WHO Director General Speeches [selected]
https://www.who.int/director-general/speeches
Selected
No new digest content identified
::::::
::::::
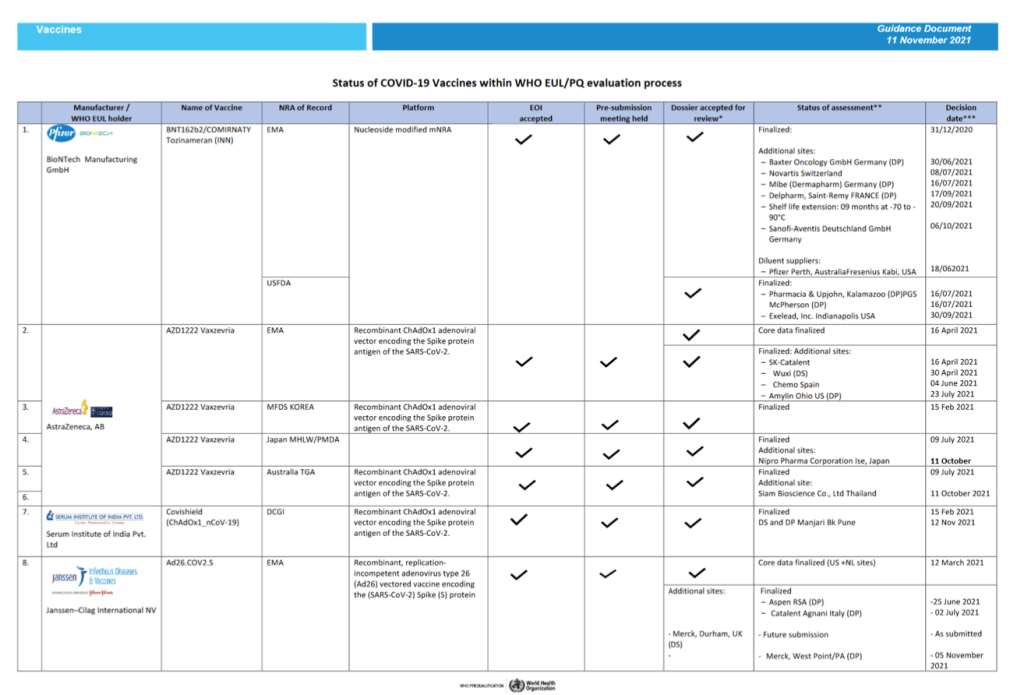
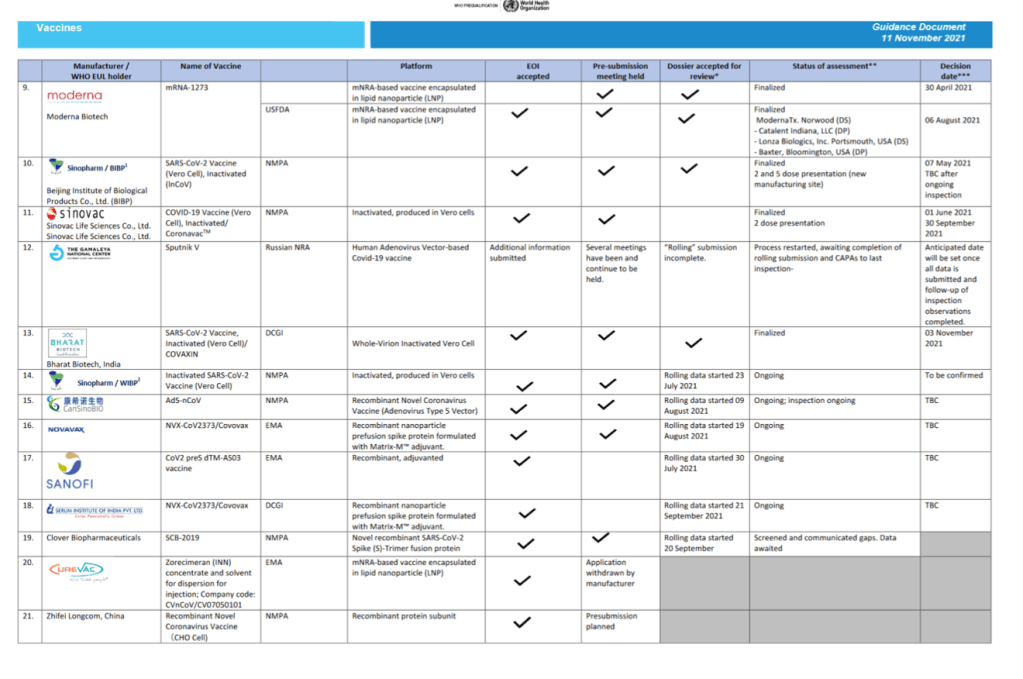
Status of COVID-19 Vaccines within WHO EUL/PQ evaluation process 11 November 2021
For 24 vaccine candidates, presents Manufacturer, Name of Vaccine, NRA of Record, Platform, EOI Accepted Status, Pre-submission Meeting Held Status, Dossier Accepted for Review, Status of Assessment; Anticipated/Completed Decision Date
[Full scale view available at title link above]

::::::
COVID Vaccine Developer/Manufacturer Announcements
[relevant press releases/announcement from organizations from WHO EUL/PQ listing above]
AstraZeneca
Press Releases
New analyses of two AZD7442 COVID-19 Phase III trials in high-risk populations confirm robust efficacy and long-term prevention
18 November 2021
Two billion doses of AstraZeneca’s COVID-19 vaccine supplied to countries across the world less than 12 months after first approval
16 November 2021
Bharat Biotech
Press Releases – No new digest announcements identified
BioCubaFarma – Cuba
Últimas Noticias – [Website not responding at inquiry; receiving 403-Forbidden]
CanSinoBIO
News – [Website not responding at inquiry]
Clover Biopharmaceuticals – China
News
Nov 18,2021
Clover and CEPI Expand Partnership for COVID-19 Vaccine Candidate to Include Evaluation as a Booster
:: CEPI commits up to an additional US$36.9 million for a total investment of up to $397.4 million to support the development of Clover’s COVID-19 vaccine candidate, SCB-2019 (CpG 1018/Alum)
:: CEPI funding supports development of SCB-2019 (CpG 1018/Alum) for primary vaccination across all age groups (pediatric, adolescent, adults, and elderly) and development as a potential booster
Curevac [Bayer Ag – Germany]
News
November 18, 2021
CureVac Publishes in Nature Preclinical Data of Second-Generation COVID-19 Candidate, CV2CoV, Demonstrating Comparable Antibody Levels to Licensed mRNA Vaccine
Gamaleya National Center
Latest News and Events – No new digest announcements identified [See Russia/RFID below]
IMBCAMS, China
Home – No new digest announcements identified
Janssen/JNJ
Press Releases – No new digest announcements identified
Moderna
Press Releases
November 19, 2021
U.S. CDC Advisory Committee on Immunization Practices Recommends Booster Vaccination with Moderna’s COVID-19 Vaccine
November 19, 2021
Moderna Announces FDA Authorization of Booster Dose of COVID-19 Vaccine in the U.S. for Adults 18 Years and Older
November 18, 2021
Moderna Announces First Participant Dosed in Phase 2/3 Study of its mRNA Respiratory Syncytial Virus (RSV) Vaccine
November 17, 2021
Moderna Files for Authorization of its COVID-19 Vaccine with Health Canada to Include Children Ages 6-11 Years
November 15, 2021
Health Canada Authorizes Booster Dose of Moderna’s COVID-19 Vaccine in Individuals 18 Years of Age and Older
Novavax
Press Releases
Novavax Statement on Publication of Positive Results from First Study of Co-administered COVID-19 and Influenza Vaccines Nov 18, 2021
Novavax Confirms European Medicines Agency Review of COVID-19 Vaccine Filing for Conditional Marketing Authorization Nov 17, 2021
Novavax and Serum Institute of India Receive Emergency Use Authorization for COVID-19 Vaccine in the Philippines Nov 17, 2021
Novavax Announces Submission of Biologics License Application in South Korea for Approval of NVX-CoV2373 Nov 15, 2021
Pfizer
Recent Press Releases
11.19.2021
Pfizer and BioNTech Receive Expanded U.S. FDA Emergency Use Authorization of COVID-19 Vaccine Booster to Include Individuals 18 and Older
11.16.2021
Pfizer Seeks Emergency Use Authorization for Novel COVID-19 Oral Antiviral Candidate
11.16.2021
Pfizer and The Medicines Patent Pool (MPP) Sign Licensing Agreement for COVID-19 Oral Antiviral Treatment Candidate to Expand Access in Low- and Middle-Income Countries
[See COVID above for detail]
Sanofi Pasteur
Press Releases – No new digest announcements identified
Serum Institute of India
NEWS & ANNOUNCEMENTS
Wednesday, November 17, 2021
Novavax and Serum Institute of India Receive Emergency Use Authorization for COVID-19 Vaccine in the Philippines
Sinopharm/WIBPBIBP
News – No new digest announcements identified
Sinovac
Press Releases – No new digest announcements identified
Vector State Research Centre of Viralogy and Biotechnology
Home – No new digest announcements identified
Zhifei Longcom, China
[Anhui Zhifei Longcom Biologic Pharmacy Co., Ltd.]
[No website identified]
::::::
GSK
Press releases for media
17 November 2021 GSK and Vir Biotechnology announce United States government agreements to purchase sotrovimab, a COVID-19 treatment
Merck
News releases – No new digest announcements identified
Novartis
News – No new digest announcements identified
SK Biosciences
Press releases
SK bioscience Submits Biologics License Application to the KMFDS for Novavax Covid-19 Vaccine Candidate
The first submission of a protein-based COVID-19 vaccine candidate in Korea…
2021. 11. 15
Valneva
Press Releases
November 15, 2021
Valneva to Present its Chikungunya Vaccine Candidate at the ASTMH 2021 Annual Meeting