WHO Director General Speeches [selected]
https://www.who.int/director-general/speeches
Selected
25 November 2021
Speech
Director-General’s opening remarks at Member States Information Session on COVID-19 – 25 November 2021
24 November 2021
Speech
WHO Director-General’s opening remarks at the media briefing on COVID-19 – 24 November 2021
Data suggest that before the arrival of the Delta variant, vaccines reduced transmission by about 60%. With Delta, that has dropped to about 40%. We cannot say this clearly enough: even if you are vaccinated, continue to take precautions to prevent becoming infected yourself, and to infecting someone else who could die. And we continue to call on all governments to implement a comprehensive and tailored approach of public health and social measures to prevent transmission, take the pressure off health systems and save lives.
Yesterday, C-TAP and the Medicines Patent Pool finalized a licensing agreement with the Spanish National Research Council for a serological antibody test. This is the first transparent, global and non-exclusive license for a COVID-19 health tool. I would like to thank the government of Spain for its support for C-TAP, and I also thank His Excellency President Carlos Alvarado Quesada of Costa Rica for his leadership in initiating C-TAP last year. We hope this will be the first of many licenses to be shared through C-TAP.
With the WTO Ministerial Conference next week and with the vast majority of countries now firmly supporting a waiver on intellectual property rights under the TRIPS agreement, I hope that consensus can be found and that we move forward.
Today marks the end of Antimicrobial Awareness Week. Antimicrobials, including antibiotics, antivirals, antifungals and antiparasitics, are the backbone of modern medicine. But the overuse and misuse of antimicrobials are undermining the effectiveness of these essential medicines. By using antimicrobials responsibly, and by following the advice of your health care provider, we can all play a part in preserving antimicrobials and preventing drug resistance.
23 November 2021
Speech
WHO Director-General’s opening remarks at the First meeting of the WHO Scientific Advisory Group for the Origins of Novel Pathogens (SAGO)
23 November 2021
Speech
WHO Director-General’s opening remarks at the 2021 SADC seminar on TRIPS waiver – 23 November 2021
…Let me leave you with three requests:
First, we seek the support of the Canadian government for the TRIPS waiver for COVID-19 products at the WTO Ministerial Conference that begins this week.
Second, we seek the support of all countries in speeding up negotiations that result in a text that countries can implement easily in their national legislation.
WHO strongly recommends that the waiver apply not only to vaccines, but also to diagnostics, therapeutics and other tools to prevent, diagnose and treat COVID-19.
Third, we urge all SADC members to explore all avenues to rapidly scale-up production, including through the use of TRIPS flexibilities, technology pools, voluntary licenses, and by investing in local production…
::::::
::::::
Status of COVID-19 Vaccines within WHO EUL/PQ evaluation process 11 November 2021
For 24 vaccine candidates, presents Manufacturer, Name of Vaccine, NRA of Record, Platform, EOI Accepted Status, Pre-submission Meeting Held Status, Dossier Accepted for Review, Status of Assessment; Anticipated/Completed Decision Date
[Full scale view available at title link above]
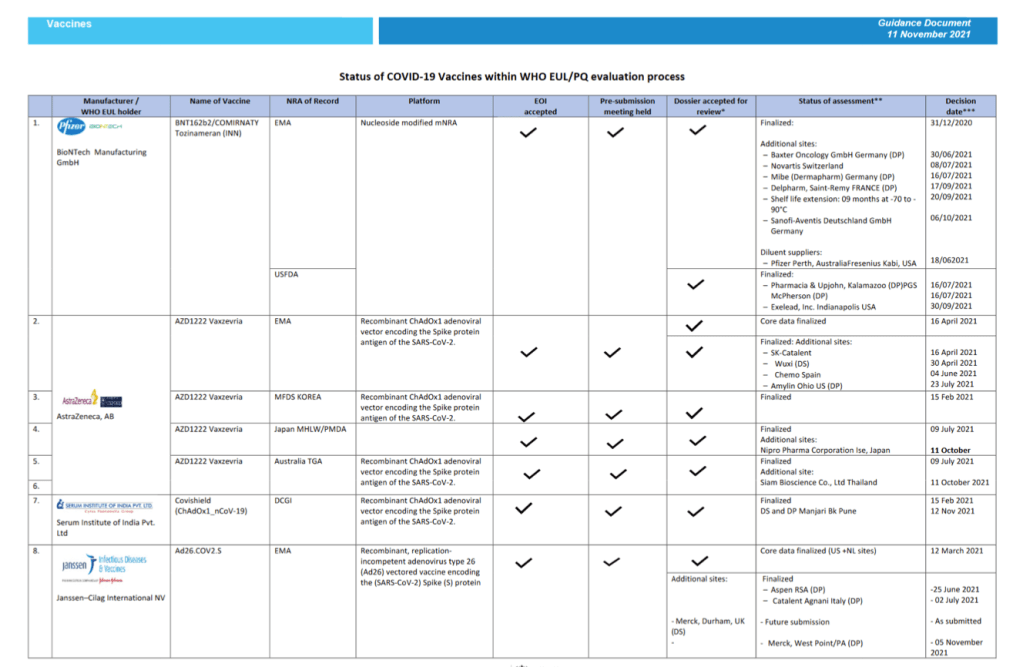
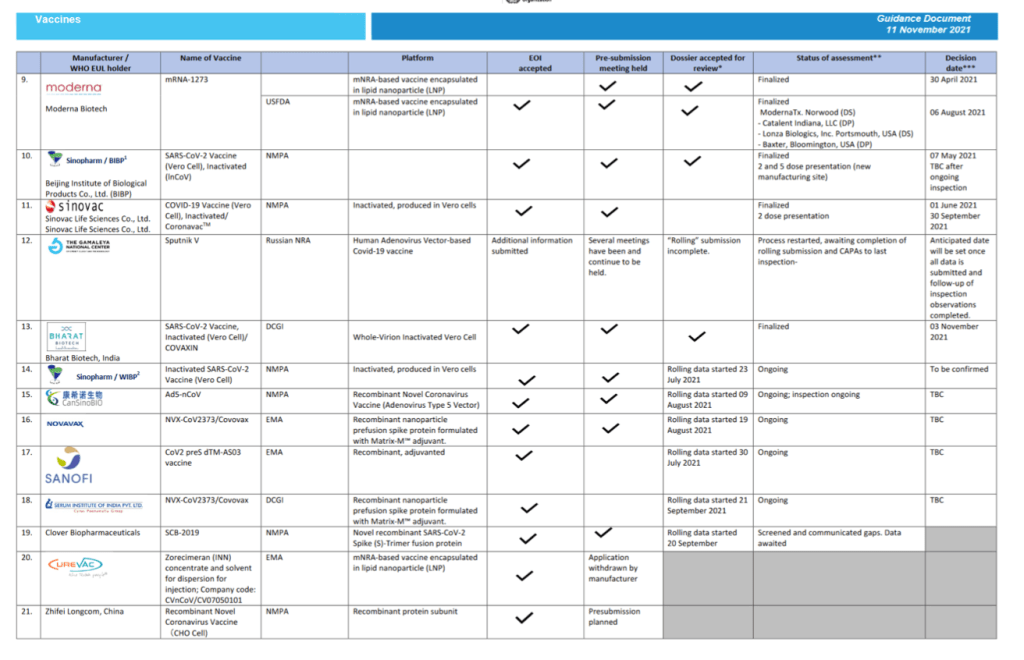
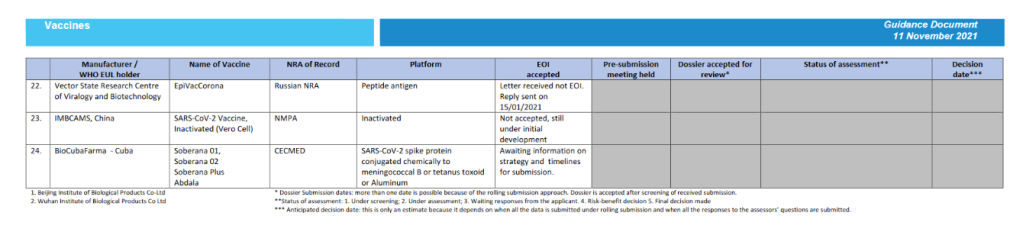
::::::
COVID Vaccine Developer/Manufacturer Announcements
[relevant press releases/announcement from organizations from WHO EUL/PQ listing above]
AstraZeneca
Press Releases
AstraZeneca unveils The Discovery Centre (DISC) in Cambridge
23 November 2021
:: State-of-the-art R&D facility designed to foster collaboration across world-class science ecosystem
:: Sustainable environmental engineering aligned to Company’s zero carbon future
AstraZeneca will today, in the presence of His Royal Highness The Prince of Wales, formally unveil The Discovery Centre (DISC) in Cambridge, UK – a state-of-the-art research and development (R&D) facility designed to the world’s highest environmental standards and accommodating over 2,200 research scientists.
The new £1bn facility will include the most advanced robotics, high-throughput screening and AI-driven technology. It will support AstraZeneca’s focus on specialised and precision medicines and foster the discovery and development of next generation therapeutics, including nucleotide-based, gene-editing and cell therapies…
Bharat Biotech
Press Releases – No new digest announcements identified
BioCubaFarma – Cuba
Últimas Noticias – [Website not responding at inquiry; receiving 403-Forbidden]
CanSinoBIO
News – [Website not responding at inquiry]
Clover Biopharmaceuticals – China
News
Nov 26,2021
Clover’s COVID-19 Vaccine Candidate Administered as Heterologous Booster in Investigator-Led Phase 2 Clinical Trial
The Phase 2 trial will evaluate SCB-2019 (CpG 1018/Alum) as a booster dose in individuals previously vaccinated with CoronaVac or recombinant Covid-19 vaccine (AstraZeneca/Fiocruz)
The Phase 2 trial is an investigator initiated study, sponsored by the Instituto D’Or de Pesquisa e Ensino (IDOR) and funded by the Bill & Melinda Gates Foundation
The study will evaluate the immunogenicity and safety of SCB-2019 in approximately 520 healthy adult participants at multiple sites in Brazil
Initial safety and immunogenicity data are anticipated in the first half of 2022
Curevac [Bayer Ag – Germany]
News – No new digest announcements identified
Gamaleya National Center
Latest News and Events – No new digest announcements identified [See Russia/RFID below]
IMBCAMS, China
Home – No new digest announcements identified
Janssen/JNJ
Press Releases
Nov 24, 2021 Canada
Johnson & Johnson COVID-19 Vaccine Fully Approved by Health Canada to Prevent COVID-19 in Individuals 18 years and Older
Vaccine demonstrates protection against COVID-19 related hospitalization and death, and across different variants
Moderna
Press Releases
November 26, 2021
Moderna Announces Strategy to Address Omicron (B.1.1.529) SARS-CoV-2 Variant
:: Company testing three existing COVID-19 vaccine booster candidates against the Omicron variant
:: Company announcing a new variant-specific vaccine candidate against Omicron (mRNA-1273.529)
CAMBRIDGE, Mass.–(BUSINESS WIRE)–Nov. 26, 2021– Moderna, Inc., (Nasdaq: MRNA) a biotechnology company pioneering messenger RNA (mRNA) therapeutics and vaccines, today announced updates to its strategy to address SARS-CoV-2 variants of concern, given the emergence of the B.1.1.529 (Omicron) variant.
The recently described Omicron variant includes mutations seen in the Delta variant that are believed to increase transmissibility and mutations seen in the Beta and Delta variants that are believed to promote immune escape. The combination of mutations represents a significant potential risk to accelerate the waning of natural and vaccine-induced immunity. A booster dose of an authorized vaccine represents the only currently available strategy for boosting waning immunity. The Moderna COVID-19 vaccine (mRNA-1273) is authorized as a booster for many populations at the 50 µg dose level. The Company is working rapidly to test the ability of the current vaccine dose to neutralize the Omicron variant and data is expected in the coming weeks.
Since early 2021, Moderna has advanced a comprehensive strategy to anticipate new variants of concern. This strategy includes three levels of response should the currently authorized 50 µg booster dose of mRNA-1273 prove insufficient to boost waning immunity against the Omicron variant…
Novavax
Press Releases
Novavax Files for Interim Authorization of COVID-19 Vaccine in Singapore Nov 24, 2021
Pfizer
Recent Press Releases
11.25.2021
Pfizer and BioNTech Receive Positive CHMP Opinion for COMIRNATY® in Children 5 to <12 Years of Age in the European Union
NEW YORK and MAINZ, Germany, November 25, 2021—Pfizer Inc. (NYSE: PFE, “Pfizer”) and BioNTech SE (Nasdaq: BNTX, “BioNTech”) today announced that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) issued a positive opinion on the administration of the companies’ COVID-19 vaccine COMIRNATY® in children 5 to under 12 years of age. The European Commission (EC) will review the CHMP recommendation and is expected to make a final decision on a variation to the Conditional Marketing Authorization in the near future…
11.22.2021
Follow-Up Data From Phase 3 Trial of Pfizer-BioNTech COVID-19 Vaccine Support Safety and High Efficacy in Adolescents 12 Through 15 Years of Age
:: Pfizer-BioNTech vaccine demonstrated 100% efficacy against COVID-19 in longer-term analysis, with no serious safety concerns identified
:: Data will support planned submissions for full regulatory approval of the vaccine in this age group in the U.S. and worldwide
Sanofi Pasteur
Press Releases – No new digest announcements identified
Serum Institute of India
NEWS & ANNOUNCEMENTS
Serum Institute of India (SII) re-starts COVAX exports; passes 1billion COVISHIELD dose
:: Total number of COVISHIELD doses produced now surpasses 1.25bn mark.
:: Resumption of global distribution of COVISHIELD via the COVAX mechanism and to low- and middle-income countries.
[See Milestones above for detail]
Sinopharm/WIBPBIBP
News – No new digest announcements identified
Sinovac
Press Releases
The 2nd International Conference on the Latest Researches, Experience Sharing on SINOVAC’s COVID-19 Vaccine Was Held 2021/11/26
Vector State Research Centre of Viralogy and Biotechnology
Home – No new digest announcements identified
Zhifei Longcom, China
[Anhui Zhifei Longcom Biologic Pharmacy Co., Ltd.]
[No website identified]
::::::
GSK
Press releases for media – No new digest announcements identified
Merck
News releases
Merck and Ridgeback Biotherapeutics Provide Update on Results from MOVe-OUT Study of Molnupiravir, an Investigational Oral Antiviral Medicine, in At Risk Adults With Mild-to-Moderate COVID-19 Nov 26, 2021
Novartis
News – No new digest announcements identified
SK Biosciences
Press releases – No new digest announcements identified
Valneva
Press Releases
Valneva Signs Purchase Agreement with European Commission for its Inactivated COVID-19 Vaccine VLA2001
Saint-Herblain (France), November 23, 2021 – Valneva SE (Nasdaq: VALN; Euronext Paris: VLA), a specialty vaccine company, today announced that it has signed an Advance Purchase Agreement (APA) with the European Commission (EC) to supply up to 60 million doses of its inactivated COVID-19 vaccine candidate, VLA2001, over two years. The agreement follows the announcement made earlier this month that the EC had approved the APA[1].