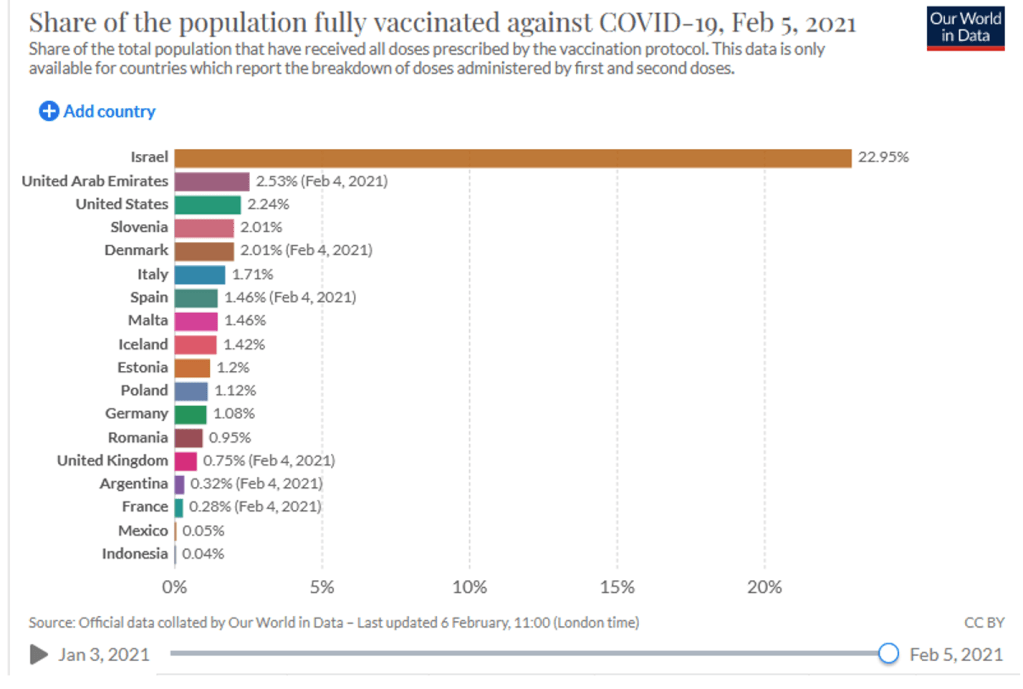
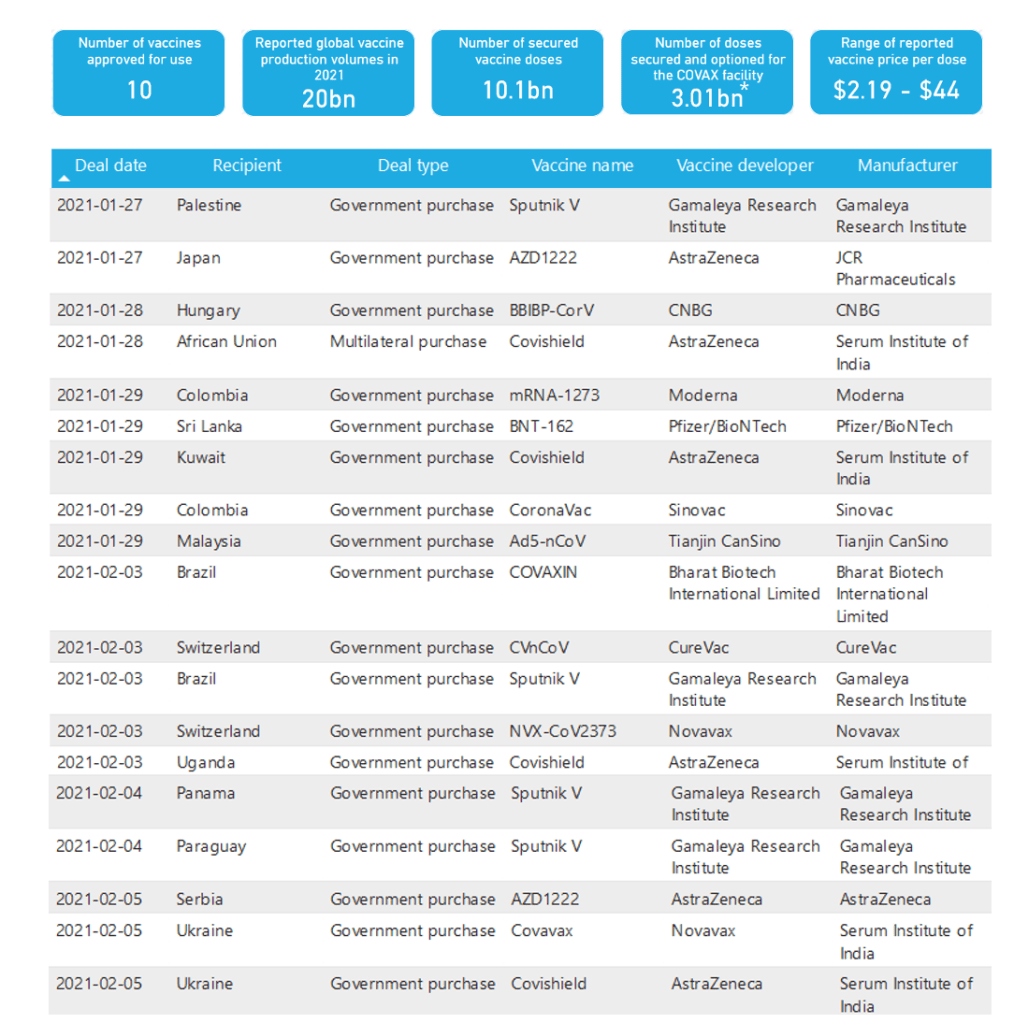
Media/Policy Watch
This watch section is intended to alert readers to substantive news, analysis and opinion from the general media and selected think tanks and similar organizations on vaccines, immunization, global public health and related themes. Media Watch is not intended to be exhaustive, but indicative of themes and issues CVEP is actively tracking. This section will grow from an initial base of newspapers, magazines and blog sources, and is segregated from Journal Watch above which scans the peer-reviewed journal ecology.
We acknowledge the Western/Northern bias in this initial selection of titles and invite suggestions for expanded coverage. We are conservative in our outlook in adding news sources which largely report on primary content we are already covering above. Many electronic media sources have tiered, fee-based subscription models for access. We will provide full-text where content is published without restriction, but most publications require registration and some subscription level.
The Atlantic
http://www.theatlantic.com/magazine/
Accessed 13 Feb 2021
Ideas
How to Beat the Pandemic by Summer
Averting a wave of new COVID-19 fatalities could require some dramatic, untested, and controversial strategies.
13 Feb 2021
Derek Thompson Staff writer at The Atlantic
After nearly a year of social isolation and sacrifice in the long war on COVID-19, the end stage of the pandemic is finally in sight. Millions of Americans are being vaccinated each week, and the number of coronavirus-related hospitalizations in the United States has plunged by more than 40 percent in the past month.
But this final stage will still be lethal—perhaps more so than most people imagine. More Americans were reported dead of COVID-19 on Friday, February 5, than on any day in all of 2020. The U.S. is still on pace to have more than 80,000 COVID-19 fatalities a month. Meanwhile, variants of the coronavirus that emerged in the United Kingdom, Brazil, and South Africa are spreading quickly. These variants are more contagious and more deadly than the original virus, and they threaten to stall or even reverse our progress.
“If we don’t accelerate the pace of vaccinations, we’re looking at an apocalypse,” says Peter Hotez, a vaccine scientist at Baylor College of Medicine. “We’ve got to figure out a way to get ahead of the variants to avoid 1 million deaths by the end of this year.”…
Health
What If We Never Reach Herd Immunity?
Hitting the threshold might actually be impossible. But vaccines can still help end the pandemic.
Sarah Zhang, February 9, 2021
BBC
http://www.bbc.co.uk/
Accessed 13 Feb 2021
HARDtalk
Kirill Dmitriev: Russia’s Sputnik V a vaccine for humankind?
Kirill Dmitriev, head of a ten billion dollar sovereign wealth fund which backed Sputnik V, on Russia’s willingness to share its vaccine in the global fight against Covid19
15 February 2021 :: Audio – 23 minutes
Covid: Oxford-AstraZeneca vaccine to be tested on children
13 Feb 2021
A new trial is to test how well the Oxford-AstraZeneca coronavirus vaccine works in children.
Some 300 volunteers will take part, with the first vaccinations in the trial taking place later in February.
Researchers will assess whether the jab produces a strong immune response in children aged between six and 17.
The vaccine is one of two being used to protect against serious illness and death from Covid in the UK, along with the Pfizer-BioNTech jab.
As many as 240 children will receive the vaccine – and the others a control meningitis jab – when the trial gets under way…
The Economist
http://www.economist.com/
Accessed 13 Feb 2021
Feb 13th 2021
How well will vaccines work?
Covid-19 may become endemic. Governments need to start thinking about how to cope
EVEN MIRACLES have their limits. Vaccines against the coronavirus have arrived sooner and worked better than many people dared hope. Without them, the pandemic threatened to take more than 150m lives. And yet, while the world rolls up a sleeve, it has become clear that expecting vaccines to see off covid-19 is mistaken. Instead the disease will circulate for years, and seems likely to become endemic.
Fear, uncertainty and doubt
Vaccine hesitancy is putting progress against covid-19 at risk
If the world is to tame the virus, the doubts will need to be fought
Vaccine efficacy
When covid-19 vaccines meet the new variants of the virus
A lot depends on blocking transmission not just disease
Financial Times
https://www.ft.com/
Accessed 13 Feb 2021
Covid-19 vaccines
Can Covax deliver the vaccines much of the world needs?
February 12, 2021
Coronavirus Business Update
Developing world struggles to get its share of Covid vaccines
February 12, 2021
Why the world needs a Covid-19 exit strategy
The public needs to know when, how and how quickly restrictions will be lifted
The editorial board
February 12, 2021
February is one of the busiest months of the year for travel bookings in the northern hemisphere. Families plan how they will spend their summer holidays and try to grab a bargain spot in the sun. Not this year, however. The uncertainty over how long coronavirus restrictions will remain in place means that while some have taken a risk and booked a break, most are sitting on the sidelines, unsure of what to do. This confusion is emblematic of the wider uncertainties that exist over the reopening of economies. Tired of severe curbs of civil liberties — which have lasted far longer than most anticipated — many people are asking when governments will finally set out a path for life to return to something like normal, how it will happen, and how fast.
This anxiety has only been heightened in recent weeks by signs that in some cases restrictions, instead of easing as lockdowns take effect, are in fact being toughened to guard against more infectious and possibly more deadly new mutations of the virus. In the UK, tough border controls will take effect from Monday for people arriving in the country, including jail terms for those offending. Germany is poised to reinstate border controls with some areas of Austria and the Czech Republic, while in Australia the state of Victoria has announced a snap five-day lockdown after an outbreak.
A global, well-funded vaccination campaign is the only way to end the pandemic for good. But despite relatively high inoculation rates in some countries, it is now clear that vaccinating enough people to achieve herd immunity will take much longer than hoped. Covax, the global initiative to distribute vaccines equitably, aims to deliver at least 2bn doses by the end of this year but that will not cover much more than a third of its target population. The new variants mean that some of the measures we have become accustomed to during lockdown will have to stay in place for some time. While some curbs on freedom will be accepted, others — bans on foreign travel and on seeing family and friends — cannot be tolerated indefinitely. A point will come where the public will rebel against restrictions that make life unbearably bleak. In the UK, scientific advisers are calling for a debate on the terms of allowing a “big wave of infection”.
The solution is for politicians to provide as much certainty as is possible. They must make it clear what will trigger the lifting of restrictions and what will not be possible in the short-to-medium term. In the UK, the government has so far produced little evidence of the costs and benefits of current restrictions. Priorities must be set, supported by appropriate economic and epidemiological modelling. Concern is rife over the impact of the lockdowns on young people and the loss of education. Reopening schools must therefore be a priority. Teachers will need to be vaccinated. Fast testing and accurate and quick contact tracing is critical. How to reboot travel must also be a consideration. For this to happen, vaccination passports would be essential, but with the aim of facilitating travel, not prohibiting travel. A recent policy document proposes that areas in Europe where the virus has been eliminated are declared green zones in which civil liberties are restored. These zones expand as more regions achieve elimination. Such an approach, while appealing, will require careful, global co-ordination.
The pandemic has severely damaged economies and societies. As far as is scientifically possible, the public deserves to know the route out of current restrictions. It is time for politicians to play their part and move from crisis response to forward planning.
Opinion Coronavirus pandemic
Another pandemic need never happen
Today’s technology is such that scientists can credibly make this claim for the first time in history
Richard Hatchett
The writer is chief executive of the Coalition for Epidemic Preparedness Innovations
… Humankind cannot prevent viruses from jumping the species barrier and on to humans. But with the right research and development investments, and a deliberate focus on eliminating barriers to rapid vaccine development, I believe we can eliminate the risk of a pandemic with the impact of Covid-19 from occurring again. It might cost tens of billions of dollars. But as a global insurance policy, that has got to be a bargain.
The Big Read Covid-19 vaccines
Vaccines vs variants: the race to immunise the developing world
The spread of new Covid strains has made it even more urgent to launch rapid vaccination programmes in poorer countries
David Pilling in London, Stephanie Findlay in New Delhi and Bryan Harris in São Paulo
February 11 2021
Opinion Coronavirus treatment
The west should pay attention to Russia and China’s vaccine diplomacy
Beijing and Moscow are using jabs to court poorer nations — but the EU and US are barely noticing Anne-Sylvaine Chassan
February 10 2021
Coronavirus pandemic
WHO dismisses coronavirus lab leak theory as ‘extremely unlikely’
Scientists visiting Chinese city of Wuhan conclude that bats were most plausible source of virus
Christian Shepherd in Beijing
February 9 2021
Top of Form
Bottom of Form
Forbes
http://www.forbes.com/
Accessed 13 Feb 2021
Editors’ Pick |
13 Feb 2021
Why Vaccine Passports Are ‘Inevitable,’ Explained By Tony Blair
The former British Prime Minister says they are the fastest way back to normal.
By Suzanne Rowan Kelleher Forbes Staff
Foreign Affairs
http://www.foreignaffairs.com/
Accessed 13 Feb 2021
[No new, unique, relevant content]
Foreign Policy
http://foreignpolicy.com/
Accessed 13 Feb 2021
Argument
Why Europe Is Falling Behind on Vaccines
It’s a perfect storm of under procurement, overzealous regulators, and anti-vaccine populations.
By Eyck Freymann, Elettra Ardissino
| February 8, 2021, 12:02 PM
The Guardian
http://www.guardiannews.com/
Accessed 13 Feb 2021
[No new, unique, relevant content]
New Yorker
http://www.newyorker.com/
Accessed 13 Feb 2021
Our Local Correspondents
Andrew Cuomo’s Refusal to Vaccinate Inmates Is Indefensible
A new lawsuit argues that withholding the COVID-19 vaccine from New York’s prisoners puts lives in danger, threatens public health, and endangers civil rights.
By Eric Lach 13 Feb 2021
New York Times
http://www.nytimes.com/
Accessed 13 Feb 2021
World
California extends vaccine eligibility to people with disabilities after backlash.
The state will extend Covid-19 vaccinations to people over 16 who are debilitated or immunocompromised by cancer and other diseases and chronic conditions.
By Shawn Hubler Feb 12
Europe
With the Economy on the Ropes, Hungary Goes All In on Mass Vaccination
Hungary became the first European Union country to administer Russia’s Sputnik vaccine, and will soon start with one from China — posing a challenge to the bloc’s joint vaccination strategy.
By Benjamin Novak Feb 12
U.S.
‘Open season’ for vaccine eligibility could come in April, Fauci says.
Dr. Anthony Fauci said most members of the general public could become eligible to get the vaccine in April, but it would take “several more months” after that to distribute shots.
By Jacey Fortin Feb 11
World
Vaccines are the new diplomatic currency, and other news from around the world.
By Mujib Mashal and Vivian Yee Feb 11
Washington Post
https://www.washingtonpost.com/
Accessed 13 Feb 2021
Lack of health services and transportation impede access to vaccine in communities of color
Akilah Johnson · Health · Feb 13, 2021
Brazil governors seek own vaccine supplies as stocks run low
Feb 13, 2021
SAO PAULO — Brazilian state governors are pursuing their own vaccine supply plans, with some expressing concern that President Jair Bolsonaro’s government won’t deliver the shots required to avoid interrupting immunization efforts.
Governors are under pressure from mayors, some of whose vaccine stocks have already been depleted, including three cities in the metropolitan area of Rio de Janeiro. Northeastern Bahia state’s capital Salvador suspended vaccination on Thursday because supplies are dwindling. Brazil’s two biggest cities, Rio and Sao Paulo, are expected to be without shots in a matter of days…
Biden says 300 million Americans can be vaccinated by July
National · Feb 11, 2021