population genetics, provides an idea of which mutations are tolerated and which are likely pathogenic. The effects of mutations, from single-nucleotide polymorphisms to the loss of full genes, and epigenetic changes that modify gene expression in single cells, tumors, and individuals are being investigated in ever-increasing numbers. These efforts have made it easier to identify how both common and rare variants, which may differ among populations, affect the genetic architecture underlying complex traits and disease.
This special issue examines well-earned successes in applying human genomic research toward understanding human evolution, cancer, polygenic traits, and functional genomics. Yet, there is still much ground ahead to cover.
Science
Volume 373| Issue 6562| 24 Sep 2021
https://www.science.org/toc/science/current
Policy Forum
Regulating forensic genetic genealogy
BY Natalie Ram, Erin E. Murphy, Sonia M. Suter
23 Sep 2021: 1444-1446
NO ACCESS
Maryland ’s new law provides a model for others
Science
Volume 373| Issue 6562| 24 Sep 2021
https://www.science.org/toc/science/current
Research Articles
Vaccine nationalism and the dynamics and control of SARS-CoV-2
BY Caroline E. Wagner et al.
24 Sep 2021
Open Access
Stockpiling and control
A triumph that has emerged from the catastrophe of the severe acute respiratory syndrome coronavirus 2 pandemic has been the rapid development of several potent vaccines. However, 18 months into the pandemic and more than 6 months after vaccine approval, wealthy countries remain the major beneficiaries. Wagner et al. model the consequences of vaccine stockpiling in affluent countries on disease rates in lower- and middle-income countries and the consequences for the eruption of new variants that could jeopardize the early success of vaccines. For countries that can readily access vaccines, it would be better to share vaccines equitably to lower disease burdens in countries with less access, reduce the cost of having to be constantly vigilant for case imports, and minimize virus evolution.
Structured Abstract
INTRODUCTION
The COVID-19 pandemic has imposed massive public health burdens and economic costs, with more than 220 million confirmed cases and more than 4.5 million deaths globally so far. In some countries, the rapid deployment of safe and effective vaccines has reduced cases, hospitalizations, and deaths. However, many regions across the world have little access to doses, and infections are resurging. Furthermore, sustained transmission has led to the emergence of novel variants of the causative severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus with increased transmissibility, and against which vaccines may be less effective. Thus, understanding the effects of vaccine nationalism and allocation on the dynamics and control of SARS-CoV-2 is critical.
RATIONALE
We extend recent models of SARS-CoV-2 dynamics to consider the number of cases and potential for viral evolution in two hypothetical regions, one with high access and one with low access to vaccines. This modified framework allows for variation in the strength and duration of natural and vaccinal immunity, which are aspects of the virus and host response that remain uncertain, thus enabling general explorations of a number of potential scenarios. To model the sharing of vaccines, we begin by only coupling the two regions through their vaccination rates. For different immuno-epidemiological scenarios of SARS-CoV-2, we examine the medium- and long-term dynamics in both regions as a function of the fraction of vaccines shared. Then, we couple the regions through immigration and introduce potential transmission increases due to evolution. We use this coupled model to explore local and combined infection numbers and clinical burdens, as well as potential viral adaptation.
RESULTS
In general, we find that stockpiling vaccines by countries with high availability leads to large increases in infections in countries with low vaccine availability, the magnitude of which depends on the strength and duration of natural and vaccinal immunity. A number of additional subtleties arise when the populations and transmission rates in each country differ depending on evolutionary assumptions and vaccine availability. Furthermore, the movement of infected individuals between countries combined with the possibility of evolutionary increases in viral transmissibility may greatly magnify local and combined infection numbers, suggesting that countries must invest in surveillance strategies to prevent case importation. Dose sharing is likely a high-return strategy because equitable allocation brings nonlinear benefits and also alleviates costs of surveillance (e.g., border testing, genomic surveillance) in settings where doses are sufficient to maintain cases at low numbers. Across a range of immunological scenarios, we find that vaccine sharing is also a powerful tool to decrease the potential for antigenic and transmission evolution of the virus, especially if infections after the waning of natural immunity contribute most to evolutionary potential.
CONCLUSION
Intuitively, our results indicate that unequal vaccine allocation will result in sustained transmission and increased case numbers in regions with low vaccine availability and thus to a higher associated clinical burden compared with a vaccinated population. Under certain scenarios, sustained local transmission could lead to an increased potential for antigenic evolution, which may result in the emergence of variants with novel antigenicity and/or transmissibility and affect epidemiological characteristics globally. Overall, our work underlines the importance of rapid, equitable vaccine deployment and the necessity to export vaccines to regions with low availability in parallel to their becoming available in regions with high access. Coordinated vaccination campaigns across the world, combined with improved surveillance and appropriate nonpharmaceutical interventions to prevent case importation, are imperative.
Science Translational Medicine
Volume 13| Issue 612| 22 Sep 2021
https://www.science.org/toc/stm/current
Reviews
Drug repurposing: Misconceptions, challenges, and opportunities for academic researchers
BY C. Glenn Begley et al.
22 Sep 2021
NO ACCESS
The assumption that drug repurposing efficiently delivers new drugs ignores the realities of drug development required to achieve regulatory approval.
Travel Medicine and Infectious Diseases
Volume 43 September–October 2021
https://www.sciencedirect.com/journal/travel-medicine-and-infectious-disease/vol/43/suppl/C
Editorial No access
Middle East respiratory syndrome coronavirus – The need for global proactive surveillance, sequencing and modeling
Jaffar A. Al-Tawfiq, Eskild Petersen, Ziad A. Memish, Stanley Perlman, Alimuddin Zumla
Article 102118
Travel Medicine and Infectious Diseases
Volume 43 September–October 2021
https://www.sciencedirect.com/journal/travel-medicine-and-infectious-disease/vol/43/suppl/C
Discussion Full text access
Launching COVID-19 vaccination in Saudi Arabia: Lessons learned, and the way forward
Abdullah Assiri, Jaffar A. Al-Tawfiq, Monira Alkhalifa, Hessa Al Duhailan, … Hani Jokhdar
Article 102119
Vaccine
Volume 39, Issue 37 Pages 5233-5332 (31 August 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/37
Conference info Abstract only
A global agenda for older adult immunization in the COVID-19 era: A roadmap for action
Lois A. Privor-Dumm, Gregory A. Poland, Jane Barratt, David N. Durrheim, … Paolo Bonanni
Pages 5240-5250
Vaccine
Volume 39, Issue 37 Pages 5233-5332 (31 August 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/37
Discussion Full text access
An overview of procurement, pricing, and uptake of COVID-19 vaccines in Pakistan
Adeel Siddiqui, Ali Ahmed, Maria Tanveer, Muhammad Saqlain, … Syed Shahzad Hasan
Pages 5251-5253
Vaccine
Volume 39, Issue 37 Pages 5233-5332 (31 August 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/37
Discussion Full text access
Geo-prioritization framework for COVID-19 vaccine allocation in India
Akshay Mahajan, Jasmine Kaur, Chirag Sidana, Shashwat Shivam, Harpreet Singh
Pages 5254-5256
Vaccine
Volume 39, Issue 37 Pages 5233-5332 (31 August 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/37
Review article Abstract only
HPV vaccine acceptance in West Africa: A systematic literature review
Rose Wilson
Pages 5277-5284
Vaccine
Volume 39, Issue 37 Pages 5233-5332 (31 August 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/37
Research article Full text access
How to ensure vaccine safety: An evaluation of China’s vaccine regulation system
Jiao Feng, Qin LI
Pages 5285-5294
Vaccine
Volume 39, Issue 37 Pages 5233-5332 (31 August 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/37
Research article Full text access
Optimal planning of the COVID-19 vaccine supply chain
Georgios P. Georgiadis, Michael C. Georgiadis
Pages 5302-5312
Vaccine
Volume 39, Issue 37 Pages 5233-5332 (31 August 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/37
Research article Open access
Effectiveness of a single-dose mass dengue vaccination in Cebu, Philippines: A case-control study
Michelle Ylade, Kristal An Agrupis, Jedas Veronica Daag, Maria Vinna Crisostomo, … Anna Lena Lopez
Pages 5318-5325
Vaccines
https://www.mdpi.com/journal/vaccines
Open Access Systematic Review
Worldwide Vaccination Willingness for COVID-19: A Systematic Review and Meta-Analysis
by Kimberly R. Nehal, Lieke M. Steendam, Maiza Campos Ponce, Marinka van der Hoeven and
G. Suzanne A. Smit
Vaccines 2021, 9(10), 1071; https://doi.org/10.3390/vaccines9101071 (registering DOI) – 24 Sep 2021
Abstract
Countries across the globe are currently experiencing a third or fourth wave of SARS-CoV-2 infections; therefore, the need for effective vaccination campaigns is higher than ever. However, effectiveness of these campaigns in disease reduction is highly dependent on vaccination uptake and coverage in […]
Vaccines
https://www.mdpi.com/journal/vaccines
Open Access Article
COVID-19 Vaccine Hesitancy and Resistance in India Explored through a Population-Based Longitudinal Survey
by Srikanth Umakanthan, Sonal Patil, Naveen Subramaniam and Ria Sharma
Vaccines 2021, 9(10), 1064; https://doi.org/10.3390/vaccines9101064 (registering DOI) – 24 Sep 2021
Abstract
Background: The World Health Organization has recommended that individual governments identify vaccine hesitancy areas. It is proposed that the governments, with the aid of local organizations, educate and implement social insights on the vaccination so that high population levels are covered with this […
Vaccines
https://www.mdpi.com/journal/vaccines
Open Access Article
Is It All a Conspiracy? Conspiracy Theories and People’s Attitude to COVID-19 Vaccination
by Zheng Yang, Xi Luo and Hepeng Jia
Vaccines 2021, 9(10), 1051; https://doi.org/10.3390/vaccines9101051 – 22 Sep 2021
Abstract
A large body of research has found that people’s beliefs in conspiracy theories about infectious diseases negatively impacts their health behaviors concerning vaccination. Conspiracy belief-based vaccination hesitancy has become more rampant after the global outbreak of COVID-19. However, some important questions remain unanswered. […]
medRxiv
medRxiv is a free online archive and distribution server for complete but unpublished manuscripts (preprints) in the medical, clinical, and related health sciences. Preprints are preliminary reports of work that have not been certified by peer review. They should not be relied on to guide clinical practice or health-related behavior and should not be reported in news media as established information. medRxiv is for the distribution of preprints – complete but unpublished manuscripts – that describe human health research conducted, analyzed, and interpreted according to scientific principles…
https://www.medrxiv.org/content/about-medrxiv
[Accessed 25 Sep 2021]
Selected Content
A systematic review of COVID-19 vaccine efficacy and effectiveness against SARS-CoV-2 infection and disease
Melissa M Higdon, Brian Wahl, Carli B Jones, Joseph G Rosen, Shaun A Truelove, Anurima Baidya, Anjalika A Nande, Parisa A ShamaeiZadeh, Karoline K Walter, Daniel R Feikin, Minal K Patel, Maria Deloria Knoll, Alison L Hill
medRxiv 2021.09.17.21263549; doi: https://doi.org/10.1101/2021.09.17.21263549
Predicting the Effectiveness of the Pfizer-BioNTech BNT162b2 Vaccine from SARS-CoV-2 Variants Neutralisation Data
Oleg Volkov, Svetlana Borozdenkova, Alexander Gray
medRxiv 2021.09.06.21263160; doi: https://doi.org/10.1101/2021.09.06.21263160
Consequences of COVID-19 vaccine allocation inequity in Chicago
Sharon Zeng, Kenley M. Pelzer, Robert D. Gibbons, Monica E. Peek, William F. Parker
medRxiv 2021.09.22.21263984; doi: https://doi.org/10.1101/2021.09.22.21263984
Real-world Effectiveness of 2-dose SARS-CoV-2 Vaccination in Kidney Transplant Recipients
Caitríona M. McEvoy, Anna Lee, Paraish S. Misra, Gerald Lebovic, Ron Wald, Darren A. Yuen
medRxiv 2021.09.21.21263457; doi: https://doi.org/10.1101/2021.09.21.21263457
Effectiveness of vaccination in preventing severe SARS CoV-2 infection in South India-a hospital-based cross-sectional study
A Charles Pon Ruban, Aazmi M, Shantaraman K.
medRxiv 2021.09.17.21263670; doi: https://doi.org/10.1101/2021.09.17.21263670
Effectiveness of COVID-19 Vaccines: Eight Months Post Single Dose Vaccination
Naif Khalaf Alharbi, Jaffar A. Al-Tawfiq, Suliman Alghnam, Amal Alwehaibe, Abrar Alasmari, Suliman A. Alsagaby, Faizah Alotaibi, Faisal Alsubaie, Majid Alshomrani, Fayssal M. Farahat, Mohammad Bosaeed, Ahmad Alharbi, Omar Aldibasi, Abdullah M. Assiri
medRxiv 2021.09.18.21263262; doi: https://doi.org/10.1101/2021.09.18.21263262
Impact of COVID-19 on the quality of life (QoL) of patients living with Sickle Cell Disorder (SCD) IN Lagos, Nigeria
Annette Akinsete, Larry Ajuwon, Michael Ottun, Hammed Adelabu, Jorden Veeneman
medRxiv 2021.09.17.21263748; doi: https://doi.org/10.1101/2021.09.17.21263748
Estimating vaccine confidence levels among healthcare staff and students of a tertiary institution in South Africa
Elizabeth O. Oduwole, Tonya Esterhuizen, Hassan Mahomed, Charles S. Wiysonge
medRxiv 2021.09.17.21263739; doi: https://doi.org/10.1101/2021.09.17.21263739
Stakeholders’ views on an institutional dashboard with metrics for responsible research
Tamarinde Haven, Martin Holst, Daniel Strech
medRxiv 2021.09.16.21263493; doi: https://doi.org/10.1101/2021.09.16.21263493
COVID-19 Vaccine Hesitancy in India: An Exploratory Analysis
Sandip K. Agarwal, Maharnab Naha
medRxiv 2021.09.15.21263646; doi: https://doi.org/10.1101/2021.09.15.21263646
Toward Using Twitter Data to Monitor Covid-19 Vaccine Safety in Pregnancy
Ari Z. Klein, Karen O’Connor, Graciela Gonzalez-Hernandez
medRxiv 2021.09.15.21263653; doi: https://doi.org/10.1101/2021.09.15.21263653
COVID-19 vaccine hesitancy and vaccine passports: Vaccination or restriction?
Shohei Okamoto, Kazuki Kamimura, Kohei Komamura
medRxiv 2021.09.15.21263559; doi: https://doi.org/10.1101/2021.09.15.21263559
Immunogenicity and safety of an inactivated SARS-CoV-2 vaccine in people living with HIV-1
Yanmeng Feng, Yifan Zhang, Zhangyufan He, Haojie Huang, Xiangxiang Tian, Gang Wang, Daihong Chen, Yanqin Ren, Liqiu Jia, Wanhai Wang, Jing Wu, Lingyun Shao, Wenhong Zhang, Heng Tang, Yanmin Wan
medRxiv 2021.09.14.21263556; doi: https://doi.org/10.1101/2021.09.14.21263556
Monitoring the COVID-19 immunisation programme through a National Immunisation Management System – England’s experience
Elise Tessier, Julia Stowe, Camille Tsang, Yuma Rai, Eleanor Clarke, Anissa Lakhani, Ashley Makwana, Heather Heard, Tim Rickeard, Freja Kirsebom, Catherine Quinot, Shreya Lakhani, Linda Power, Michael Edelstein, Andy Evans, Mary Ramsay, Jamie Lopez-Bernal, Joanne White, Charlotte Gower, Nick Andrews, Colin Campbell
medRxiv 2021.09.14.21263578; doi: https://doi.org/10.1101/2021.09.14.21263578
Monitoring the COVID-19 immunisation programme through a National Immunisation Management System – England’s experience
Elise Tessier, Julia Stowe, Camille Tsang, Yuma Rai, Eleanor Clarke, Anissa Lakhani, Ashley Makwana, Heather Heard, Tim Rickeard, Freja Kirsebom, Catherine Quinot, Shreya Lakhani, Linda Power, Michael Edelstein, Andy Evans, Mary Ramsay, Jamie Lopez-Bernal, Joanne White, Charlotte Gower, Nick Andrews, Colin Campbell
medRxiv 2021.09.14.21263578; doi: https://doi.org/10.1101/2021.09.14.21263578
COVID-19 Acceleration and Vaccine Status in France – August 2021
Christelle Baunez, Mickael Degoulet, Stéphane Luchini, Patrick A. Pintus, Miriam Teschl
medRxiv 2021.09.18.21263773; doi: https://doi.org/10.1101/2021.09.18.21263773
Covid-19 in the Phase 3 Trial of mRNA-1273 During the Delta-variant Surge
Lindsey R. Baden, Hana M. El Sahly, Brandon Essink, Dean Follmann, Kathleen M. Neuzil, Allison August, Heather Clouting, Gabrielle Fortier, Weiping Deng, Shu Han, Xiaoping Zhao, Brett Leav, Carla Talarico, Bethany Girard, Yamuna D. Paila, Joanne E. Tomassini, Florian Schödel, Rolando Pajon, Honghong Zhou, Rituparna Das, Jacqueline Miller
medRxiv 2021.09.17.21263624; doi: https://doi.org/10.1101/2021.09.17.21263624
The Relationship of Vaccine Uptake and COVID-19 Infections among Nursing Home Staff and Residents in Missouri
Stephen Scroggins, Matthew Ellis, Enbal Shacham
medRxiv 2021.09.16.21263714; doi: https://doi.org/10.1101/2021.09.16.21263714
COVID-19 vaccination rates among health care workers by immigrant background. A nation-wide registry study from Norway
Kristian Bandlien Kraft, Ingeborg Elgersma, Trude Marie Lyngstad, Petter Elstrøm, Kjetil Telle
medRxiv 2021.09.17.21263619; doi: https://doi.org/10.1101/2021.09.17.21263619
Correlates of COVID-19 vaccine acceptance, hesitancy and refusal among employees of a safety net California county health system with an early and aggressive vaccination program: Results from a cross-sectional survey
Nicole M. Gatto, Jerusha E. Lee, Donatella Massai, Susanna Zamarripa, Bijan Sasaninia, Dhruv Khurana, Kelsey Michaels, Deborah Freund, Judi Nightingale, Anthony Firek, for the Riverside University Health System (RUHS) Comparative Effectiveness and Clinical Outcomes Research Center (CECORC)-Claremont Graduate University (CGU) COVID-19 Research Group
medRxiv 2021.09.14.21263588; doi: https://doi.org/10.1101/2021.09.14.21263588
Think Tanks et al
Brookings [to 25 Sep 2021]
http://www.brookings.edu/
Op-Ed
Africa must produce its own vaccines
Landry Signé
Thursday, September 23, 2021
Center for Global Development [to 25 Sep 2021]
http://www.cgdev.org/page/press-center
Accessed 25 Sep 2021
Takeaways from the White House COVID Summit: Ending this Pandemic and Preparing for the Next
September 24, 2021
The global COVID response effort has been in desperate need of a shot in the arm. Yesterday’s US-hosted Global COVID-19 Summit, on the sidelines of the UN General Assembly, aimed to provide just that. Here are a few of the highlights, and what was missing.
Amanda Glassman and Erin Collinson
Early COVID-19 Vaccination Not Only Prevents Deaths, But Can Save Money Too, New Findings Show
September 24, 2021
In January 2021, India embarked on one of the world’s…
Hiral Anil Shah, Nimalan Arinaminpathy and Javier Guzman
Four Priorities for the New Leader of USAID’s Global Health Bureau
September 22, 2021
Next week, Atul Gawande, the prominent author, surgeon, researcher, and—most recently—presidential nominee to lead USAID’s Bureau for Global Health
Julia Kaufman, Sarah Rose and Janeen Madan Keller
Chatham House [to 25 Sep 2021]
https://www.chathamhouse.org/
Accessed 25 Sep 2021
[No new digest content identified]
CSIS
https://www.csis.org/
Accessed 25 Sep 2021
Blog Post
Russia’s New Frontier in Southeast Asia: Vaccine Diplomacy
September 21, 2021
Report
Beyond COVAX: The Importance of Public-Private Partnerships for Covid-19 Vaccine Delivery to Developing Countries
September 13, 2021 | Conor M. Savoy Public-private partnerships are critical to meeting the demand in developing countries to rapidly scale supply and distribution systems for Covid-19 vaccines in support of existing multilateral efforts such as COVAX.
Kaiser Family Foundation
https://www.kff.org/search/?post_type=press-release
[No new digest content identified]
Rand [to 25 Sep 2021]
https://www.rand.org/pubs.html
Selected Research Reports, Featured Journal Articles
Report
Identifying Strategies to Boost COVID-19 Vaccine Acceptance in the United States
Sep 9, 2021
This report recommends strategies to help boost COVID-19 vaccine acceptance in the United States in order to reach herd immunity and end the pandemic.
Florian F. Schmitzberger, Kirstin W. Scott, Wilson Nham, Kusum Mathews, Lucy Schulson, Sydney Fouche, Nasma Berri, Alex Shehab, Ashwin Gupta, Rama A. Salhi, Neil Kamdar, Jennifer Bouey, Mahshid Abir
Rockefeller Foundation [to 25 Sep 2021]
https://www.rockefellerfoundation.org/
Selected Reports/Press Releases
Sep 14 2021 Press Releases
Wellcome and The Rockefeller Foundation Fund New data.org initiative, Epiverse, Analyzing Emergence and Spread of Pandemics
data.org to lead global, distributed pandemic analysis with a privacy-preserving approach
New York | September 14, 2021 Today, data.org is announcing funding from Wellcome and The Rockefeller Foundation’s pandemic prevention institute …
Sep 14 2021 Press Releases
Over 90% of U.S. Employers Surveyed Require or Encourage Covid-19 Vaccinations
Arizona State University Survey of Employers, supported by The Rockefeller Foundation, shows companies are increasing support for Covid-19 vaccinations and testing to keep employees safe. PHOENIX, Ariz. | Sept. 14, 2021 …
World Economic Forum [to 25 Sep 2021]
https://agenda.weforum.org/news/
Media
India Completes First Drone Delivery of Vaccines, Start of New Pilot Programme with World Economic Forum
News 25 Sep 2021
Majority Want to Prioritize Equitable Access to COVID-19 Vaccines Before Boosters
News 10 Sep 2021
Health experts say at least 60% of world’s population needs to be vaccinated by 2022 to get the current pandemic under control – but only 0.4% of doses have been administered in low-income countries
Majority would receive a booster shot, but think priority goes to people yet to have a full dose
Read the full report
Vaccines and Global Health: The Week in Review is a weekly digest summarizing news, events, announcements, peer-reviewed articles and research in the global vaccine ethics and policy space. Content is aggregated from key governmental, NGO, international organization and industry sources, key peer-reviewed journals, and other media channels. This summary proceeds from the broad base of themes and issues monitored by the Center for Vaccine Ethics & Policy in its work: it is not intended to be exhaustive in its coverage. You are viewing the blog version of our weekly digest, typically comprised of between 30 and 40 posts below all dated with the current issue date
.– Request an Email Summary: Vaccines and Global Health : The Week in Review is published as a single email summary, scheduled for release each Saturday evening before midnight (EDT in the U.S.). If you would like to receive the email version, please send your request to david.r.curry@centerforvaccineethicsandpolicy.org.
– pdf version: A pdf of the current issue is available here:
– blog edition: comprised of the approx. 35+ entries posted below.
– Twitter: Readers can also follow developments on twitter: @vaxethicspolicy.
.
– Links: We endeavor to test each link as we incorporate it into any post, but recognize that some links may become “stale” as publications and websites reorganize content over time. We apologize in advance for any links that may not be operative. We believe the contextual information in a given post should allow retrieval, but please contact us as above for assistance if necessary.
Support this knowledge-sharing service: Your financial support helps us cover our costs and to address a current shortfall in our annual operating budget. Click here to donate and thank you in advance for your contribution.
.
David R. Curry, MS
Executive Director
Center for Vaccine Ethics and Policy
Milestones :: Perspectives :: Research
COVID
Editor’s Note:
As is obvious to all, the sheer volume of strategic announcements, regulatory actions, country program decisions, commentary, and, indeed, misinformation around COVID response continues at extraordinary levels. Our weekly digest strives to present a coherent and comprehensive snapshot, but cannot be exhaustive, If you recognize a missed strategic development, a new source of rigorous analysis, or an insight/commentary that would benefit our common understanding, please advise me…we will review all suggestions and consider them for inclusion in a subsequent edition: david.r.curry@ge2p2global.org
We are seeking access to modelling which engages scenarios and articulates imperatives around a COVID-19 pandemic end-game through at least a 2025 horizon. We assess that WHO must be conducting or contracting for such modeling – or should recognize an imperative to be doing so in its global health governance role. If we have missed such modeling in progress, we would be delighted to be advised of it and will include it in our coverage.
Milestones :: Perspectives :: Research
WHO calls on world leaders at the UN General Assembly to focus on vaccine equity, pandemic preparedness, and getting the SDGs back on track
17 September 2021 News release Geneva
WHO is urging leaders attending the 76th session of the United Nations General Assembly (UNGA) to guarantee equitable access to COVID-19 vaccines and other life-saving tools; ensure the world is better prepared to respond to future pandemics; and renew efforts to achieve the Sustainable Development Goals (SDGs).
The COVID-19 pandemic has already claimed the lives of nearly 5 million people around the globe, and the virus continues to circulate actively in all regions of the world.
Vaccines are the most critical tool to end the pandemic and save lives and livelihoods. More than 5.7 billion vaccine doses have been administered globally, but 73% of all doses have been administered in just 10 countries. High-income countries have administered 61 times more doses per inhabitant than low-income countries. The longer vaccine inequity persists, the more the virus will keep circulating and evolving, and the longer the social and economic disruption will continue.
WHO’s targets are to vaccinate at least 40% of the population of every country by the end of this year, and 70% by the middle of next year. These targets are achievable if countries and manufacturers make a genuine commitment to vaccine equity.
WHO is calling on countries to fulfil their dose-sharing pledges immediately and to swap their near-term vaccine deliveries with COVAX and AVAT (African COVID-19 Vaccine Acquisition Task Team); WHO is also calling on manufacturers to prioritize supplies to COVAX and partners, and for countries and manufacturers to facilitate the sharing of technology, know-how and intellectual property to support regional vaccine manufacturing.
Even as countries focus on ending this pandemic, the world must also prepare for future pandemics and other health emergencies.
COVID-19 caught the world – including wealthy nations – unprepared for a pandemic of this speed and scale. It hit vulnerable populations particularly hard and exacerbated inequalities.
WHO urges all countries to break the cycle of ‘panic and neglect’ seen after previous health emergencies, and commit adequate financial resources, as well as political will, to strengthening health emergency preparedness across the globe…
Milestones :: Perspectives :: Research
Statement by White House Press Secretary Jen Psaki on COVID-19 Summit
September 17, 2021 • Statements and Releases
On Wednesday September 22 President Biden will convene a virtual COVID Summit on the margins of the UN General Assembly. This meeting is about expanding and enhancing our shared efforts to defeat COVID-19, building out from previous gatherings of world leaders and ministers in fora like the G7, G20, and Act Accelerator to rally civil society, NGOs, philanthropists, and industry along with world leaders and align on a common vision for defeating COVID-19 together.
We will be asking participants to commit to of a higher level of ambition across four themes:
:: Vaccinate the World by enhancing equitable access to vaccines and getting shots in arms.
:: Save Lives Now by solving the oxygen crisis, and making tests, therapeutics and PPE more available.
:: Build Back Better by establishing a sustainable health security financing mechanism, and global leadership for emerging threats.
:: Calling the World to Account by aligning around targets, tracking progress, and supporting one another in fulfilling our commitments.
We will have more to share on the COVID Summit in the coming days.
Milestones :: Perspectives :: Research
International organizations, vaccine manufacturers agree to intensify cooperation to deliver COVID-19 vaccines
16 September 2021
The heads of the International Monetary Fund, World Bank Group, World Health Organization and World Trade Organization met with the CEOs of leading vaccine manufacturing companies to discuss strategies to improve access to COVID-19 vaccines, especially in low— and lower middle-income countries and in Africa. The Task Force expressed concerns that without urgent steps the world is unlikely to achieve the end-2021 target of vaccinating at least 40% of the population in all countries — a critical milestone to end the pandemic and for global economic recovery
The Task Force members noted that, despite adequate total global vaccine production in the aggregate, the doses are not reaching low- and lower middle-income countries in sufficient amounts, resulting in a crisis of vaccine inequity. The Task Force encouraged countries that have contracted high amounts of vaccine doses, and vaccine manufacturers, to come together in good faith to urgently accelerate COVID-19 vaccine supplies to COVAX and AVAT, two multilateral mechanisms that are crucial for equitable distribution of vaccines.
Task Force members welcomed the willingness of the CEOs to work collectively with them to end vaccine inequity and their readiness to form a technical working group with the Task Force to exchange and coordinate information on vaccine production and deliveries.
The Task Force stressed that if the 40% coverage threshold is to be reached in all countries by the end of 2021, the following actions need to be taken immediately by governments and vaccine manufacturers:
:: Release doses to low- and lower middle-income countries: Task Force members take note that countries with high vaccination rates have collectively pre-purchased over two billion doses in excess of what is required to fully vaccinate their populations. The Task Force calls again on those countries to urgently:
swap their near-term delivery schedules with COVAX and AVAT,
fulfill their dose donation pledges with unearmarked upfront deliveries to COVAX, and
iii) release vaccine companies from options and contracts so those doses can be delivered to people in low- and lower middle-income countries. In addition, vaccine manufacturers should prioritize and fulfill their contracts to COVAX and AVAT.
:: Transparency on supply of vaccines: To ensure that doses reach countries that need them the most, particularly low- and lower middle-income countries, the Task Force calls on vaccine manufacturers to share details on month-by-month delivery schedules for all vaccine shipments, especially for COVAX and AVAT. In its remarks, WHO emphasized its call for a moratorium on booster doses until the end of 2021, with the exception of the immune-compromised, to help optimize supply to low-income countries.
:: Eliminate export restrictions, prohibitions: The Task Force calls on all countries to urgently address export restrictions, high tariffs and customs bottlenecks on COVID-19 vaccines and the raw materials and supplies required for the production and timely distribution of vaccines.
:: Regulatory streamlining and harmonization: The Task Force calls on all regulatory authorities around the world to create regulatory consistency and standardization on the approval of vaccines, and to support the acceptance of the WHO Emergency Use Listing procedure. In parallel, efforts should be made to boost production of vaccines, diagnostics and treatments globally and expedite equitable delivery of such lifesaving tools to developing countries.
Milestones :: Perspectives :: Research
Leaders Make Urgent Call To Accelerate Vaccination Globally And In Africa
14 September 2021 News release Geneva
WHO Director-General Dr Tedros Adhanom Ghebreyesus and a group of global health leaders today issued an urgent call for vaccine equity globally and in Africa in particular. The leaders stressed that the worst pandemic in the last hundred years will not end unless and until, there is genuine global cooperation on vaccine supply and access. They also reiterated the WHO’s global vaccination target for 70% of the population of all countries to be vaccinated by mid- 2022.
Dr Tedros was joined by Dr Seth Berkley, CEO Gavi, Strive Masiyima, AU Special Envoy for COVID- 19, Dr John Nkengasong, Africa CDC Director, Professor Benedict Oramah, President and Chairman of the Board of Directors, Afreximbank, Dr Vera Songwe, UN Under- Secretary- General and Executive Secretary of the Economic Commission For Africa and Dr Matshidiso Moeti, WHO Regional Director for Africa.
The press conference followed two days of meetings among the leaders, with Richard Hatchett, Chief Executive Officer of CEPI joining the meetings as well…
Milestones :: Perspectives :: Research
Africa faces 470 million COVID-19 vaccine shortfall in 2021
WHO – Africa Region
16 September 2021
Brazzaville – As the COVAX Facility is forced to slash planned COVID-19 vaccine deliveries to Africa by around 150 million this year, the continent faces almost 500 million doses short of the global year-end target of fully vaccinating 40% of its population. This shortfall comes as Africa tops 8 million COVID-19 cases this week.
With the cutback COVAX is now expected to deliver 470 million doses to Africa this year. These will be enough to vaccinate just 17% of the population, far below the 40% target. An additional 470 million doses are needed to reach the end-year target even if all planned shipments via COVAX, a multilateral initiative aimed at guaranteeing global access to lifesaving COVID-19 vaccines, and the African Union are delivered.
“Export bans and vaccine hoarding have a chokehold on vaccine supplies to Africa. As long as rich countries lock COVAX out of the market, Africa will miss its vaccination goals. The huge gap in vaccine equity is not closing anywhere near fast enough. It is time for vaccine manufacturing countries to open the gates and help protect those facing the greatest risk,” said Dr Matshidiso Moeti, World Health Organization (WHO) Regional Director for Africa.
As export bans, challenges in boosting production at COVAX manufacturing sites and delays in filing for regulatory approvals for new vaccines constrain deliveries, COVAX has called for donor countries to share their supply schedules to give more clarity on deliveries.
COVAX has also called for countries with enough vaccines to give up their place in the queue for deliveries.Manufacturers must deliver to COVAX in line with firm commitments, and countries that are well-advanced with vaccinations must expand and accelerate donations, ensuring doses are available in larger, more predictable volumes and with longer shelf lives.
About 95 million more doses are set to arrive in Africa via COVAX throughout September, which will be the largest shipment the continent receives for any month so far. Yet even as deliveries pick up, Africa has been able to fully vaccinate just 50 million people, or 3.6% of its people.
Around 2% of the nearly 6 billion doses given globally have been administered in Africa. The European Union and the United Kingdom have vaccinated over 60% of their people and high-income countries have administered 48 times more doses per person than low-income nations.
“The staggering inequity and severe lag in shipments of vaccines threatens to turn areas in Africa with low vaccination rates into breeding grounds for vaccine-resistant variants. This could end up sending the whole world back to square one,” said Dr Moeti…
Milestones :: Perspectives :: Research
UNHCR warns of dire consequences for refugees from COVID-19 underfunding
This is a summary of what was said by UNHCR’s Chief of Public Health Section, Ann Burton – to whom quoted text may be attributed – at today’s press briefing at the Palais des Nations in Geneva.
17 September 2021 |
UNHCR, the UN Refugee Agency, calls for more global attention and funding support, to counter the impact of COVID-19 on forcibly displaced people worldwide.
The COVID-19 emergency tops the list of UNHCR’s top-10 underfunded situations in 2021. Only one third of the budgeted requirements of US$ 924 million has been received, leaving a yawning gap in UNHCR’s ability to protect the most vulnerable from the fallout of the pandemic.
Despite progress in many locations where UNHCR is working, we continue to see new cases and people continue to die. While safe and effective vaccines can relieve pressures on health systems and save lives, vaccine inequity continues to hit the hardest in many refugee-hosting states. We know that 86 per cent of refugees are hosted in developing countries. However, some 80 per cent of all vaccine doses have been given in high- and upper middle-income countries.
At the same time, low-income countries, hosting the bulk of the world’s refugees, have the least resilient health systems and are struggling to cope with the needs of their own populations – before we add the extra needs posed by hosting refugees. As UNHCR, we reiterate our call on states to share excess doses with COVAX in a timely way, to address the global vaccine inequity and avoid prolonging the pandemic.
Until now, we have been very encouraged by the overwhelming response of hosting states in including refugees in the vaccine roll-out and urge them to continue to do so. However, we have seen that many barriers to vaccine access remain. UNHCR stands ready to support states to overcome some of these barriers – provided we have the means to do so – for example, by creating information materials in refugee languages suitable for low literacy levels.
The pandemic hurts forcibly displaced and stateless people in ways that reach far beyond the risk posed by the virus itself. And the failure to adequately fund the response only deepens their plight.
Milestones :: Perspectives :: Research
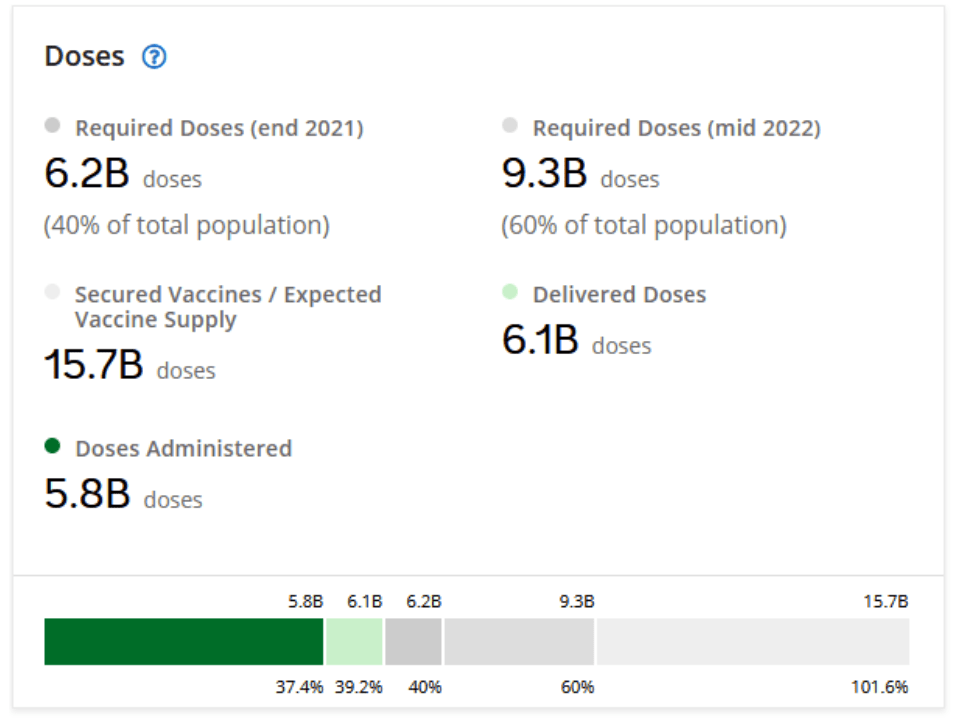
COVID Vaccines – OCHA:: HDX
COVID-19 Data Explorer: Global Humanitarian Operations
COVID-19 Vaccine Roll-out
Sep 18, 2021 | COVAX (WHO,GAVI,CEPI), UNDESA, Press Reports | DATA
Global COVID-19 Figures: 227M total confirmed cases; 4.7M total confirmed deaths
Global vaccines administered: 5.88B
Number of Countries: 29 [29 week ago]
COVAX Allocations Round 4-6 (Number of Doses): 120M [120M week ago]
COVAX Delivered (Number of Doses): 87M [85M week ago]
Other Delivered (Number of Doses): 130M [110M week ago]
Total Delivered (Number of Doses): 200M [210M week ago]
Total Administered (Number of Doses): 190M [170M week ago]
::::::
Multilateral Leaders Task Force on COVID-19 [IMF, World Bank Group, WHO, WTO]
A joint initiative from the International Monetary Fund, World Bank Group, World Health Organization, and World Trade Organization to accelerate access to COVID-19 vaccines, therapeutics and diagnostics by leveraging multilateral finance and trade solutions, particularly in low- and middle-income countries.
Website accessed 18 Sep 2021: https://data.covid19taskforce.com/data The global view below is complemented by country-specific dashboards here.