Milestones :: Perspectives :: Research
Europe: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
European Medicines Agency
News & Press Releases
News: EMA welcomes new Head of International Affairs (new)
Last updated: 01/10/2021
EMA is pleased to announce the appointment of Martin Harvey Allchurch as its new head of International Affairs as of 1 October 2021.
Martin Harvey Allchurch brings a wealth of EMA and international experience to this role, having worked in a number of management positions at the Agency, as well as at Unitaid, the World Health Organisation-hosted partnership for access to innovative healthcare in low- and middle-income countries…
News: Meeting highlights from the Pharmacovigilance Risk Assessment Committee (PRAC) 27-30 September 2021 (new)
PRAC, Last updated: 01/10/2021
…PRAC assessment of rare cases of venous thromboembolism with COVID-19 Vaccine Janssen
The PRAC has concluded that there is a possible link to rare cases of venous thromboembolism (VTE) with COVID-19 Vaccine Janssen…
…The PRAC has now reviewed new evidence from the study described above, as well as new evidence from another large clinical study. In this second study, there was no increase in venous thromboembolic events among individuals who received COVID-19 Vaccine Janssen. The PRAC also reviewed evidence from the post marketing setting – that is data gathered while the vaccine is used in the context of vaccination campaigns. When taking all evidence into account, the committee concluded that there is a reasonable possibility that rare cases of VTE are linked to vaccination with COVID-19 Vaccine Janssen.
The committee is therefore recommending listing VTE as a rare side effect of COVID-19 Vaccine Janssen in the product information, together with a warning to raise awareness among healthcare professionals and people taking the vaccine, especially those who may have an increased risk of VTE…
News: Transatlantic Taskforce continues international fight against antimicrobial resistance (new)
Last updated: 30/09/2021
News: EMA implements new measures to minimise animal testing during medicines development (new)
Last updated: 29/09/2021
::::::
European Centre for Disease Prevention and Control
https://www.ecdc.europa.eu/en
Latest Updates
Publication
Rapid Risk Assessment: Assessing SARS-CoV-2 circulation, variants of concern, non-pharmaceutical interventions and vaccine rollout in the EU/EEA, 16th update
Risk assessment
30 Sep 2021
Since its emergence in March 2021, the B.1.617.2 (Delta) variant of concern (VOC) has rapidly become predominant across the European Union/European Economic Area (EU/EEA). More than 99% of newly reported cases are attributed to this variant. The Delta variant has demonstrated a significant transmission advantage relative to previously circulating SARS-CoV-2 strains. However, full vaccination remains protective against severe outcomes such as hospitalisation, admission to intensive care and death. Currently available vaccines have played a crucial role in limiting viral circulation and in particular, limiting the impact of infections by the Delta variant.
Despite the fact that over 565 million vaccine doses have been administered in the EU/EEA so far, only 61.1% (range: 18.4–79.4%) of the total population in the EU/EEA have been fully vaccinated to date. The total population includes children and adolescents for whom the vaccine is not available or who may not be included in national target groups yet. There is considerable inter-country and sub-national variation in vaccine uptake, resulting in large proportions of the EU/EEA population remaining susceptible to SARS-CoV-2 infection.
Modelling scenarios that consider vaccination coverage, vaccine effectiveness, natural immunity and population contact rates—in the context of continued Delta circulation—indicate that the potential burden of disease risk in the EU/EEA from the Delta variant is high between now to the end of November, unless vaccination coverage can be increased rapidly in the total population in the next few weeks.
News
High risk of autumn surge in COVID-19 cases and deaths in countries with insufficient vaccination coverage, warns ECDC
News story – 30 Sep 2021
EU/EEA countries that have not yet achieved high enough COVID-19 vaccination coverage in their total populations, which are planning to relax non-pharmaceutical interventions during the next two weeks, run a high risk of experiencing a significant surge of cases, hospitalisations and mortality from now until the end of November. This is indicated by a new SARS-CoV-2 Rapid Risk Assessment published today by the European Centre for Disease Prevention and Control (ECDC).
::::::
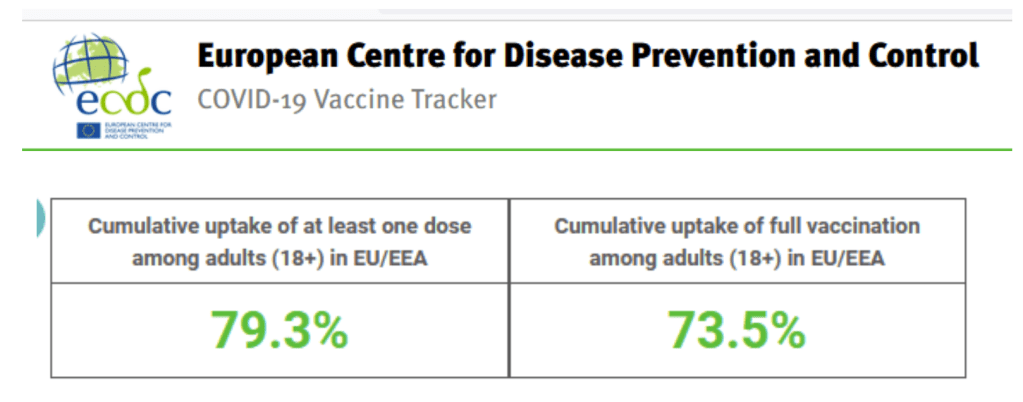
Accessed 2 Oct 2021
https://vaccinetracker.ecdc.europa.eu/public/extensions/COVID-19/vaccine-tracker.html#uptake-tab
::::::
European Commission
https://ec.europa.eu/commission/presscorner/home/en
Press release 2 October 2021
EU Humanitarian Air Bridge delivers life-saving medical aid to Afghanistan
Press release 1 October 2021
Code of Practice on disinformation: Commission welcomes new prospective signatories and calls for strong and timely revision
The topic of disinformation remains high on the Commission’s agenda. Eight new prospective signatories joined the revision process of the Code of Practice on disinformation during the latest signatories Assembly meeting yesterday.
Press release 28 September 2021
European Health Union: Towards a reform of EU’s pharmaceutical legislation
Today, as part of its work to create a future-proof and crisis-resilient regulatory framework for the pharmaceutical sector, the Commission has published a public consultation on the revision of the EU’s pharmaceutical legislation.