COVID IP – WIPO Report
New COVID-19 Research: Universities and Research Organizations Highly Active in Vaccine Patenting During Pandemic’s Early Days; China, U.S.-based Applicants Lead in Vaccine and Therapeutics Innovation
Mar 9, 2022 PR/2022/887
Universities and research organizations filed nearly as many patent applications as corporations for COVID-19 vaccines during the early months of the global pandemic, with China and U.S.-based innovators most actively patenting new anti-COVID19 vaccine and therapeutic technologies, according to a WIPO report on pandemic-era innovation trends released today.
“This report underlines that collaboration – across organizations, agencies, sectors and borders – is essential if we are to make meaningful progress in addressing the global challenges that we face,” said Mr. Tang, who was joined at an event launching the report by World Health Organization Director General Tedros Adhanom Ghebreyesus and World Trade Organization Director General Ngozi Okonjo-Iweala.
Among the report’s key findings
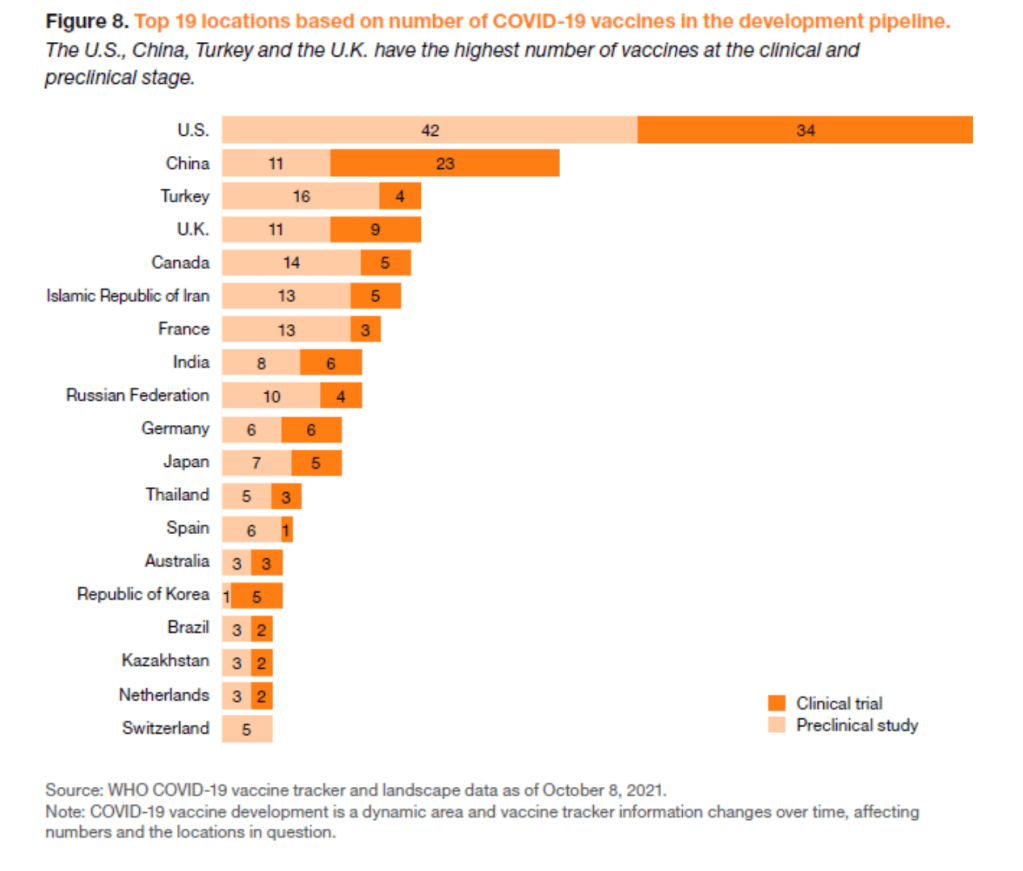
In the first 21 months of the pandemic, close to 5,300 patent applications relating to COVID-19 were filed across 49 patent offices.
This included nearly 1,500 filings related to therapeutics and over 400 filings related to vaccines.
For vaccine filings, universities and public research organizations accounted for 44 percent of the total, compared with 49 percent by companies. As one point of comparison, universities and public research organizations accounted for only 8 percent of total international patent applications filed with WIPO in 2021.
The top 10 applicant locations for vaccines were China, the U.S., the Russian Federation, the U.K., Republic of Korea, Germany, India, Austria, Switzerland and Australia.
China, the U.S. and India were the top origins for therapeutics. India and the Republic of Korea saw higher filing activity for therapeutics than vaccines.
Initial data from top patent offices showed comparatively quick grants for patent applications related to COVID-19, with innovators utilizing general fast-track avenues or specific COVID-19 measures designed to bring new products to the public quickly. By comparison, they were processed faster than patent applications in the field of chemistry and bioscience in the same period (January 2020 to September 2021).
The report also highlights how research organizations and universities worked together with private industry to help speed the development of life-saving COVID-19 vaccines and therapeutics, and confirms that accelerated innovation and vaccine development during the pandemic were possible thanks to research breakthroughs and technological advancements pre-pandemic.
WIPO Patent Landscape Report – COVID-19-related vaccines and therapeutics
Preliminary insights on related patenting activity during the pandemic
Author(s): World Intellectual Property Organization (WIPO) |
Publication year: 2022 :: 83 pages
Download: https://www.wipo.int/publications/en/details.jsp?id=4589