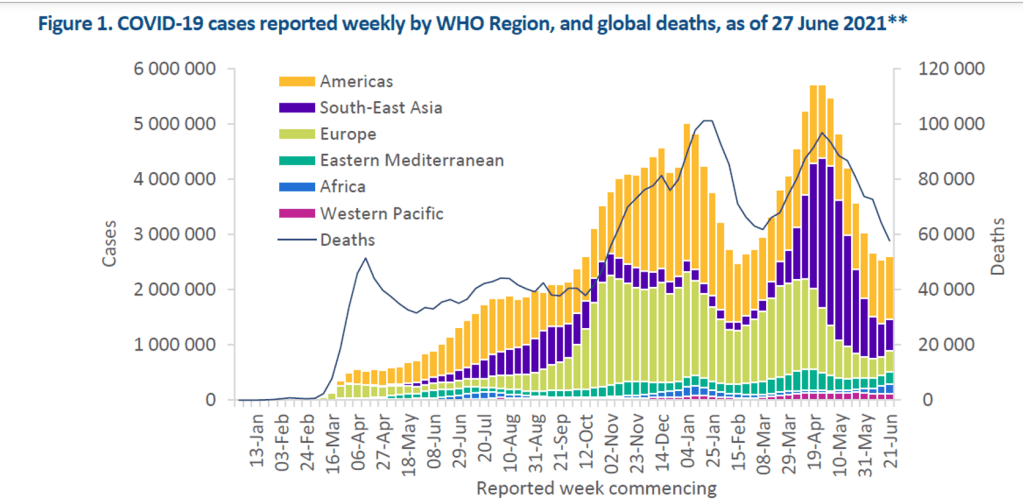
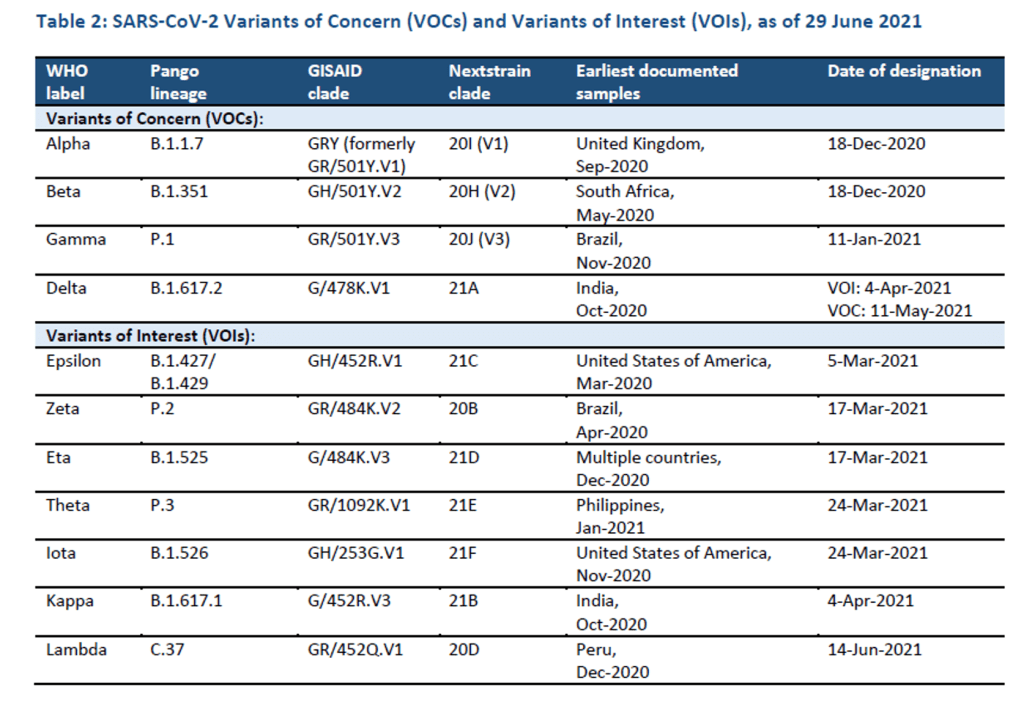
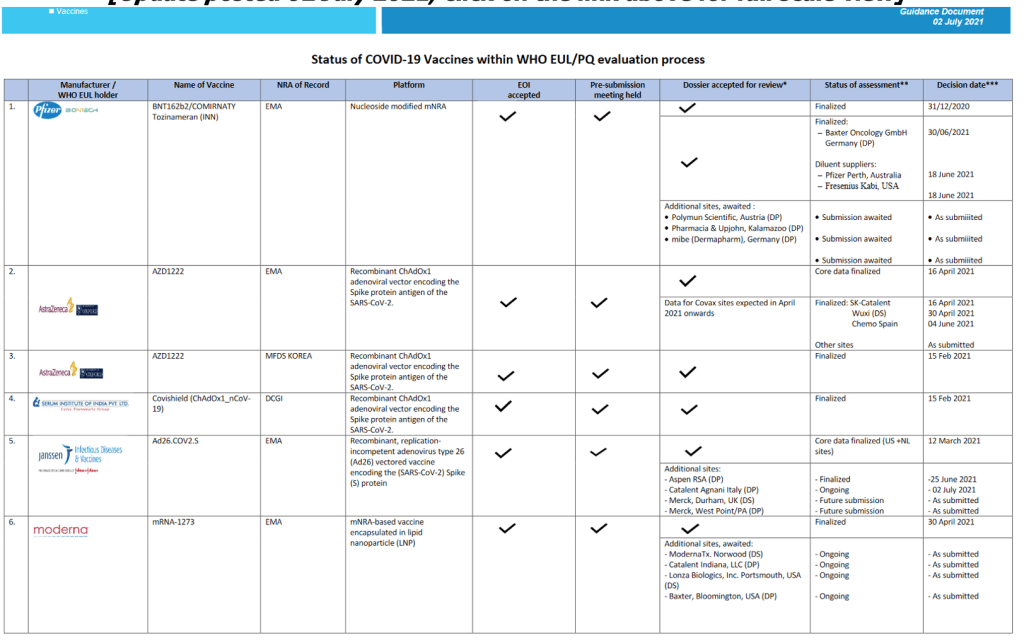
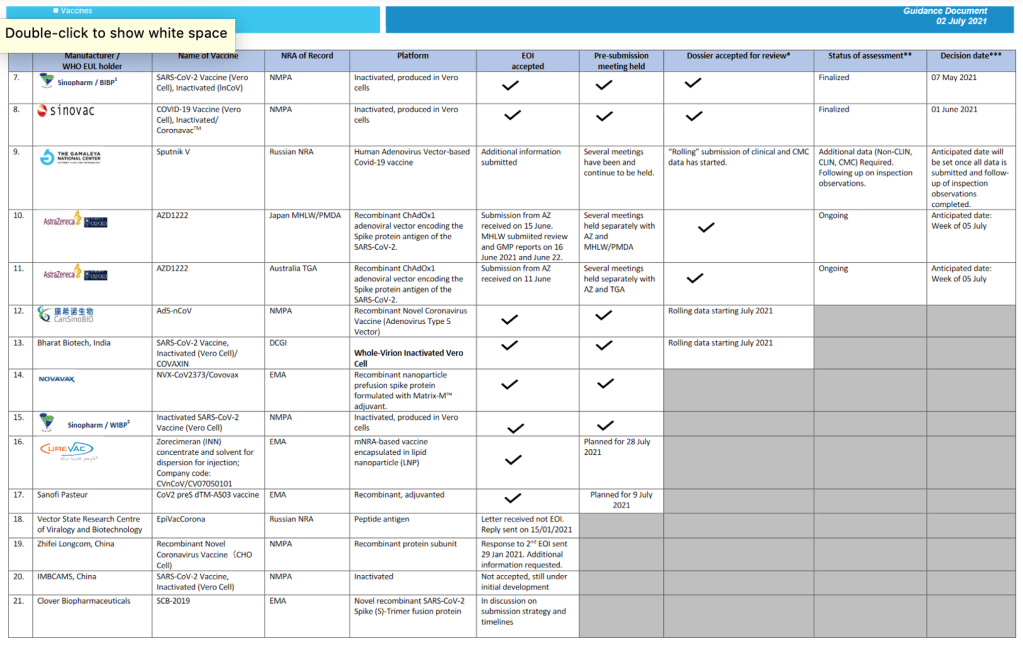
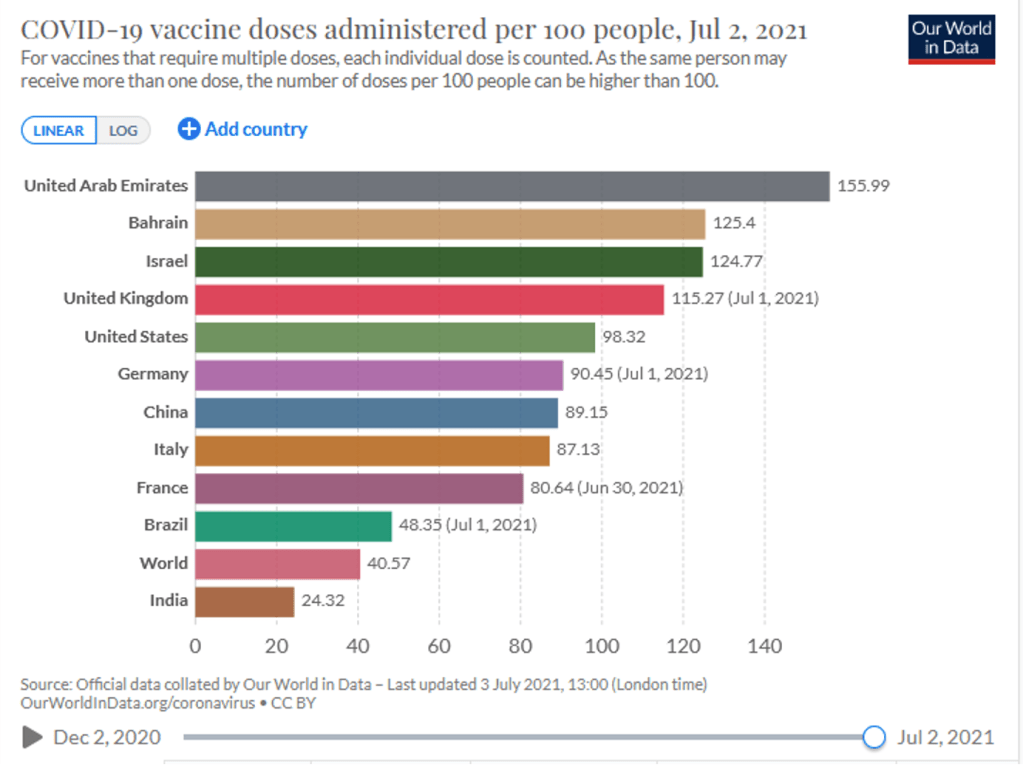
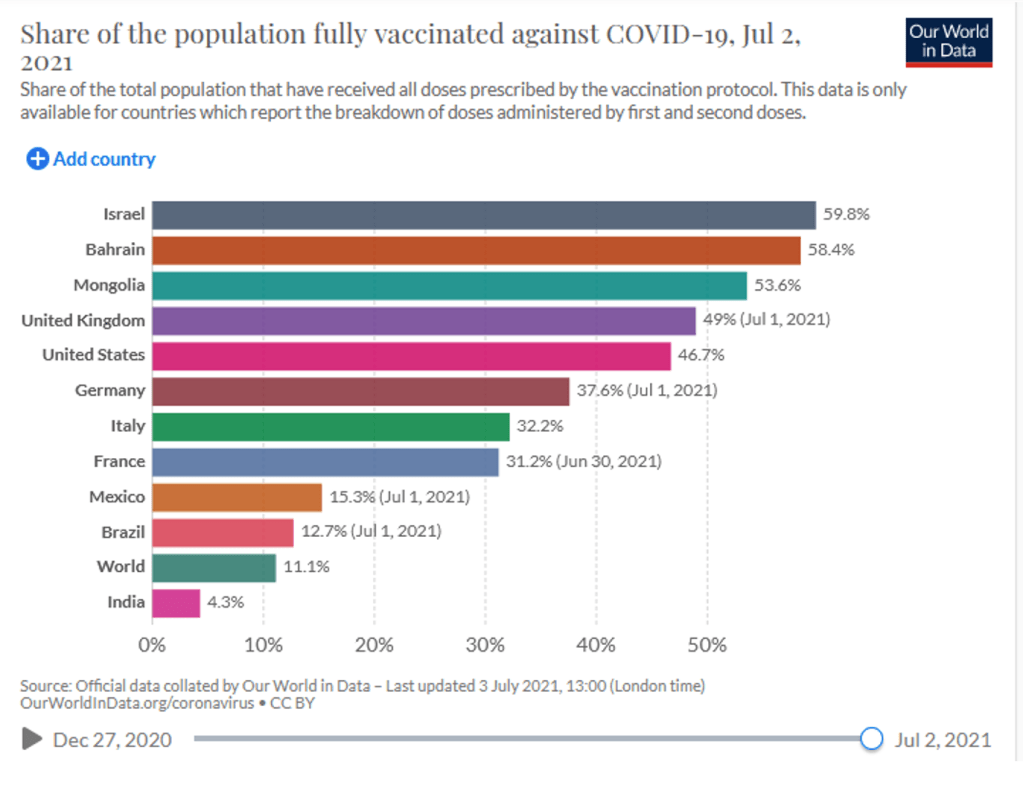
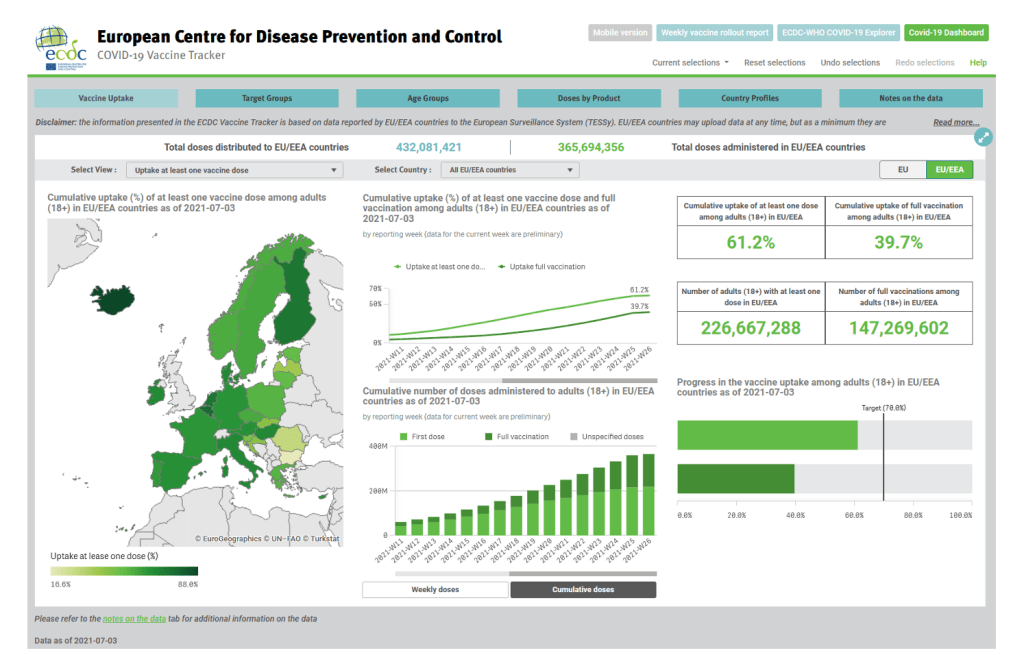
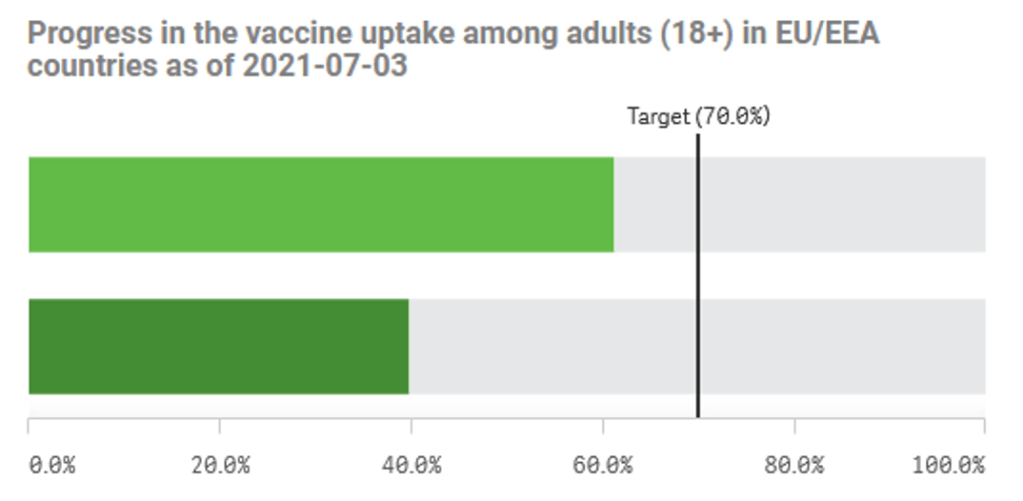
Organization Announcements
Editor’s Note:
Careful readers will note that the number and range of organizations now monitored in our Announcements section below has grown as the impacts of the pandemic have spread across global economies, supply chains and programmatic activity of multilateral agencies and INGOs.
Paul G. Allen Frontiers Group [to 3 Jul 2021]
https://alleninstitute.org/what-we-do/frontiers-group/news-press/
News
No new digest content identified.
BARDA – U.S. Department of HHS [to 3 Jul 2021]
https://www.phe.gov/about/barda/Pages/default.aspx
News
No new digest content identified.
BMGF – Gates Foundation [to 3 Jul 2021]
https://www.gatesfoundation.org/ideas/media-center
Press Releases and Statements
Press release
Jun 30, 2021
Gates Foundation Commits $2.1 Billion to Advance Gender Equality Globally
Commitments at the Generation Equality Forum will advance women’s economic empowerment, strengthen health and family planning, and accelerate women in leadership as women suffer disproportionately from the pandemic
Bill & Melinda Gates Medical Research Institute [to 3 Jul 2021]
https://www.gatesmri.org/
The Bill & Melinda Gates Medical Research Institute is a non-profit biotech organization. Our mission is to develop products to fight malaria, tuberculosis, and diarrheal diseases—three major causes of mortality, poverty, and inequality in developing countries. The world has unprecedented scientific tools at its disposal; now is the time to use them to save the lives of the world’s poorest people
No new digest content identified.
CARB-X [to 3 Jul 2021]
https://carb-x.org/
News
No new digest content identified.
Center for Vaccine Ethics and Policy – GE2P2 Global Foundation [to 3 Jul 2021]
https://centerforvaccineethicsandpolicy.net/
News/Analysis/Statements
:: Past weekly editions and posting of all segments of Vaccines and Global Health: The Week in Review are available here.
:: [NEW] Informed Consent: A Monthly Review – July 2021 is now posted here
CEPI – Coalition for Epidemic Preparedness Innovations [to 3 Jul 2021]
http://cepi.net/
Latest News
Joint COVAX Statement on the Equal Recognition of Vaccines
COVAX 01 Jul 2021
[See COVID above for detail]
From lab to potential jab: Clover’s COVID-19 vaccine story
CEPI’s early investment in Clover has been key to accelerating development and equitable access to this promising vaccine.
COVAX 01 Jul 2021
CIOMS – COUNCIL FOR INTERNATIONAL ORGANIZATIONS OF MEDICAL SCIENCES [to 3 Jul 2021]
https://cioms.ch/
News; Publications
CIOMS June 2021 Newsletter
The CIOMS June 2021 Newsletter is available here.
DARPA – Defense Advanced Research Projects Agency [to 3 Jul 2021
https://www.darpa.mil/news
News
No new digest content identified.
Duke Global Health Innovation Center [to 3 Jul 2021]
https://dukeghic.org/
No new digest content identified.
EDCTP [to 3 Jul 2021]
http://www.edctp.org/
The European & Developing Countries Clinical Trials Partnership (EDCTP) aims to accelerate the development of new or improved drugs, vaccines, microbicides and diagnostics against HIV/AIDS, tuberculosis and malaria as well as other poverty-related and neglected infectious diseases in sub-Saharan Africa, with a focus on phase II and III clinical trials
30 June 2021
Annual Report 2020 – focused delivery on promises
Emory Vaccine Center [to 3 Jul 2021]
http://www.vaccines.emory.edu/
Vaccine Center News
No new digest content identified.
European Vaccine Initiative [to 3 Jul 2021]
http://www.euvaccine.eu/
Latest News
No new digest content identified
FDA [to 3 Jul 2021]
https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/default.htm
Press Announcements /Selected Details
July 2, 2021 – Coronavirus (COVID-19) Update: July 2, 2021
Today, the FDA authorized the use, under the emergency use authorization (EUA) for the Janssen COVID-19 vaccine, of an additional batch of vaccine drug substance manufactured at the Emergent facility. To date a total of four batches of Janssen drug substance that were manufactured at the Emergent facility have been authorized…
June 29, 2021 – Coronavirus (COVID-19) Update: June 29, 2021
The FDA has sent to Congress its Report on Drug Shortages for Calendar Year 2020, which includes a section on the agency’s drug shortage efforts in response to COVID-19…
Fondation Merieux [to 3 Jul 2021]
http://www.fondation-merieux.org/
News, Events
No new digest content identified
Gavi [to 3 Jul 2021]
https://www.gavi.org/
News Releases
1 July 2021
6 million children benefitting from the first ever multi-antigen vaccination campaign
[Zimbabwe] Harare, 1 July 2021 – UNICEF, Gavi, the Vaccine Alliance and WHO have partnered with the Ministry of Health and Child Care to launch a new vaccine campaign introducing typhoid conjugate vaccine (TCV) into the routine immunisation schedule across the country.
The TCV campaign, the first of its kind in the region, was made possible through funding from Gavi and the multi-donor Health Development Fund (HDF) supported by the European Union, the United Kingdom, Sweden, Irish Aid and Gavi.
“Before the discovery of antibiotics, typhoid would kill as many as one in five people who contracted it,” said Thabani Maphosa, Managing Director of Country Programmes at Gavi. “The rise of extreme drug resistant typhoid risks bringing us back to levels of mortality not seen since the 19th century, posing a risk to all of us. That’s why typhoid conjugate vaccine is so important and why the government of Zimbabwe deserves praise for introducing this lifesaver into its routine immunisation programme.”
The campaign has integrated TCV with the inactivated polio vaccine (IPV) and human papillomavirus (HPV) vaccination. In addition, children aged 6 to 59 months received a vitamin A supplementation. After the initial campaign, TCV will be administered routinely to all children at 9 months of age to protect them from typhoid fever which has become endemic in Zimbabwe with outbreaks continuing to affect communities, particularly children…
1 July 2021
Joint COVAX Statement on the equal recognition of vaccines
[See COVID above for detail]
30 June 2021
Indonesia introduces Pneumococcal Conjugate Vaccine (PCV) across the country
:: Gavi, UNICEF and partners collaborate on nationwide introduction, which will see all children from the age of two months protected from pneumonia by 2022
:: The country has received 1.6 million doses of the vaccine procured through the Gavi Pneumococcal Advanced Market Commitment mechanism
:: Anuradha Gupta: “Congratulations to the Government of Indonesia on this milestone, which will save the lives of thousands of children across the country for years to come”
30 June 2021
Gavi signs agreement with Clover Biopharmaceuticals for supply to COVAX
:: The signed advance purchase agreement (APA) will make up to 414 million doses of the Clover vaccine candidate available to the COVAX Facility, pending WHO Emergency Use Listing (EUL)
:: Supply will be available to self-financing participants of the COVAX Facility as well as lower-income economies eligible to be supported by the Gavi COVAX Advance market Commitment (AMC), expected to commence Q4 2021
27 June 2021
US-donated vaccine deliveries through COVAX set to begin, with first shipment to Honduras
:: Deliveries of US-donated doses through COVAX to begin today
:: Anticipated delivery of the Moderna vaccine to Honduras comes after announcements by President Biden of significant dose-sharing pledges; further deliveries to follow in coming days and weeks
GHIT Fund [to 3 Jul 2021]
https://www.ghitfund.org/newsroom/press
GHIT was set up in 212 with the aim of developing new tools to tackle infectious diseases that
No new digest content identified.
Global Fund [to 3 Jul 2021]
https://www.theglobalfund.org/en/news/
News & Stories
News
WHO and Global Fund Sign Cooperation Agreement to Scale Up HIV, TB and Malaria Interventions and Strengthen Health Systems
30 June 2021
The World Health Organization and the Global Fund to Fight AIDS, Tuberculosis and Malaria signed a cooperation and financing agreement to implement 10 strategic initiatives to accelerate the end of AIDS, tuberculosis and malaria as epidemics and strengthen systems for health. This new agreement, which will cover the 2021-2023 implementation period, aims to address some of the persistent challenges that impede progress against the three diseases and protect hard-won gains from new pandemics like COVID-19.
In 2019, a total of 1.4 million people died from tuberculosis and an estimated 409,000 people died from malaria. In 2020, 690,000 people died from AIDS-related illnesses…
Global Research Collaboration for Infectious Disease Preparedness [GloPID-R] [to 3 Jul 2021]
https://www.glopid-r.org/news/
News
No new digest content identified.
Hilleman Laboratories [to 3 Jul 2021]
http://www.hillemanlabs.org/
Website reports “under maintenance” at inquiry
Human Vaccines Project [to 3 Jul 2021]
http://www.humanvaccinesproject.org/
News
News webpage not responding at inquiry
IAVI [to 3 Jul 2021]
https://www.iavi.org/newsroom
Latest News
No new digest content identified.
International Coalition of Medicines Regulatory Authorities [ICMRA]
http://www.icmra.info/drupal/en/news
Selected Statements, Press Releases, Research
No new digest content identified.
ICRC [to 3 Jul 2021]
https://www.icrc.org/en/whats-new
Selected News Releases, Statements, Reports
Sexual Violence, Conflict, and COVID-19: An Invisible Pandemic
Despite clear legal prohibitions, sexual violence remains widespread during armed conflicts and other situations of violence, as well as in detention. It occurs in various contexts and has grave humanitarian consequences.
24-06-2021 | Article
The challenges for humanitarian action in today’s conflicts: the perspective from the International Committee of the Red Cross
Excellencies, colleagues.It is a pleasure to address you today and I extend my warm appreciation to Minister of Defence Sergey Shoygu.
23-06-2021 | Statement
International Generic and Biosimilar Medicines Association [IGBA]
https://www.igbamedicines.org/
News
No new digest content identified.
IFFIm
http://www.iffim.org/
Press Releases/Announcements
No new digest content identified.
IFRC [to 3 Jul 2021]
http://media.ifrc.org/ifrc/news/press-releases/
Selected Press Releases, Announcements
Africa
As COVID-19 cases surge in Africa, Red Cross warns that insufficient funding is impeding the response
Nairobi/Geneva, 2 July 2021 – Halting an increasing trend of COVID-19 cases in Africa will require additional funding. This was announced by the International Federation of Red Cross and Red Crescent Societies (IFRC), amid a worrying surge of cases in …
2 July 2021
Blind spots continue to prevent access to COVID-19 vaccines for refugees and migrants, new Red Cross and Red Crescent report says
Geneva, 30 June 2021 – New research conducted by the Red Cross Red Crescent Global Migration Lab across more than 50 countries reveals that refugees and migrants continue to face serious obstacles in accessing COVID-19 vaccines. Despite some progress m …
30 June 2021
Indonesia
Indonesia: On edge of catastrophe as COVID-19 overwhelms hospitals
Kuala Lumpur/Jakarta/Geneva, 29 June 2021: Urgent increases are needed in medical care, testing and vaccinations as Indonesia teeters on the edge of a COVID-19 catastrophe. The more dangerous and deadly Delta COVID-19 variant is fuelling infections tha …
29 June 2021
Institut Pasteur [to 3 Jul 2021]
https://www.pasteur.fr/en/press-area
Press Documents
Press Info
01.07.2021
Modelling study of benefits and risks associated with the COVID-19 vaccine Vaxzevria
On April 7, 2021, the European Medical Agency (EMA) concluded that thrombosis in combination with thrombocytopenia has…
Press release
29.06.2021
Neonatal meningitis: the immaturity of microbiota and epithelial barriers implicated
Meningitis is associated with high mortality and frequently causes severe sequelae. Newborn infants are particularly…
IOM / International Organization for Migration [to 3 Jul 2021]
http://www.iom.int/press-room/press-releases
News
IOM’s Emergency Director Urges Swift, Durable Solutions to Protracted Rohingya Crisis
2021-06-29 16:08
Cox’s Bazar – Almost 900,000 Rohingya refugees in congested camps in Bangladesh’s Cox’s Bazar District desperately need urgent action to secure their future, nearly four years after they were forced to flee Myanmar, a senior International Organization for…
IVAC [to 3 Jul 2021]
https://www.jhsph.edu/research/centers-and-institutes/ivac/index.html
Updates; Events
Johns Hopkins Launches ‘Vira’ Vax ChatBot
July 2021
COVID-19 vaccination is lagging in young adults nationally. Vira, the Vax Chatbot is designed to support and meet young people where they are with simple answers to common COVID-19 vaccine questions. Vira is adaptive, learning by listening to your questions and feedback to help scientists respond to emerging questions. […]
IVI [to 3 Jul 2021]
http://www.ivi.int/
Selected IVI News, Announcements, Events
No new digest content identified.
JEE Alliance [to 3 Jul 2021]
https://www.jeealliance.org/
Selected News and Events
No new digest content identified.
Johns Hopkins Center for Health Security [to 3 Jul 2021]
https://www.centerforhealthsecurity.org/news/center-news/
Center News
Johns Hopkins Center for Health Security Shares Recommendations to Improve Nation’s Public Health and Medical Preparedness and Response Programs
June 28, 2021 – As the U.S. Congress continues to respond to and begin recovery from the COVID-19 pandemic, they must also prepare for the next pandemic, which could appear at any time. For over 20 years, the Johns Hopkins Center for Health Security has worked to protect people’s health from epidemics and disasters and ensure community resilience by examining how innovations, policies, and programs can strengthen health security.
The Center has identified 13 proposals that, if empowered through bipartisan legislation, would improve the nation’s public health, and increase medical preparedness and response programs for future public health events.
Proposals:
Expedite Development of Medical Countermeasures for Unknown Viral Threats
Invest in Ongoing and Just-in-Time Social Science Research to Ensure Public Support for Outbreak Management
Authorize the National Center for Epidemic Forecasting and Outbreak Analytics
Prioritize Funding for Development of At-Home Diagnostic Technology
Support Innovation and Stockpile Strategies to Provide Better Respiratory Protective Devices
Create a National Center for Pandemic and Disaster Nursing Research
Develop New Vaccine Delivery Platforms to Rapidly Immunize Millions in a Pandemic
Develop a National Strategy to Combat Health-Related Misinformation and Disinformation
Create Deployable Research Response Teams
Create a Central Repository for Serosurveys and Public Health Observational Studies
Evaluate the Hospital Preparedness Program’s Effectiveness during the Pandemic
Enlist the Community Health Sector in Achieving Adequate Preparedness and Response for Vulnerable Populations
Develop a Coherent Strategy and Substructure to Facilitate Comprehensive Recovery from an Infectious Disease Disaster
Read full summaries of the proposals.
MSF/Médecins Sans Frontières [to 3 Jul 2021]
http://www.msf.org/
Latest [Selected Announcements]
Coronavirus COVID-19 pandemic
COVID-19 pandemic threatens to deteriorate in Brazil as 500,000 deaths recorded
Project Update 30 Jun 2021
As the weather gets colder in Brazil and the pandemic rolls on, the COVID-19 situation in the country has again become deeply concerning, with national and international experts warning of a third devastating wave.
The response remains fragmented and decentralised while the central authorities continue to disregard science, including the importance of masks and physical distancing, in their health messaging.
Sadly, Brazilians now account for over 500,000 COVID-19 deaths and the average COVID-19 fatality rate has crept above 2,000 people a day for the first time since May. Likewise, the number of new cases has also risen to more than 70,000 a day. This is the highest average since the peak of cases during the second wave in early May…
National Academy of Medicine – USA [to 3 Jul 2021]
https://nam.edu/programs/
Selected News/Programs
No new digest content identified.
National Vaccine Program Office – U.S. HHS [to 3 Jul 2021]
https://www.hhs.gov/vaccines/about/index.html
Upcoming Meetings/Latest Updates
No new digest content identified.
NIH [to 3 Jul 2021]
http://www.nih.gov/news-events/news-releases
News Releases
NIH COVID-19 testing initiative funds additional research projects to safely return children to in-person school
July 2, 2021 — The awards are the second installment of the Safe Return to School Diagnostic Testing Initiative.
Investigational malaria vaccine gives strong, lasting protection
June 30, 2021 — Phase 1 trials conducted at NIH Clinical Center.
Two U.S. Phase 1 clinical trials of a novel candidate malaria vaccine have found that the regimen conferred unprecedentedly high levels of durable protection when volunteers were later exposed to disease-causing malaria parasites. The vaccine combines live parasites with either of two widely used antimalarial drugs—an approach termed chemoprophylaxis vaccination. A Phase 2 clinical trial of the vaccine is now underway in Mali, a malaria-endemic country. If the approach proves successful there, chemoprophylaxis vaccination, or CVac, potentially could help reverse the stalled decline of global malaria. Currently, there is no vaccine in widespread use for the mosquito-transmitted disease.
The trials were conducted at the National Institutes of Health (NIH) Clinical Center in Bethesda, Maryland. They were led by Patrick E. Duffy, M.D., of the NIH National Institute of Allergy and Infectious Diseases (NIAID), and Stephen L. Hoffman, M.D., CEO of Sanaria Inc., Rockville, Maryland.
The Sanaria vaccine, called PfSPZ, is composed of sporozoites, the form of the malaria parasite transmitted to people by mosquito bites. Sporozoites travel through blood to the liver to initiate infection…
NIH-funded screening study builds case for frequent COVID-19 antigen testing
June 30, 2021 — Rapid antigen tests perform on par with lab tests when used every three days.
Adjuvant developed with NIH funding enhances efficacy of India’s COVID-19 vaccine
June 29, 2021 — Adjuvants are formulated as part of a vaccine to boost immune responses and enhance effectiveness.
PATH [to 3 Jul 2021]
https://www.path.org/media-center/
Press Releases
PATH announces new five-year strategy
June 29, 2021—PATH announced the launch of a new strategy that will guide the organization’s efforts over the next five years.
The result of a year-long development process that included input from more than 400 local stakeholders across 50 countries, PATH Strategy 2025 aims to respond to current global crises, to the needs and priorities of PATH’s local partners, and to their resounding call for more equitable and inclusive approaches to global public health.
“Our new strategy is very exciting, not only for PATH, but for our entire sector. Over the next five years, we will transform PATH into an even better agent of change for global health,” said Nikolaj Gilbert, President and CEO of PATH.
PATH Strategy 2025 outlines three main priorities: preparing for and responding to emerging health threats, improving lives with science and technology, and increasing health system capacity and resilience.
The five-year plan also outlines four change strategies—equity in health, community-focused priorities, respectful partnerships, and inclusive innovation—that will shape how PATH approaches its work. To learn more about PATH Strategy 2025, visit path.org/strategy2025.
PATH welcomes introduction of the US International Pandemic Preparedness and COVID-19 Response Act of 2021
June 29, 2021 by PATH
Sabin Vaccine Institute [to 3 Jul 2021]
http://www.sabin.org/updates/pressreleases
Statements and Press Releases
No new digest content identified.
UNAIDS [to 3 Jul 2021]
http://www.unaids.org/en
Selected Press Releases/Reports/Statements
1 July 2021
Report shows big COVID-19-related HIV prevention programme service disruptions, but highlights that HIV service innovations and adaptations are possible
30 June 2021
UNAIDS, IOM: People on the move living with HIV must have access to COVID-19 vaccines
[See COVID above for detail]
28 June 2021
The journey towards comprehensive sexuality education
UNHCR Office of the United Nations High Commissioner for Refugees [to 3 Jul 2021]
http://www.unhcr.org/en-us/media-centre.htmlS
Selected News Releases, Announcements
Refugee health challenges remain high amid COVID-19
1 Jul 2021
Malaria remained the single most common cause of illness among refugees in 2020, while psychological distress caused by COVID-19, and acute malnutrition constituted major threats to refugees’ health and well-being, according to data released today by UNHCR, the UN Refugee Agency, in its Annual Public Health Global Review.
In a year marked by the pandemic, UNHCR’s key focus was to advocate for the inclusion of refugees in COVID-19 national response plans. The agency also worked to support national health systems by procuring personal protective equipment, other equipment like oxygen concentrators, COVID-19 tests and medicines, and by increasing intensive care capacity in countries such as Lebanon and Bangladesh…
United Nations Human Rights Council [to 3 Jul 2021]
https://www.ohchr.org/EN/HRBodies/HRC/Pages/NewsSearch.aspx?NTID=PRS&MID=HR_COUNCIL
Council News
COVID, State surveillance, smartphones, mega tech-corporations and artificial intelligence are only a few of the themes which will remain recurrent throughout the life of the mandate on privacy
02/07/2021
Access to reliable information sources is the obvious antidote to disinformation, Special Rapporteur on freedom of opinion and expression tells Human Rights Council
02/07/2021
COVID-19 Pandemic Brought on a Substantive Setback for Persons with Leprosy who must be Recognised as a Vulnerable Group with Regard to Vaccination, Special Rapporteur Tells Human Rights Council
01/07/2021
UNICEF [to 3 Jul 2021]
https://www.unicef.org/media/press-releases
Press Releases, News Notes, Statements [Selected]
News note 07/01/2021
UNICEF signs COVID-19 vaccine supply agreement with Janssen Pharmaceutica NV
COPENHAGEN/NEW YORK, 1 July 2021 – UNICEF has signed a long-term agreement with Janssen Pharmaceutica NV for the supply of COVID-19 vaccine on behalf of the COVAX Facility.
Through the supply agreement, UNICEF, along with procurement partners including the Pan-American Health Organization (PAHO), will have access to up to 200 million doses of the single-dose vaccine in 2021 for 92 countries supported by the Advance Market Commitment and also some self-financing countries. This is the 6th such agreement that UNICEF has completed for COVID-19 vaccines, previous agreements have been announced with the Serum Institute of India, Pfizer, AstraZeneca, Human Vaccine and Moderna…
Press release 07/01/2021
Billions of people will lack access to safe water, sanitation and hygiene in 2030 unless progress quadruples – warn WHO, UNICEF
Latest estimates reveal that 3 in 10 people worldwide could not wash their hands with soap and water at home during the COVID-19 pandemic
Statement 07/01/2021
Joint COVAX statement on the equal recognition of vaccines
[See COVID above for detail]
Press release 06/30/2021
Indonesia introduces pneumococcal conjugate vaccine (PCV) across the country
:: Gavi, UNICEF and partners collaborate on nationwide introduction, which will see all children from the age of two months protected from pneumonia by 2022
:: The country has received 1.6 million doses of the vaccine procured through the Gavi Pneumococcal Advanced Market Commitment mechanism
:: Anuradha Gupta: “Congratulations to the Government of Indonesia on this milestone, which will save the lives of thousands of children across the country for years to come”
Statement 06/27/2021
Statement on U.S. donation of 1.5 million doses of COVID-19 vaccine to Honduras
Statement by Jean Gough, UNICEF Regional Director for Latin America and the Caribbean
PANAMA CITY, 27 June 2021 – “UNICEF welcomes the first United States Government donation of COVID-19 vaccines via COVAX’s dose-sharing mechanism, arriving today in Honduras, and calls for more largescale vaccine donations, in particular to hard-hit Latin America and the Caribbean.
“With only less than 62,000 Hondurans reportedly fully vaccinated, these 1.5 million doses of vaccine represent long-awaited relief to Honduras, which has one of the lowest vaccination rates in Latin America. Moreover, these doses are a glimmer of hope to the region and all countries waiting anxiously for COVAX doses. The United States should be applauded for stepping up to support Honduras, the region, and the world…
Unitaid [to 3 Jul 2021]
https://unitaid.org/
Featured News
No new digest content identified.
Vaccine Equity Initiative [to 3 Jul 2021]
https://vaccineequitycooperative.org/news/
News
No new digest content identified.
Vaccination Acceptance & Demand Initiative [Sabin) [to 3 Jul 2021]
https://www.vaccineacceptance.org/
Announcements
No new digest content identified.
Vaccine Confidence Project [to 3 Jul 2021]
http://www.vaccineconfidence.org/
News, Research and Reports
Coronavirus global impact
Launched April 2, 2020 and recurring every 3 days, Premise Data is utilizing its global network of Contributors to assess economic, social, and health sentiment surrounding the coronavirus (COVID-19).
Vaccine Education Center – Children’s Hospital of Philadelphia [to 3 Jul 2021]
http://www.chop.edu/centers-programs/vaccine-education-center
News
No new digest content identified.
Wellcome Trust [to 3 Jul 2021]
https://wellcome.ac.uk/news
News and reports
News
From equality to global poverty: the Covid-19 effects on societies and economies
29 June 2021
The Covid-19 pandemic is a social and an economic crisis just as much as it is a health one – its repercussions, severe and far-reaching, are being felt across the world.
The Wistar Institute [to 3 Jul 2021]
https://www.wistar.org/news/press-releases
Press Releases
Jun. 29, 2021
Wistar Scientists Discover Blood-based Biomarkers to Predict HIV Remission After Stopping Antiretroviral Therapy
The newly identified sugar and metabolic signatures may help predict duration and probability of HIV remission after antiretroviral therapy interruption during cure-directed clinical trials.
WFPHA: World Federation of Public Health Associations [to 3 Jul 2021]
https://www.wfpha.org/
Latest News
Recommendations to Drive Value-based Decision-making in Vaccination
Jun 30, 2021
The Value(S) of Vaccination: Building the Scientific Evidence according to a Value-Based Healthcare Approach
Jun 29, 2021
World Bank [to 3 Jul 2021]
http://www.worldbank.org/en/news/all
Selected News, Announcements
Over 350,000 People to Obtain COVID-19 Vaccination in Liberia
WASHINGTON, June 30, 2021 – The World Bank today approved Additional Financing to the Liberia COVID-19 Emergency Preparedness and Response Project to enable affordable and equitable access to COVID-19…
Date: June 30, 2021 Type: Press Release
World Bank Financing for COVID-19 Vaccine Rollout Exceeds $4 Billion for 50 Countries
WASHINGTON, June 30, 2021 – The World Bank announced today that it is providing over $4 billion for the purchase and deployment of COVID-19 vaccines for 51 developing countries, half of which are…
Date: June 30, 2021 Type: Press Release
[See COVID above for detail]
Benin: World Bank Support for the COVID-19 Vaccination Rollout
WASHINGTON, June 29, 2021 – The World Bank has approved $30 million in additional financing from the International Development Association (IDA)* to help Benin vaccinate its population against COVID-19…
Date: June 29, 2021 Type: Press Release
Sudan Gets a Boost from World Bank Group for COVID-19 Response
KHARTOUM, June 29, 2021—The Government of Sudan and the World Bank signed a grant agreement of $100 million to boost the country’s COVID-19 response and expand vaccination coverage in the country. This…
Date: June 29, 2021 Type: Press Release
World Bank Approves $24 Million to Support Zambia with COVID-19 Vaccines
WASHINGTON, June 29, 2021 – The World Bank approved $24 million in additional financing to the Republic of Zambia for the acquisition and equitable distribution of COVID-19 (coronavirus) vaccines. The…
Date: June 29, 2021 Type: Press Release
World Customs Organization – WCO [to 3 Jul 2021]
http://www.wcoomd.org/
Latest News – Selected Items
30 June 2021
WCO Deputy Secretary General addresses the WTO Technical Symposium on COVID-19 vaccine supply chain and regulatory transparency
The Deputy Secretary General of the World Customs Organization (WCO), Mr. Ricardo Treviño, joined the Deputy Director-General of the World Trade Organization (WTO), Ms. Anabel González and the Assistant Director-General of the World Health Organization (WHO), Dr. Mariângela Simão in the opening of the WTO Technical Symposium on COVID-19 vaccine supply chain and regulatory transparency that was held online on 29 June 2021.
The Technical Symposium was organized as a follow-up to the WTO event on COVID-19 and vaccine equity that was held on 14 April 2021 with the participation of the WCO Secretary General.
The 29 June 2021 follow-up event was aimed at contributing to improved public and policymaker understanding of the operation of global COVID-19 vaccine supply chains, the cross-border movement of vaccine inputs, and the need for greater regulatory transparency and convergence. The Technical Symposium was also aimed at mapping vaccine production and trade and discussing trade measures that support or constrain the achievement of the rapid scale-up of vaccine production and distribution…
World Organisation for Animal Health (OIE) [to 3 Jul 2021]
https://www.oie.int/en/for-the-media/press-releases/2021/
Press Releases
Ten years on, Rinderpest eradication highlights the value of vaccines
News 28 June 2021
The global eradication of Rinderpest gives reason to be optimistic when it comes to fighting infectious animal diseases. While the recent COVID-19 pandemic has shown the devastating impact emerging infectious diseases can have, it is also a reminder of the critical role vaccines can play in protecting us all.
This text was originally published on the iD4D website under Creative Commons BY NC ND 4.0 International
WTO – World Trade Organisation [to 3 Jul 2021]
http://www.wto.org/english/news_e/news_e.htm
WTO News and Events
Improving trade data for COVID-19 products key to better response policies: WTO
1 July 2021
Improving trade data on products needed to combat the COVID-19 pandemic — including vaccines and their components — is key to ensuring that the right policies are in place to facilitate their distribution, according to a new information note issued by the WTO Secretariat on 1 July.
WTO issues new update on trade in medical goods in the context of COVID-19
30 June 2021
The WTO Secretariat has published a further update of its information note on trade in medical goods in the context of tackling COVID-19. The update provides figures on trade in medical goods covering all of 2020.
World Bank, IMF, WHO, WTO heads call for urgent action to accelerate global vaccine access
30 June 2021
The heads of the World Bank Group, International Monetary Fund, World Health Organization and the WTO convened on 30 June the first meeting of the Task Force on COVID-19 Vaccines, Therapeutics and Diagnostics for Developing Countries. David Malpass, Kristalina Georgieva, Dr Tedros Adhanom Ghebreyesus and Dr Ngozi Okonjo-Iweala said urgent action is needed to stop the rising human toll due to the pandemic and halt further divergence in the economic recovery between advanced economies and the rest. Following the meeting, they issued a joint statement.
[See COVID above for detail]
WTO technical symposium feeds into continuing efforts to deal with COVID-19 pandemic
29 June 2021
The WTO held a technical symposium on 29 June 2021 to address the main challenges to vaccine supply chain and regulatory transparency in the context of COVID-19. Representatives of the medical and pharmaceutical industry, international organizations and the WTO Secretariat explored new ways to cooperate towards finding practical solutions to scale up the global COVID-19 response and address gaps in the global production and distribution of vaccines, personal protective equipment and other medical technologies.
[See COVID above for detail]
::::::
ARM [Alliance for Regenerative Medicine] [to 3 Jul 2021]
Press Releases – Alliance for Regenerative Medicine (alliancerm.org)
Selected Press Releases
ARM and NIIMBL Release Project A-Gene to Bring Quality by Design Principles to Gene Therapy Manufacturing
June 24, 2021, Washington, DC
The Alliance for Regenerative Medicine (ARM) and the National Institute for Innovation in Manufacturing Biopharmaceuticals (NIIMBL) today released Project A-Gene, a multistakeholder collaboration to incorporate Quality by Design (QbD) principles into a manufacturing case study of a viral vector commonly used in gene therapies…
BIO [to 3 Jul 2021]
https://www.bio.org/press-releases
Press Releases, Lewtters, Testimony, Comments [Selected]
BIO Letter to USTR Ambassador Tai on Global Vaccine Distribution
June 30, 2021
We would like to provide you some updates on the U.S. and global biotechnology industry’s efforts to scale-up global vaccine production to address the global imbalance in access to COVID vaccines, and to request a close working relationship with…
BIO letter to HHS Secretary Xavier Becerra on the importance of funding R&D for COVID-19 therapeutics at Biomedical Advanced Research and Development Agency (BARDA).
June 25, 2021
The partnership between the biopharmaceutical industry and the U.S. Government is largely responsible for where we are with the COVID-19 pandemic today. In just 16 months from the declaration of the pandemic, cases have dramatically dropped as more…
A Declaration from Members of the World’s Biotechnology Sector On Global Access to COVID Vaccines & Treatments and the Role of Intellectual Property
June 24, 2021
[See COVID above for detail]
DCVMN – Developing Country Vaccine Manufacturers Network [to 3 Jul 2021]
http://www.dcvmn.org/
News; Upcoming events
No new digest content identified.
ICBA – International Council of Biotechnology Associations [to 3 Jul 2021]
https://internationalbiotech.org/news/
News
A Declaration from Members of the World’s Biotechnology Sector On Global Access to COVID Vaccines & Treatments and the Role of Intellectual Property
Jul. 01 2021
[See COVID above for detail]
IFPMA [to 3 Jul 2021]
http://www.ifpma.org/resources/news-releases/
Selected Press Releases, Statements, Publications
No new digest content identified.
International Alliance of Patients’ Organizations – IAPO [to 3 Jul 2021]
https://www.iapo.org.uk/news/topic/6
Press and media [Selected]
No new digest content identified.
PhRMA [to 3 Jul 2021]
http://www.phrma.org/
Latest News [Selected]
3 things that support robust biopharmaceutical research and development
June 29, 2021
Let’s take a closer look at three key pillars of the biopharmaceutical ecosystem that are central to the success of America’s research and development:
1. Private-public collaboration
2. A robust intellectual property (IP) system
3. Strong private sector investment in R&D