Coronavirus [COVID-19] – WHO
Public Health Emergency of International Concern (PHEIC)
https://www.who.int/emergencies/diseases/novel-coronavirus-2019
Weekly Epidemiological and Operational updates
Last update: 5 Jun 2021
Confirmed cases :: 172 242 495 [week ago: 169 118 995]
Confirmed deaths :: 3 709 397 [week ago: 3 519 175]
Vaccine doses administered: 1 638 006 899 [week ago: 1 546 316 352]
::::::
Weekly epidemiological update on COVID-19 – 1 June 2021
In the past week, the number of new COVID-19 cases and deaths continues to decrease, with over 3.5 million new cases and 78 000 new deaths reported globally. Although the number of global cases and deaths continued to decrease for a fifth and fourth consecutive week respectively, case and death incidences remain at high levels and significant increases have been reported in many countries in all regions.
In this edition, special focus updates are provided on:
:: SARS-CoV-2 Variants of Interest (VOIs) and Variants of Concern (VOCs), including the introduction of new labels for public communications, updates on VOI and VOC classifications and the global geographical distribution of VOCs Alpha (B.1.1.7), Beta (B.1.351), Gamma (P.1) and Delta (B.1.617.2).
:: Lessons learned during the early phases of rolling out COVID-19 vaccines, with a particular focus on low-and-middle income countries (LMICs).
[Excerpt, p. 5]

Weekly operational update on COVID-19 31 May 2021
Overview
In this edition of the COVID-19 Weekly Operational Update, highlights of country-level actions and WHO support to countries include:
:: Launch of “Strengthening Civil Society Engagement in the COVID-19 Response” in Guyana
:: Infection prevention and control (IPC) critical for COVID-19 care and recovery in India
:: Intra-Action Review (IAR) in Montenegro
:: Vanuatu the seventh country in the Pacific islands to receive COVID-19 vaccine doses from the COVAX Facility
:: Indian Sign Language course extends OpenWHO reach and accessibility amidst the COVID-19 pandemic
:: Utilizing message testing to ensure behavioural messages resonate with the intended audience and a global consultative meeting on Intra-Action Reviews (IARs) and simulation exercises
:: Regular updates on WHO’s resource requirements and funds received to support countries in implementing the COVID-19 Strategic Preparedness and Response Plan (SPRP) 2021, WHO/PAHO procurement of critical supplies, and progress on a subset of indicators from the SPRP 2021 Monitoring and Evaluation Framework
::::::
::::::
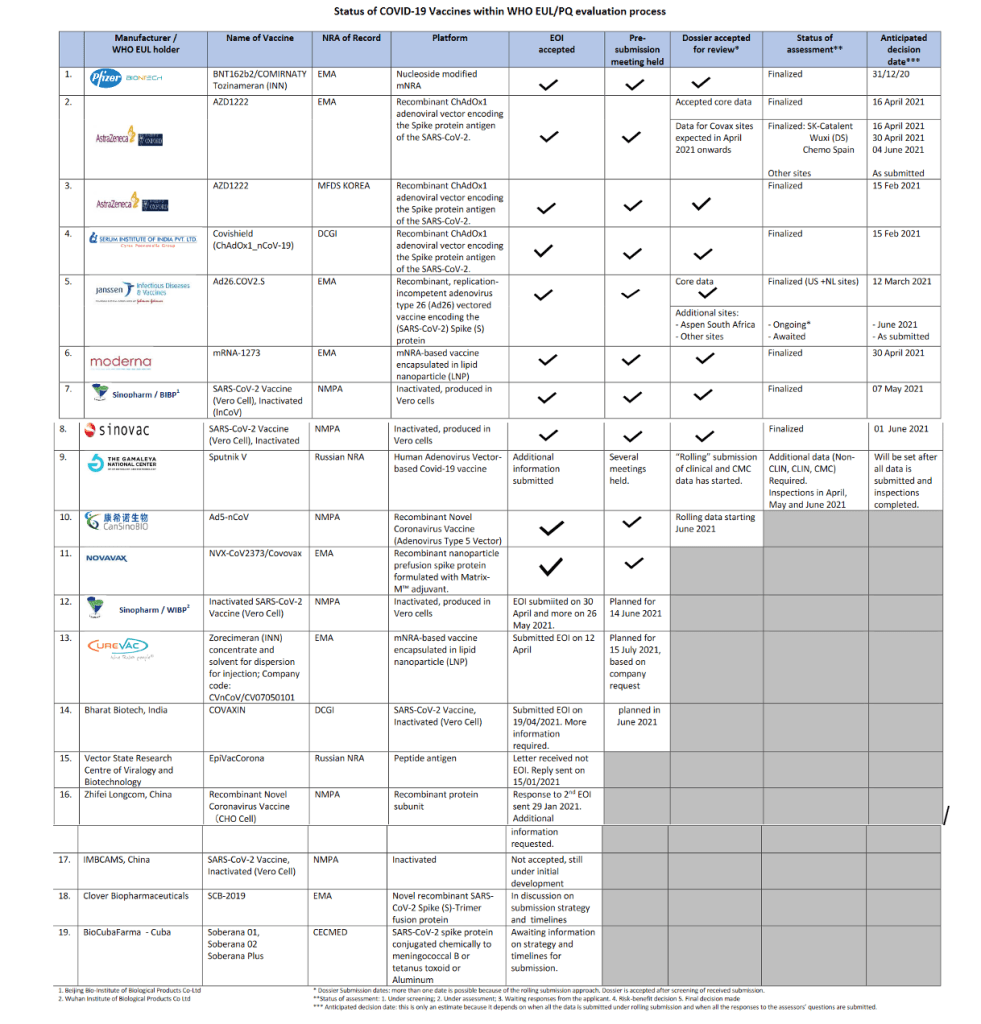
Draft landscape and tracker of COVID-19 candidate vaccines
28 May 2021 | Publication
The COVID-19 candidate vaccine landscape and tracker database compiles detailed information on COVID-19 vaccine candidates in development.
The landscape is updated regularly – twice a week (Tuesday and Friday, 17:00 CET).

Status of COVID-19 Vaccines within WHO EUL/PQ evaluation process 28 May 2021
For 19 vaccine candidates, presents Manufacturer, Name of Vaccine, NRA of Record, Platform, EOI Accepted Status, Pre-submission Meeting Held Status, Dossier Accepted for Review, Status of Assessment; Anticipated/Completed Decision Date
[click on the link above for full scale view]
03 June 2021
WHO validates Sinovac COVID-19 vaccine for emergency use and issues interim policy recommendations
1 June 2021 News release
WHO today validated the Sinovac-CoronaVac COVID-19 vaccine for emergency use, giving countries, funders, procuring agencies and communities the assurance that it meets international standards for safety, efficacy and manufacturing. The vaccine is produced by the Beijing-based pharmaceutical company Sinovac.
“The world desperately needs multiple COVID-19 vaccines to address the huge access inequity across the globe,” said Dr Mariângela Simão, WHO Assistant-Director General for Access to Health Products. “We urge manufacturers to participate in the COVAX Facility, share their knowhow and data and contribute to bringing the pandemic under control.”…
In the case of the Sinovac-CoronaVac vaccine, the WHO assessment included on-site inspections of the production facility.
The Sinovac-CoronaVac product is an inactivated vaccine. Its easy storage requirements make it very manageable and particularly suitable for low-resource settings.
WHO’s Strategic Advisory Group of Experts on Immunization (SAGE) has also completed its review of the vaccine. On the basis of available evidence, WHO recommends the vaccine for use in adults 18 years and older, in a two-dose schedule with a spacing of two to four weeks. Vaccine efficacy results showed that the vaccine prevented symptomatic disease in 51% of those vaccinated and prevented severe COVID-19 and hospitalization in 100% of the studied population.
Few older adults (over 60 years) were enrolled in clinical trials, so efficacy could not be estimated in this age group. Nevertheless, WHO is not recommending an upper age limit for the vaccine because data collected during subsequent use in multiple countries and supportive immunogenicity data suggest the vaccine is likely to have a protective effect in older persons. There is no reason to believe that the vaccine has a different safety profile in older and younger populations. WHO recommends that countries using the vaccine in older age groups conduct safety and effectiveness monitoring to verify the expected impact and contribute to making the recommendation more robust for all countries…