Milestones :: Perspectives :: Research
Europe: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
European Medicines Agency
News & Press Releases
Meeting highlights from the Pharmacovigilance Risk Assessment Committee (PRAC) 30 August – 2 September 2021
News 03/09/2021
COVID-19 vaccines: EMA reviewing cases of multisystem inflammatory syndrome
EMA’s safety committee (PRAC) is assessing whether there is a risk of multisystem inflammatory syndrome (MIS) with COVID-19 vaccines following a report of MIS with Comirnaty. The case occurred in a 17-year old male in Denmark who has since fully recovered.
Some cases of MIS were also reported in the EEA following vaccination with other COVID-19 vaccines.1
MIS is a serious inflammatory condition affecting many parts of the body and symptoms can include tiredness, persistent severe fever, diarrhoea, vomiting, stomach pain, headache, chest pain and difficulty breathing. MIS has previously been reported following COVID-19 disease. The Danish patient, however, had no history of COVID-19.
MIS is rare and its incidence rate before the COVID-19 pandemic estimated from 5 European countries was around 2 to 6 cases per 100,000 per year in children and adolescents below 20 years of age and below 2 cases per 100,000 per year in adults aged 20 years or more.
At this stage, there is no change to the current EU recommendations for the use of COVID-19 vaccines…
News: ECDC and EMA highlight considerations for additional and booster doses of COVID-19 vaccines (new)
Last updated: 02/09/2021
Based on current evidence, there is no urgent need for the administration of booster doses of vaccines to fully vaccinated individuals in the general population, according to a technical report issued yesterday by the European Centre for Disease Prevention and Control (ECDC). The report also notes that additional doses should already be considered for people with severely weakened immune systems as part of their primary vaccination.
Evidence on vaccine effectiveness and duration of protection shows that all vaccines authorised in the EU/EEA are currently highly protective against COVID-19-related hospitalisation, severe disease and death, while about one out of three adults in the EU/EEA over 18 years is still currently not fully vaccinated. In this situation, the priority now should be to vaccinate all those eligible individuals who have not yet completed their recommended vaccination course. To complement vaccination efforts, it is also crucial to continue applying measures such as physical distancing, hand and respiratory hygiene, and using face masks where needed, in particular in high-risk settings such as long-term care facilities or hospital wards with patients at risk of severe COVID-19…
::::::
European Centre for Disease Prevention and Control
https://www.ecdc.europa.eu/en
Latest Updates
News
ECDC awards 77 M€ to strengthen EU/EEA Member States’ capacities to detect and identify SARS-CoV-2 variants
News story – 3 Sep 2021
ECDC has awarded more than 77 M€ to 24 EU/EEA countries to strengthen whole genome sequencing and RT-PCR infrastructures within the countries’ national public health programmes.
The immediate objective is to reinforce countries’ capacities for early detection and enhanced monitoring of emergent and known SARS-CoV-2 variants. The support is a response to a request to ECDC by the European Commission to implement a number of activities that were launched as part of the “HERA Incubator ” aiming to support the rapid detection of SARS-CoV-2 variants across the EU…
News
ECDC and EMA highlight considerations for additional and booster doses of COVID-19 vaccines
Press release – 2 Sep 2021
Based on current evidence, there is no urgent need for the administration of booster doses of vaccines to fully vaccinated individuals in the general population, according to a technical report issued by the European Centre for Disease Prevention and Control (ECDC). The report also notes that additional doses should already be considered for people with severely weakened immune systems as part of their primary vaccination.
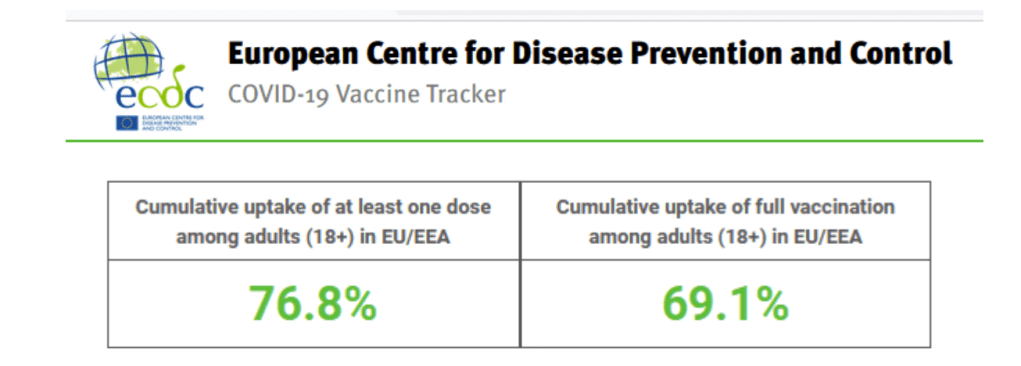
Data as of 2021-09-03
https://vaccinetracker.ecdc.europa.eu/public/extensions/COVID-19/vaccine-tracker.html#uptake-tab
European Commission
https://ec.europa.eu/commission/presscorner/home/en
Questions and answers 3 September 2021
Questions and Answers: The EU and AstraZeneca agree on COVID-19 vaccine supply and on ending litigation
Press release 3 September 2021
Coronavirus: The EU and AstraZeneca agree on COVID-19 vaccine supply and on ending litigation
Today the EU and AstraZeneca have reached an agreement which will secure the delivery of the remaining COVID-19 vaccine doses to Member States under the terms of the Advance Purchase Agreement concluded on 27 August 2020 with AstraZeneca.
Press release 31 August 2021
Coronavirus: 70% of the EU adult population fully vaccinated
Today, the EU has reached a crucial milestone with 70% of the adult population now fully vaccinated. In total, over 256 million adults in the EU have now received a full vaccine course.