COVID Therapeutics
Therapeutics and COVID-19: living guideline
WHO 14 January 2022
Overview
The WHO Therapeutics and COVID-19: living guideline contains the Organization’s most up-to-date recommendations for the use of therapeutics in the treatment of COVID-19. The latest version of this living guideline is available in pdf format (via the ‘Download’ button) and via an online platform, and is updated regularly as new evidence emerges.
This eighth version of the WHO living guideline now contains 14 recommendations, including three new recommendations regarding Janus kinase (JAK) inhibitors and sotrovimab. No further updates to the previous existing recommendations were made in this latest version.
::::::
WHO recommends two new drugs to treat COVID-19
14 January 2022 Geneva
Statement
WHO has recommended two new drugs for COVID-19, providing yet more options for treating the disease. The extent to which these medicines will save lives depends on how widely available and affordable they will be.
The first drug, baricitinib, is strongly recommended for patients with severe or critical COVID-19. It is part of a class of drugs called Janus kinase (JAK) inhibitors that suppress the overstimulation of the immune system. WHO recommends that it is given with corticosteroids.
Baricitinib is an oral drug, used in the treatment of rheumatoid arthritis. It provides an alternative to other arthritis drugs called Interleukin-6 receptor blockers, recommended by WHO in July 2021.
WHO has also conditionally recommended the use of a monoclonal antibody drug, sotrovimab, for treating mild or moderate COVID-19 in patients who are at high risk of hospitalization. This includes patients who are older, immunocompromised, having underlying conditions like diabetes, hypertension, and obesity, and those unvaccinated.
Sotrovimab is an alternative to casirivimab-imdevimab, a monoclonal antibody cocktail recommended by WHO in September 2021. Studies are ongoing on the effectiveness of monoclonal antibodies against Omicron but early laboratory studies show that sotrovimab retains its activity.
The panel of experts developing the guidelines also looked at two other drugs for severe and critical COVID-19: ruxolitinib and tofacitinib. Given their uncertain effects, WHO made a conditional recommendation against their use.
Today’s recommendations, forming the eighth update of WHO’s living guidelines on therapeutics and COVID-19, are based on evidence from seven trials involving over 4,000 patients with non-severe, severe, and critical COVID-19.
WHO is in discussions with manufacturers to secure global supply capacity and equitable and sustainable access to the newly recommended therapeutics. The Access to COVID-19 Tools Accelerator (ACT-A) Therapeutics pillar has been engaging with pharmaceutical companies to seek comprehensive access plans for low- and middle-income countries, so that these treatments can be rapidly deployed everywhere, not just in rich countries. The ACT-A is also looking to expand licensing scope to make the products more affordable.
The two newly recommended drugs – baricitinib and sotrovimab – have been invited for WHO Prequalification, which assesses the quality, efficacy and safety of priority health products to increase access in lower income countries.
::::::
Editor’s Note:
We reviewed WHO COVID and ACT Tool Accelerator websites to identify additional detail on the “WHO discussions” involving manufacturers referenced above [regarding global supply capacity and equitable and sustainable access to treatments/therapeutics]. We found no announcements or additional detail.
Further, we reviewed the Outcomes Statement of the 8th ACT-Accelerator Facilitation Council meeting of 9 December 2021. The statement makes no mention of discussions on supply and equitable access to COVID therapeutics.
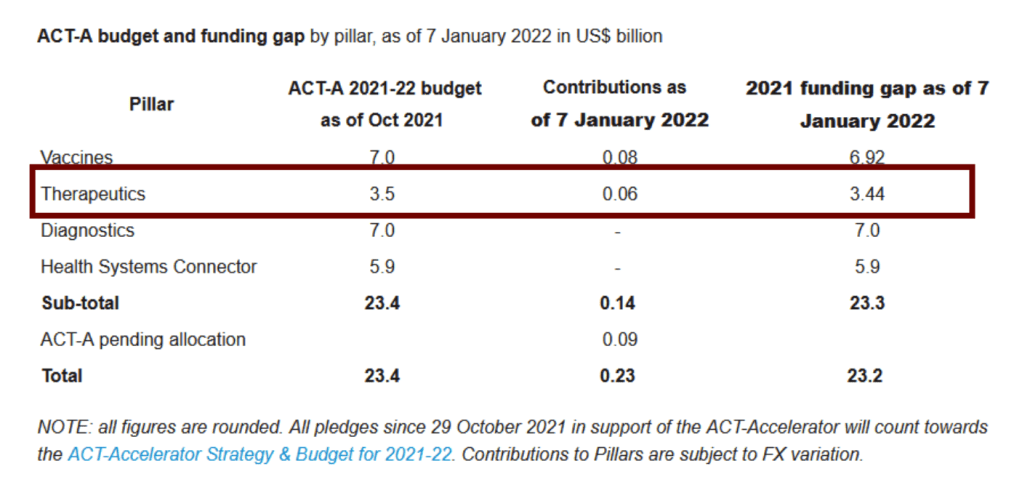
We also note that the Access to COVID-19 tools funding commitment tracker [see full announcement below] reports that the “Therapeutics” ACT-A 2021-22 budget of US$3.5 billion has received contributions of only USD$.06 billion as if January 7, 2022, a USD$3.44 billion shortfall.
Finally, we note that the ACT-Accelerator Strategic Plan & Budget: October 2021 to September 2022 [28 October 2021] addresses therapeutics:
3.3 Therapeutics
Co-convened by Unitaid and Wellcome, with WHO
The Therapeutics Pillar supports access to safe and effective therapeutics of assured quality to save
millions of lives from COVID-19 and to minimise its global health and economic threat.
Context. With the challenges of an evolving pandemic and difficulty in large scale uptake of
vaccines, therapeutics will remain critical in the COVID-19 response, saving lives and reducing
the burden on health systems (see Section 1). Among existing options, medical oxygen and
corticosteroids are mainstays of treatment for severe and critical COVID-19, along with
recent additions to clinical guidelines. ACT-A is working to drive uptake of existing products
and support equitable access to novel therapies for LICs and LMICs – despite challenges in
supply, price, and implementation. To date, there have been few recommended therapeutics
to treat mild and moderate cases in outpatient settings. But if proven effective, emerging
oral outpatient drugs – deployed with tests -could fundamentally change the response. One
example – subject to clinical review – could be the antiviral compound molnupiravir, for which
promising interim results were recently announced. Effective oral outpatient drugs would offer
a widespread, scalable way to stop disease progression, ease strain on health systems and
reduce deaths.
Setting a target. Approximately 200 million COVID-19 cases are expected globally in the next 12 months20, with potential for increased case numbers due to new variants or continued transmission amongst the vaccinated21. The Therapeutics Pillar aims to treat up to ~120 million22 of these cases (6-8 million severe and 113 million mild / moderate), focusing on LICs, LMICs and UMICs.
Therapeutics Pillar priorities: To achieve that target by September 2022, the Therapeutics
Pillar will prioritize deployment of existing effective products, including medical oxygen for
severe and critical cases, introduction of new COVID-19 therapies once proven, especially
those with a profile conducive to access in LICs and LMICs, and strengthening of research
efforts to enrich the therapeutic clinical pipeline. Alongside this, there will be a need to ensure
country readiness for effective implementation of tools, including supporting a test-and-treat
strategy for outpatient care in eligible patients…