Revista Panamericana de Salud Pública/Pan American Journal of Public Health (RPSP/PAJPH)
https://www.paho.org/journal/en
Management and impact of interventions to reduce COVID-19 cases in Costa Rica
Original research | Spanish |
13 Apr 2022
Revista Panamericana de Salud Pública/Pan American Journal of Public Health (RPSP/PAJPH)
https://www.paho.org/journal/en
Management and impact of interventions to reduce COVID-19 cases in Costa Rica
Original research | Spanish |
13 Apr 2022
Risk Management and Healthcare Policy
https://www.dovepress.com/risk-management-and-healthcare-policy-archive56
[Accessed 16 Apr 2022]
Original Research
Acceptance of COVID-19 Vaccine Among High-Risk Occupations in a Port City of China and Multifaceted Strategies for Increasing Vaccination Coverage: A Cross-Sectional Study
Sun Y, Li B, Li N, Li B, Chen P, Hao F, Sun C
Risk Management and Healthcare Policy 2022, 15:643-655
Published Date: 14 April 2022
Science
Volume 376| Issue 6590| 15 Apr 2022
https://www.science.org/toc/science/current
Policy Forum
Getting genetic ancestry right for science and society
BY Anna C. F. Lewis, et al.
14 Apr 2022: 250-252
We must embrace a multidimensional, continuous view of ancestry and move away from continental ancestry categories
Science Translational Medicine
Volume 14| Issue 640| 13 Apr 2022
https://www.science.org/toc/stm/current
Reviews
Point-of-care diagnostic tests for tuberculosis disease
BY Jia Mei Hong, et al.
06 Apr 2022
Advances in biomarkers and microfluidics technology facilitate the development of TB disease diagnostic point-of-care tests.
Vaccine
Volume 40, Issue 13 Pages 1913-2122 (18 March 2022)
https://www.sciencedirect.com/journal/vaccine/vol/40/issue/13
Review article Open access
Epidemiology and economic burden of meningococcal disease in Germany: A systematic review
S. Gruhn, J. Witte, W. Greiner, O. Damm, … M. Knuf
Pages 1932-1947
Vaccine
Volume 40, Issue 13 Pages 1913-2122 (18 March 2022)
https://www.sciencedirect.com/journal/vaccine/vol/40/issue/13
Research article Open access
Long-term effectiveness of human papillomavirus vaccines among adult women: A real-world scenario
Ga Young Lee, Perapong Inthasorn, Piyawat Laowahutanont, Saranath Lawpoolsri, … Punnee Pitisuttithum
Pages 1968-1976
Vaccine
Volume 40, Issue 13 Pages 1913-2122 (18 March 2022)
https://www.sciencedirect.com/journal/vaccine/vol/40/issue/13
Research article Full text access
Promoting immunization equity in Latin America and the Caribbean: Case studies, lessons learned, and their implication for COVID-19 vaccine equity
Isabella L. Chan, Robin Mowson, Juan Pedro Alonso, Javier Roberti, … Martha Velandia-González
Vaccine
Volume 40, Issue 13 Pages 1913-2122 (18 March 2022)
https://www.sciencedirect.com/journal/vaccine/vol/40/issue/13
Research article Open access
Impact of the Sinopharm’s BBIBP-CorV vaccine in preventing hospital admissions and death in infected vaccinees: Results from a retrospective study in the emirate of Abu Dhabi, United Arab Emirates (UAE)
Farida Ismail AlHosani, Anderson Eduardo Stanciole, Bashir Aden, Andrey Timoshkin, … Farah Mustafa
Vaccine
Volume 40, Issue 13 Pages 1913-2122 (18 March 2022)
https://www.sciencedirect.com/journal/vaccine/vol/40/issue/13
Research article Open access
Unmet need for COVID-19 vaccination coverage in Kenya
Samuel K. Muchiri, Rose Muthee, Hellen Kiarie, Joseph Sitienei, … Victor A. Alegana
Pages 2011-2019
Vaccine
Volume 40, Issue 13 Pages 1913-2122 (18 March 2022)
https://www.sciencedirect.com/journal/vaccine/vol/40/issue/13
Research article Full text access
The correlates and dynamics of COVID-19 vaccine-specific hesitancy
Eric Merkley, Peter John Loewen
Pages 2020-2027
Vaccine
Volume 40, Issue 13 Pages 1913-2122 (18 March 2022)
https://www.sciencedirect.com/journal/vaccine/vol/40/issue/13
Research article Full text access
Public perceptions and the willingness to get vaccinated against COVID-19: Lessons from Israel
Oren Heller, Yung Chun, Yaniv Shlomo, Ateret Gewirtz-Meydan, … Michal Grinstein-Weiss
Vaccine
Volume 40, Issue 13 Pages 1913-2122 (18 March 2022)
https://www.sciencedirect.com/journal/vaccine/vol/40/issue/13
Research article Open access
Conspiracy theories and misinformation about COVID-19 in Nigeria: Implications for vaccine demand generation communications
Chizoba Wonodi, Chisom Obi-Jeff, Funmilayo Adewumi, Somto Chloe Keluo-Udeke, … Ruth Faden
Pre-Print Servers
Gates Open Research
https://gatesopenresearch.org/browse/articles
Open Letter metrics AWAITING PEER REVIEW
Women’s groups and COVID-19: An evidence review on savings groups in Africa [version 1; peer review: awaiting peer review]
Olayinka Adegbite, Leigh Anderson, Sybil Chidiac, Osasuyi Dirisu, Jenna Grzeslo, Julia Hakspiel, Chinmaya Holla, Emily Janoch, Krishna Jafa, Shubha Jayaram, Grace Majara, Tabitha Mulyampiti, Eve Namisango, Eva Noble, Bukola Onyishi, David Panetta, Garima Siwach, Munshi Sulaiman, Rebecca Walcott, Sapna Desai, Thomas de Hoop
Peer Reviewers Invited
Funder
The Bill and Melinda Gates Foundation
PUBLISHED 12 Apr 2022
medRxiv
https://www.medrxiv.org/content/about-medrxiv
medRxiv is a free online archive and distribution server for complete but unpublished manuscripts (preprints) in the medical, clinical, and related health sciences. Preprints are preliminary reports of work that have not been certified by peer review. They should not be relied on to guide clinical practice or health-related behavior and should not be reported in news media as established information. medRxiv is for the distribution of preprints – complete but unpublished manuscripts – that describe human health research conducted, analyzed, and interpreted according to scientific principles…
COVID-19 vaccine coverage among immigrants and refugees in Alberta: a population-based cross-sectional study
Shannon E MacDonald, Yuba Raj Paudel, Crystal Du
medRxiv 2022.04.11.22273644; doi: https://doi.org/10.1101/2022.04.11.22273644
Can We Really Trust the Findings of the COVID-19 Research? Quality Assessment of Randomized Controlled Trials Published on COVID-19
Athira S Joshy, Christy Thomas, Saphal Surendran, Krishna Undela
medRxiv 2022.04.15.22273881; doi: https://doi.org/10.1101/2022.04.15.22273881
Mondo: Unifying diseases for the world, by the world
Nicole A Vasilevsky, Nicolas A Matentzoglu, Sabrina Toro, Joe E Flack, Harshad Hegde, Deepak R Unni, Gioconda Alyea, Joanna S Amberger, Larry Babb, James P Balhoff, Taylor I Bingaman, Gully A Burns, Tiffany J Callahan, Leigh C Carmody, Lauren E Chan, George S Chang, Michel Dumontier, Laura E Failla, May J Flowers, H A Garrett Jr., Dylan Gration, Tudor Groza, Marc Hanauer, Nomi L Harris, Ingo Helbig, Jason A Hilton, Daniel S Himmelstein, Charles T Hoyt, Megan S Kane, Sebastian Kohler, David Lagorce, Martin Larralde, Antonia Lock, Irene Lopez Santiago, Donna R Maglott, Adriana J Malheiro, Birgit HM Meldal, Julie A McMurry, Moni Munoz-Torres, Tristan H Nelson, David Ochoa, Tudor I Oprea, David Osumi-Sutherland, Helen Parkinson, Zoe M Pendlington, Ana Rath, Heidi L Rehm, Lyubov Remennik, Erin R Riggs, Paola Roncaglia, Justyne E Ross, Marion F Shadbolt, Kent A Shefchek, Morgan N Similuk, Nicholas Sioutos, Rachel Sparks, Ray Stefancsik, Ralf Stephan, Doron Stupp, Jagadish Chandrabose Sundaramurthi, Imke Tammen, Courtney L Thaxton, Eloise Valasek, Alex H Wagner, Danielle Welter, Patricia L Whetzel, Lori L Whiteman, Valerie Wood, Colleen H Xu, Andreas Zankl, Xingmin A Zhang, Christopher G Chute, Peter N Robinson, Christopher J Mungall, Ada Hamosh, Melissa A Haendel
medRxiv 2022.04.13.22273750; doi: https://doi.org/10.1101/2022.04.13.22273750
There are thousands of distinct disease entities and concepts, each of which are known by different and sometimes contradictory names. The lack of a unified system for managing these entities poses a major challenge for both machines and humans that need to harmonize information to better predict causes and treatments for disease. The Mondo Disease Ontology is an open, community-driven ontology that integrates key medical and biomedical terminologies, supporting disease data integration to improve diagnosis, treatment, and translational research. Mondo records the sources of all data and is continually updated, making it suitable for research and clinical applications that require up-to-date disease knowledge.
Triangulating evidence in health sciences with Annotated Semantic Queries
Yi Liu, Tom R Gaunt
medRxiv 2022.04.12.22273803; doi: https://doi.org/10.1101/2022.04.12.22273803
Profile of Brazilian inpatients with COVID-19 vaccine breakthrough infection and risk factors for unfavorable outcome
Matheus Alexandre Santos de Jesus, Natália Satchiko Hojo-Souza, Thiago Rocha de Moraes, Daniel Ludovico Guidoni, Fernanda Sumika Hojo Souza
medRxiv 2022.04.12.22273589; doi: https://doi.org/10.1101/2022.04.12.22273589
Effectiveness of COVID-19 vaccines against hospitalization and death in Canada: A multiprovincial test-negative design study
Sharifa Nasreen, Yossi Febriani, Héctor Alexander Velásquez García, Geng Zhang, Mina Tadrous, Sarah A. Buchan, Christiaan H. Righolt, Salaheddin M. Mahmud, Naveed Zafar Janjua, Mel Krajden, Gaston De Serres, Jeffrey C. Kwong
medRxiv 2022.04.13.22273825; doi: https://doi.org/10.1101/2022.04.13.22273825
Health Economic Burden of COVID-19 in Saudi Arabia
Khalidah A. Alenzi, Hamdan S. Al-malky, Ali F. Altebainawi, Hamidah Q. Abushomi, Fahad O. Alatawi, Moosa H. Atwadi, Moosa A. Khobrani, Dlal A. Almazrou, Nariman Alrubeh, Zainab A. Alsoliabi, Abdulaziz M. Kardam, Shakr A. Alghamdi, Abdulrahman Alasiri, Thamir M. Alshammari
medRxiv 2022.04.08.22273439; doi: https://doi.org/10.1101/2022.04.08.22273439
Effectiveness of the BNT162b vaccine fourth dose in reducing SARS-CoV-2 infection among healthcare workers in Israel, a multi-center cohort study
Matan J Cohen, Yonatan Oster, Allon E Moses, Avishay Spitzer, Shmuel Benenson, the Israeli-hospitals 4th vaccine Working Group
medRxiv 2022.04.11.22273327; doi: https://doi.org/10.1101/2022.04.11.22273327
The relation between COVID-19 vaccinations and public governance to improve preparedness of next pandemic impacts and crisis management: a global study
Mario Coccia, Igor Benati
medRxiv 2022.04.10.22273663; doi: https://doi.org/10.1101/2022.04.10.22273663
Abstract
The goal of this study is to analyze the relationship between COVID-19 vaccinations and public governance performing a global analysis of more than 110 countries worldwide. Methodology applies the Independent Samples T-Test that compares the means of two independent groups (countries with high/low level of vaccinations) to determine whether there is statistical evidence that the associated population means of indicators of public governance are significantly different. Findings suggest that high levels of governance can support a better function of health systems in the rollout of vaccinations to cope with COVID-19 pandemic crisis. This study may assist long-run policy of governments to improve good governance and health systems of countries in order to reinforce the preparedness to face next pandemic threats and in general future crisis management in society.
Economic vulnerability and poor service delivery made it more difficult for shack-dwellers to comply with COVID-19 restrictions: The impracticability and inequitable burden of universal/unstratified public health policies
GTH Ellison, RB Mattes, H Rhoma, T De Wet
medRxiv 2022.04.07.22273499; doi: https://doi.org/10.1101/2022.04.07.22273499
COVID-19 vaccine for people who live and work in prisons worldwide: A scoping review
Nasrul Ismail, Lara Tavoschi, Babak Moazen, Alicia Roselló, Emma Plugge
medRxiv 2022.04.07.22273414; doi: https://doi.org/10.1101/2022.04.07.22273414
Novel Privacy Considerations for Large Scale Proteomics
Andrew C. Hill, Elizabeth M. Litkowski, Ani Manichaikul, Leslie Lange, Katherine A. Pratte, Katerina J. Kechris, Matthew DeCamp, Marilyn Coors, Victor E. Ortega, Stephen S. Rich, Jerome I. Rotter, Robert E. Gerzsten, Clary B. Clish, Jeffery Curtis, Xiaowei Hu, Debby Ngo, Wanda K O’Neal, Deborah Meyers, Eugene Bleecker, Brian D. Hobbs, Michael H. Cho, Farnoush Banaeikashani, Russell P. Bowler
medRxiv 2022.04.06.22269907; doi: https://doi.org/10.1101/2022.04.06.22269907
COVID-19 Vaccination Mandates and Vaccine Uptake
Alexander Karaivanov, Dongwoo Kim, Shih En Lu, Hitoshi Shigeoka
medRxiv 2021.10.21.21265355; doi: https://doi.org/10.1101/2021.10.21.21265355 Revision
Acceptance of and preference for COVID-19 vaccination in healthcare workers: a comparative analysis and discrete choice experiment
Chuanxi Fu, Zheng wei, Fengchang Zhu, Sen Pei, Shunping Li, Liuren Zhang, Xiaohui Sun, Yue Wu, Ping Liu, Mark Jit
medRxiv 2020.04.09.20060103; doi: https://doi.org/10.1101/2020.04.09.20060103 Revision
SARS-CoV-2 reinfections with BA.1 (Omicron) variant among fully vaccinated individuals in the northeast of Brazil
Francisco P. F. Neto, Diego G. Teixeira, Dayse C. S. da Cunha, Ingryd C. Morais, Celisa P. M. Tavares, Genilson P. Gurgel, Sanderson D. do Nascimento, David C. dos Santos, Alexandre de O. Sales, Selma M.B. Jerônimo
medRxiv 2022.04.08.22272726; doi: https://doi.org/10.1101/2022.04.08.22272726
Wellcome Open Research [to 16 Apr 2022]
https://wellcomeopenresearch.org/browse/articles
[Accessed 16 Apr 2022]
Wellcome Open Research provides all Wellcome researchers with a place to rapidly publish any results they think are worth sharing. All articles benefit from rapid publication, transparent peer review and editorial guidance on making all source data openly available.
Data Note metrics AWAITING PEER REVIEW
An open dataset of Plasmodium vivax genome variation in 1,895 worldwide samples [version 1; peer review: awaiting peer review]
MalariaGEN, Ishag Adam, Mohammad Shafiul Alam, Sisay Alemu, Chanaki Amaratunga, Roberto Amato, Voahangy Andrianaranjaka, Nicholas M Anstey, Abraham Aseffa, Elizabeth Ashley, Ashenafi Assefa, Sarah Auburn, Bridget E Barber, Alyssa Barry, Dhelio Batista Pereira, Jun Cao, Nguyen Hoang Chau, Kesinee Chotivanich, Cindy Chu, Arjen M. Dondorp, Eleanor Drury, Diego F. Echeverry, Berhanu Erko, Fe Espino, Rick Fairhurst, Abdul Faiz, María Fernanda Villegas, Qi Gao, Lemu Golassa, Sonia Goncalves, Matthew J Grigg, Yaghoob Hamedi, Tran Tinh Hien, Ye Htut, Kimberly J Johnson, Nadira Karunaweera, Wasif Khan, Srivicha Krudsood, Dominic P Kwiatkowski, Marcus Lacerda, Benedikt Ley, Pharath Lim, Yaobao Liu, Alejandro Llanos-Cuentas, Chanthap Lon, Tatiana Lopera-Mesa, Jutta Marfurt, Pascal Michon, Olivo Miotto, Rezika Mohammed, Ivo Mueller, Chayadol Namaik-larp, Paul N Newton, Thuy-Nhien Nguyen, Francois Nosten, Rintis Noviyanti, Zuleima Pava, Richard D Pearson, Beyene Petros, Aung P Phyo, Ric N Price, Sasithon Pukrittayakamee, Awab Ghulam Rahim, Milijaona Randrianarivelojosia, Julian C Rayner, Angela Rumaseb, Sasha V Siegel, Victoria J Simpson, Kamala Thriemer, Alberto Tobon-Castano, Hidayat Trimarsanto, Marcelo Urbano Ferreira, Ivan D Vélez, Sonam Wangchuk, Thomas E Wellems, Nicholas J White, Timothy William, Maria F Yasnot, Daniel Yilma
Peer Reviewers Invited
Funders
Wellcome Trust
Medical Research Council UK and the Department for International Development (DFID)
Bill and Melinda Gates Foundation
PUBLISHED 14 Apr 2022
Research Article metrics
Revised
Stage 2 Registered Report: How responsibility attributions to self and others relate to outcome ownership in group decisions [version 2; peer review: 2 approved]
Matt Jaquiery, Marwa El Zein
Peer Reviewers John A. Dewey; Nura Sidarus
Funders
Wellcome Trust
Medical Research Council
University of Oxford
LATEST VERSION PUBLISHED 11 Apr 2022
Research Article metrics
Revised
A survey to gather perspectives of DBT/Wellcome Trust India Alliance-funded researchers on public engagement with science [version 2; peer review: 2 approved]
Sarah Iqbal, Banya Kar
Peer Reviewers Marina Joubert; Mary Chambers and Han Dong Thai Tran
Funder
DBT/Wellcome Trust India Alliance
LATEST VERSION PUBLISHED 08 Apr 2022
Think Tanks
Brookings
http://www.brookings.edu/
Accessed 16 Apr 2022
[No new digest content identified]
Center for Global Development [to 16 Apr 2022]
https://www.cgdev.org/
Research [Selected]
POLICY PAPERS
Beyond India’s Lockdown: PMGKY Benefits During the COVID-19 Crisis and the State of Digital Payments
Alan Gelb et al.
April 11, 2022
India imposed a lock-down in response to the COVID-19 pandemic in March 2020 and began a gradual re-opening in June. A telephonic survey in April examined the early effectiveness of information and the massive PMGKY social protection program (Policy Paper 217). This paper analyzes a second-round sur…
Chatham House [to 16 Apr 2022]
https://www.chathamhouse.org/
Accessed 16 Apr 2022
[No new digest content identified]
CSIS
https://www.csis.org/
Accessed 16 Apr 2022
Upcoming Event
Equity in Immunization Services to Ensure “A Long Life for All”
April 28, 2022
Upcoming Event
Innovation and IP’s Role in Combatting the Covid-19 Pandemic
April 27, 2022
Upcoming Event
AMR as a Global Security Threat: Destabilizing Food Systems and Healthy Communities
April 25, 2022
Commentary
Removing Patent Rights to Lower Drug Costs Is a Dangerous Precedent
April 14, 2022 | By Walter G. Copan
Kaiser Family Foundation
https://www.kff.org/search/?post_type=press-release
Accessed 16 Apr 2022
Accessed 16 Apr 2022
[No new digest content identified]
ODI [Overseas Development Institute] [to 16 Apr 2022]
https://odi.org/en/publications/
Publications
Accessed 16 Apr 2022
[No new digest content identified]
Rand [to 16 Apr 2022]
https://www.rand.org/pubs.html
Reports, Selected Journal Articles
Report
How Extremism Operates Online: A Primer
In this Perspective, the second in a RAND Corporation series on online extremist material, the authors explore how the internet affects extremist activities and how exposure to or consumption of such content influences the behavior of internet users.
Apr 12, 2022
Alexandra T. Evans, Heather J. Williams
Vaccines and Global Health: The Week in Review is a weekly digest summarizing news, events, announcements, peer-reviewed articles and research in the global vaccine ethics and policy space. Content is aggregated from key governmental, NGO, international organization and industry sources, key peer-reviewed journals, and other media channels. This summary proceeds from the broad base of themes and issues monitored by the Center for Vaccine Ethics & Policy in its work: it is not intended to be exhaustive in its coverage. You are viewing the blog version of our weekly digest, typically comprised of between 30 and 40 posts below all dated with the current issue date
.– Request an Email Summary: Vaccines and Global Health : The Week in Review is published as a single email summary, scheduled for release each Saturday evening before midnight (EDT in the U.S.). If you would like to receive the email version, please send your request to david.r.curry@centerforvaccineethicsandpolicy.org.
– pdf version: A pdf of the current issue is available here:
– blog edition: comprised of the approx. 35+ entries posted below.
– Twitter: Readers can also follow developments on twitter: @vaxethicspolicy.
.
– Links: We endeavor to test each link as we incorporate it into any post, but recognize that some links may become “stale” as publications and websites reorganize content over time. We apologize in advance for any links that may not be operative. We believe the contextual information in a given post should allow retrieval, but please contact us as above for assistance if necessary.
Support this knowledge-sharing service: Your financial support helps us cover our costs and to address a current shortfall in our annual operating budget. Click here to donate and thank you in advance for your contribution.
.
David R. Curry, MS
Executive Director
Center for Vaccine Ethics and Policy
GAVI COVAX Advance Market Commitment Summit 2022
One Third of Humanity Still Unvaccinated, Secretary-General Tells GAVI COVAX Advance Market Commitment Summit 2022, Citing ‘Deeply Unequal World’
8 April 2022
SG/SM/21233
Following is the text of UN Secretary-General António Guterres’ video message for the GAVI COVAX Advance Market Commitment Summit 2022, “One World Protected – Break COVID Now”, in New York today:
I commend the Government of Germany and GAVI for organizing this important Summit. This gathering is a critical reminder that the COVID-19 pandemic is far from over.
We’re seeing 1.5 million new cases each day. Large outbreaks are spreading in Asia. A new wave is sweeping across Europe. And some countries are reporting their highest death rates since the start of the pandemic.
Omicron is a startling reminder of how quickly COVID-19 can mutate and spread, especially in the absence of high vaccination coverage. Some high-income countries are preparing for their second booster doses. And yet one third of humanity remains unvaccinated. This is a brutal indictment of our deeply unequal world. It’s also a prime breeding ground for new variants, more deaths and increased human and economic misery. The next variant is not a question of “if”. It’s a question of “when”.
We are far from our target of every country reaching 70 per cent vaccination coverage by the middle of this year. And with new variants emerging every four months on average, time is of the essence. Supply is not the issue. Manufacturers are producing 1.5 billion doses per month. And thanks to its remarkable procurement, shipment and delivery system, the COVAX Facility and its Advance Market Commitment mechanism has managed to deliver 1.2 billion doses so far to countries in need.
This proves that progress is possible. But, the window is closing fast. Governments and pharmaceutical companies need to work better together to deliver vaccines to every person, everywhere — not just in wealthy countries. This means countries fulfilling and accelerating dose-sharing and donation commitments to COVAX with better quality of supply. And it means having strong national vaccinedelivery systems at the ready — including efforts to counter disinformation and get vaccines into arms.
I also call on countries to commit new funding for the ACT-Accelerator and COVAX this year. And we need to help all countries prepare for future pandemics by multiplying the number of countries able to locally produce tests, vaccines and treatments. So, we can save lives quickly and equitably when the next variant or pandemic strikes. And ultimately, build stronger health systems that are accessible to all.
Investments today will not only save lives. They will strengthen overwhelmed health systems for the future, and help all countries move closer to sustainable recovery. The COVID-19 pandemic is not over. But, it can be. Let’s end it together.
::::::
World leaders commit US$ 4.8 billion to help Break COVID Now
2022 Break COVID Now Summit co-hosted by Gavi, alongside Germany (G7 President), Ghana, Indonesia (G20 President), and Senegal (African Union chair) reaffirms support for COVAX and global solidarity in fighting the COVID-19 pandemic
Summit sees US$ 4.8 billion in commitments, securing vital cash to help lower-income countries boost vaccinations now, secure equitable access for new vaccine procurements plus additional help for countries looking to procure their own vaccines in the future. Indonesia’s President, leading the G20 this year, extended his support for the launch of the Pandemic Vaccine Pool as a key instrument for response and resiliency.
Professor José Manuel Barroso, Chair of the Gavi Board, said: “We welcome this incredible show of global solidarity from so many stakeholders at a time when the world faces multiple challenges.”
Geneva, 8 April 2022 – The 2022 Break COVID Now Summit, co-hosted by Gavi, the Vaccine Alliance alongside the governments of Germany, Ghana, Indonesia and Senegal saw world leaders come together to reaffirm support for equitable access to COVID-19 vaccines and acting urgently to break COVID now.
With Germany, Indonesia and Senegal holding Presidencies in the G7, G20 and AU respectively, the Summit represented an affirmation of the international community’s support for COVAX’s multilateral approach to vaccine equity. The event also successfully secured commitments valued at US$ 4.8 billion for the Gavi COVAX Advance Market Commitment (AMC), the mechanism which supports equitable access to COVID-19 vaccines for lower-income countries.
These commitments mean a total of US$ 1.7 billion in new sovereign donor pledges towards the 2022 AMC fundraising ask, as well as US$ 2.1 billion worth of commitments via new innovative financial mechanisms provided by the EIB and the United States Development Finance Corporation (DFC), and least US$ 1 billion made available by three multilateral development banks (MDBs) – World Bank, Asian Development Bank and European Investment Bank (EIB).
The commitments made will enable COVAX to provide urgent delivery support for lower-income countries and ensure dose donations can be shipped and administered. They have also enabled Gavi to launch the Pandemic Vaccine Pool to support future procurement of new COVID-19 vaccines, on behalf of COVAX AMC participants, should they be needed. Commitments from MDBs enable low-cost financing for these countries to purchase additional vaccines beyond those that are donor-funded.
New donor commitments made to the Gavi COVAX AMC include:
Japan, co-host of the 2021 AMC Summit, pledged US$ 500 million
Germany, co-host of today’s Break COVID Now Summit, pledged EUR 400 million
Canada pledged CAD 220 million
Brazil pledged US$ 86.7 million
European Commission EUR 75 million
Australia pledged AUD 85 million
Finland pledged EUR 2 million
Iceland pledged ISK 250 million
Luxembourg pledged EUR 1 million
Vietnam pledged US$ 500,000
Regional Government of Catalonia, Spain pledged EUR 290’000
Provincial Council of Bizkaia, Spain pledged EUR 100’000
Estonia pledged EUR 40,000
Malta pledged EUR 40,000
Workday Foundation pledged US$ 300,000
CODE(RED) Campaign pledged US$ 200,000
Other donors also pledged a total of US$ 122.6 million
The pledges made today build on top of the commitments made at the 2022 One World Protected event, which launched the 2022 AMC Investment Opportunity on 19 January.
A number of countries and groups expressed intention to make additional contributions to COVAX, including in support of the Pandemic Vaccine Pool, but were unable to announce today due to their budgetary processes and the short timeline of this emergency appeal…
IMF Working Paper
A Global Strategy to Manage the Long-Term Risks of COVID-19
Authors/Editors: Ruchir Agarwal ; Gita Gopinath ; Jeremy Farrar ; Richard Hatchett ; Peter Sands
Publication Date: April 5, 2022 :: 26 pages
Electronic Access: Free Download.
Summary:
The pandemic is not over, and the health and economic losses continue to grow. It is now evident that COVID-19 will be with us for the long term, and there are very different scenarios for how it could evolve, from a mild endemic scenario to a dangerous variant scenario. This realization calls for a new strategy that manages both the uncertainty and the long-term risks of COVID-19. There are four key policy implications of such as strategy.
First, we need to achieve equitable access beyond vaccines to encompass a comprehensive toolkit.
Second, we must monitor the evolving virus and dynamically upgrade the toolkit.
Third, we must transition from the acute response to a sustainable strategy toward COVID-19, balanced and integrated with other health and social priorities.
Fourth, we need a unified risk-mitigation approach to future infectious disease threats beyond COVID-19.
Infectious diseases with pandemic potential are a threat to global economic and health security. The international community should recognize that its pandemic financing addresses a systemic risk to the global economy, not just the development need of a particular country. Accordingly, it should allocate additional funding to fight pandemics and strengthen health systems both domestically and overseas. This will require about $15 billion in grants this year and $10 billion annually after that.
Series: Working Paper No. 2022/068
Disclaimer: IMF Working Papers describe research in progress by the author(s) and are published to elicit comments and to encourage debate. The views expressed in IMF Working Papers are those of the author(s) and do not necessarily represent the views of the IMF, its Executive Board, or IMF management.
New IMF Staff Paper Strategy to Manage the Long-Term Risks of COVID-19
April 5, 2022
Washington, DC: The International Monetary Fund, in partnership with the Coalition for Epidemic Preparedness Innovations (CEPI), the Global Fund, and Wellcome Trust published today “A Global Strategy to Manage the Long-term Risks of COVID-19” working paper, which calls for a more comprehensive and integrated pandemic response from the international community.
In the joint paper, the four global organizations assert that ending the pandemic everywhere remains an urgent economic, health, and moral priority for the world—advocating for the following:
Gita Gopinath, First Deputy Managing Director, IMF:
“It is now clear that COVID-19 is likely to be with us for the long-term. Given the many possible scenarios for the evolution of COVID-19 (from benign to severe scenarios), and given the limited resources countries have, we need a new strategy.
“Countries need a more comprehensive COVID-19 toolkit for fighting the pandemic that includes vaccines, tests, treatments – and bolstering the resilience of health systems so they are in a better position to tackle both COVID-19 and other deadly diseases in a sustainable, effective way.
“Overall, health security is economic security. As recently as our January World Economic Outlook Update, we’d estimated the cumulative losses from the pandemic to reach $13.8 trillion. The international community should recognize that its pandemic financing addresses a systemic risk to the global economy. Thus, we are calling for additional funding to fight pandemics and to strengthen health systems. This will require about $15 billion in grants this year and $10 billion annually after that. The cost of inaction – for all of us – is very high. We need to act – now.
“Together with our partners on the Multilateral Leaders Taskforce and with the ACT Accelerator, the IMF stands ready to help countries meet the challenges of the pandemic and their financing needs—including through a Resilience and Sustainability Trust (RST).”
Richard Hatchett, CEO, CEPI:
“In many ways COVID-19 has shown us the potential of human ingenuity and innovation when we direct out energy and resources in fighting a common enemy. It has also tragically thrown into sharp focus a global failure to work multilaterally to ensure equal access to life saving vaccines.
“Vaccines are, and will continue to be, at the forefront of how modern societies counter infectious disease threats. They are one of our most potent tools against pandemic risks and will be critical to any future response. But if they are to truly fulfill their potential in preventing future pandemics their development must also go alongside investments in global surveillance, R&D, manufacturing, and health systems.
“During the course of this pandemic we have seen scientific advances that may well have taken decades to make in ‘peacetime’. Now, more than ever, the world now has the tools, the platforms, the concepts, and the experience to build a system that can more effectively meet the threat of future viruses.
“A future in which we are to respond to the next Disease X with new vaccines, therapeutics, and diagnostics in just 100 days is possible—but it will take vision, political will, and commensurate financial investments from governments around the world. We estimate that it will cost the world around $10 billion a year to ensure adequate global pandemic preparedness: this price tag is substantial, but failure to invest now—and build on the gains made in the response to COVID-19—will result in human and economic costs that will reverberate for generations.”
Peter Sands, Executive Director, Global Fund:
“The next phase of the fight against COVID-19 will be different. We are in for a long fight against a virus that continues to evolve. So we must shift to a more sustainable response that recognizes the linkages between responding to COVID-19, tackling the earlier pandemics of HIV, TB and malaria, and preparing for future pandemic threats. We should step up investment in systems for health, intentionally maximizing the synergies between interventions against existing infectious diseases and initiatives to prevent, detect and respond to future infectious disease outbreaks. Stronger and more resilient systems for health, including community systems, will enable us to protect everyone, everywhere from the deadliest infectious diseases.”
Jeremy Farrar, Director, Wellcome Trust:
“These last two years have shown that remarkable progress is possible when the world comes together and supports science boldly at scale, across borders. This approach gave us lifesaving Covid-19 vaccines and treatments in record time. Now is not the time to ease up – the virus’s next move is anything but certain and the risk of new variants high. We all desperately want this pandemic to be over. But simply wishing for the most optimistic scenario won’t make it so.
“We need to set our sights on developing next generation vaccines that can block transmission and won’t require endless boosters, strengthening genomic surveillance globally so we can identify and track new variants and improving global access to vaccines, treatments and tests. Leaving any countries unprotected, puts us all at risk.
“Most importantly, the response must be built on international co-operation. Only by working together can leaders achieve long-lasting and sustainable recovery from Covid-19 and prepare for the epidemic and pandemic threats of tomorrow.”
Ukraine

[Excerpts]
1.2 Access to health care in Ukraine
There are many challenges to accessing health care, with active hostilities and a lack of public transport restricting movement. Close to 1000 health facilities are in proximity to conflict areas or are in changed areas of control, multiple hospitals have been repurposed to care for wounds and half of the pharmacies in Ukraine have closed, which leaves the health system vulnerable to infrastructural damage and severe disruptions in critical services. As a consequence, there is limited or no access to medicines, health facilities, and health-care workers in some areas. Between 24 February and 6 April a total of 91 attacks1 on health care have been reported, resulting in 46 injuries and 73 deaths.2 Further attacks are being verified. Since 24 February 274 hospitals have been shelled, 13 have been completely destroyed, and 70 ambulances have been disabled by shelling…
Risk of emergence and spread of infectious diseases
Ongoing epidemics
The incidence of COVID-19 continues to decrease, with 14 120 new cases and 147 new deaths reported between 31 March and 5 April. However, these numbers should be interpreted carefully due to underreporting of COVID-19 cases and deaths. From 23 February to 6 April the seven-day average number of polymerase chain reaction tests and antigen-rapid diagnostic tests has declined significantly, with a 93% (from 42 460 to 1531) and 89% (from 51 484 to 6194) decrease, respectively. Vaccination uptake remains low, particularly in vulnerable populations, and the disruption in testing and treatment puts those most vulnerable at increased risk of severe illness and death.
Epidemic risk
:: Poor ventilation and overcrowding increase the risk of spread of respiratory infections, including COVID-19. Lack of access to water, sanitation and hygiene (WASH) heightens the risk of emergence of foodborne and waterborne diseases. According to United Nations Office for the Coordination of Humanitarian Affairs (OCHA), around six million people have either limited or no access to safe water, with active hostilities preventing repair teams from fixing damaged systems and restoring access to water while also hindering the delivery of water in the hardest-hit areas, like the Donetsk and Luhansk oblasts. Of note, an outbreak of cholera was reported in 2011 in the Mariupol region, while a single case was reported in 2016 in the Zaporizhzhya oblast, highlighting the risk of cholera outbreaks.
:: Suboptimal vaccination coverage for routine and childhood immunizations, including measles and poliomyelitis (polio), increases the risk of re-emergence and transmission of vaccine-preventable diseases. Notably, two cases of circulating vaccine-derived poliovirus type 2 (cVDPV2) were reported in Ukraine in 2021. On 1 February 2022 a national supplemental polio immunization campaign targeting nearly 140 000 children was launched, but due to the current situation it has been deprioritized and significantly slowed down.3
:: With the arrival of spring and rising temperatures, disrupted access to WASH and damage to homes may increase the risk of vector-borne diseases such as West Nile fever and tick-borne encephalitis.
AMR
AMR Action Fund Announces First Investments in Adaptive Phage Therapeutics and Venatorx Pharmaceuticals
Deals mark an important step toward AMR Action Fund’s goal of bringing two to four new antibiotics to market to take on growing threat of drug-resistant bacteria
APRIL 4, 2022, BOSTON, MA –The AMR Action Fund, the world’s largest public-private partnership investing in biotech companies that are developing antibiotics, announced today that it has invested in Adaptive Phage Therapeutics (APT) and Venatorx Pharmaceuticals. The transactions mark the Fund’s first investments and are an important step toward its goals of bringing to market new treatments for priority pathogens identified by the World Health Organization (WHO) and U.S. Centers for Disease Control and Prevention.
“From inception, the AMR Action Fund has focused on identifying investments that will yield urgently needed treatments and catalyze long-term innovation to take on the growing threat of antimicrobial resistance (AMR), which now kills more people annually than HIV/AIDS or malaria,” said Bill Burns, Board Chair of the AMR Action Fund. “Welcoming Adaptive Phage Therapeutics and Venatorx Pharmaceuticals as our first portfolio companies demonstrates that we are well on our way to fulfilling this important mission.”…
“Adaptive Phage Therapeutics and Venatorx Pharmaceuticals are poised to change the treatment landscape for drug-resistant infections and deliver significant benefit to patients,” said AMR Action Fund CEO, Henry Skinner. “While this is a major milestone for the Fund, our work is just beginning. We plan to commit over $100 million in capital this year in companies developing clinically differentiated antimicrobials with the potential to treat the most urgent unmet clinical needs, and we will continue investing in promising biotechs in the years ahead to ensure that patients around the world have the treatments they need in the ever-evolving fight against superbugs. Our investments are substantial, but we alone are not enough to take on the global challenge of AMR. It is now imperative that policymakers around the world enact market reforms to support investment in these urgently needed medications.”
APT’s approach leverages an ever-growing library of systematically discovered, selected, catalogued, and curated bacteriophages (phages), naturally occurring viruses that infect and kill bacteria, which collectively provide broad coverage against many of the world’s highest priority antibiotic-resistant bacteria. Phages from APT’s phage bank are matched to treat patients’ infections through a proprietary susceptibility assay, and are being tested on a range of infections, including prosthetic joint infections, bone infections (osteomyelitis), and lung infections. The AMR Action Fund’s investment in APT was executed as an extension to a Series B round led by Deerfield Management Company…
::::::
IFPMA – Largest pharmaceutical industry-backed collective venture fund tackling antimicrobial resistance announces first portfolio investments
April 4, 2022
::::::
Coronavirus [COVID-19] – WHO
Public Health Emergency of International Concern (PHEIC)
https://www.who.int/emergencies/diseases/novel-coronavirus-2019
Weekly Epidemiological and Operational updates
Last update: 08 Apr 2022
Confirmed cases :: 494 587 638
Confirmed deaths :: 6 170 283
Vaccine doses administered: 11 250 782 214
::::::
Weekly epidemiological update on COVID-19 – 5 April 2022
Overview
After the increase observed during the first half of March 2022, the number of new COVID-19 cases has decreased for a second consecutive week, with a 16% decline during the week of 28 March through 3 April 2022 as compared to the previous week.
The number of new weekly deaths also decreased sharply (-43%) as compared to the previous week, during which an artificial spike in deaths was observed.
Across the six WHO regions, over nine million new cases and over 26 000 new deaths were reported, and all the regions show decreasing trends both in the number of new weekly cases and new weekly deaths.
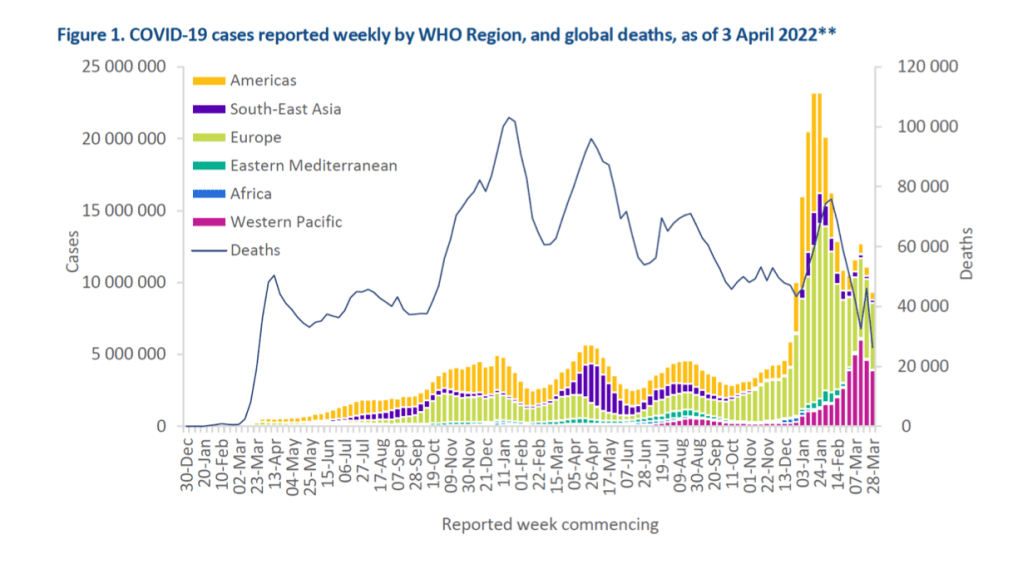
As of 3 April 2022, just over 489 million cases and over 6 million deaths have been reported globally.
In this edition, we provide an update on the geographic distribution of circulating SARS-CoV-2 variants of concern (VOCs), including the prevalence and summary of current evidence of the Omicron variant. We also provide updates on recombinants and vaccine effectiveness for the Delta and Omicron variants.
Over two-thirds of Africans exposed to virus which causes COVID-19: WHO study
07 April 2022
Brazzaville – Up to 65% of Africans have been infected by SARS-CoV-2, the virus which causes COVID-19, a World Health Organization (WHO) analysis finds. The study finds that true infections on the continent were 97 times larger than reported confirmed cases.
The analysis, which is available as a pre-print under peer review, synthesized 151 studies published on seroprevalence in Africa between January 2020 and December 2021. It found that exposure to SARS-CoV-2 skyrocketed from 3% (1.0-9.2% range) in June of 2020 to 65% (56.3-73% range) by September of 2021, or 800 million infections compared with 8.2 million cases reported at that time. The study showed that exposure to the virus rose sharply following the emergence of the Beta and the Delta variants.
The analysis revealed that the true number of infections could be as much as 97 times higher than the number of confirmed reported cases. This compares to the global average where true number of infections is 16 times higher than the number of confirmed reported cases.
However, seroprevalence varied widely within and across countries in Africa – higher in more dense urban areas than in less populated rural areas – and between age groups, with children aged 0-9 years having fewer infections compared with adults. Exposure to the virus also varied between countries and Africa’s sub-regions: seroprevalence appears to be highest in Eastern, Western and Central African regions.
The new analysis suggests that more than two-thirds of all Africans have been exposed to the COVID-19 virus. Globally seroprevalence studies have found a significant under-counting of cases occurring across the globe with 45.2% of the world’s population estimated to have been infected with the virus by September 2021. It is, however, difficult to compare figures for Africa with those of other regions, as many of the studies conducted cover different time periods.
The continent differentiates itself from other regions by its high number of asymptomatic cases, with 67% of cases having no symptoms.
“This analysis shows that current reported COVID-19 confirmed cases are only a fraction of the actual number of infections on the continent,” said Dr Matshidiso Moeti, WHO Regional Director for Africa. “This under-counting is occurring world-wide and it’s no surprise that the numbers are particularly large in Africa where there are so many cases with no symptoms.”…
Status of COVID-19 Vaccines within WHO EUL/PQ evaluation process 02 March 2022
[New additions; Full scale view available at title link above]
[Updated on 09 Apr 2022]

COVID Vaccines/Therapeutics – Developer/Manufacturer Announcements
[Selected press releases/announcements from organizations from WHO EUL/PQ listing above and other organizations]
AstraZeneca
Press Releases – No new digest announcements identified
Bharat Biotech
Press Releases – No new company announcements identified
Suspension of supply of COVID-19 vaccine (COVAXIN®)
2 April 2022
Today, WHO confirmed the suspension of supply of Covaxin (Bharat Biotech) through UN procurement agencies, and recommended that countries using the vaccine take action as appropriate.
The suspension is in response to the outcome of a WHO inspection on 14–22 March 2022, and the need to conduct process and facility upgrade to address recently identified deficiencies in good manufacturing practices (GMP).
Bharat Biotech has committed to addressing the GMP deficiencies and is developing a corrective and preventive action plan for submission to the Drugs Controller General of India (DCGI) and WHO. In the interim and as a precautionary measure, the company has indicated that it will suspend production of Covaxin for export. As a consequence, supply will be interrupted for the foreseeable future.
The risk assessment to date does not indicate change in the risk–benefit ratio. The data, available to WHO, indicate the vaccine is effective and no safety concerns exist…
BioCubaFarma – Cuba
Últimas Noticias – Website not leading at inquiry
Biological E
News
BE to Get mRNA Tech from WHO to Produce COVID-19 Vaccines
WHO selects BE as a recipient of mRNA technology
Biological E. Limited produces a number of critical & life-saving vaccines, including CORBEVAXTM
Hyderabad, April 4, 2022: Biological E. Limited (BE), one of the world’s largest vaccine manufacturers, has today announced that it has been selected as a recipient of mRNA technology from the World Health Organisation (WHO) technology transfer hub.
Biontech
Press Releases
BioNTech Granted Pandemic Preparedness Contract by German Federal Ministry of Health
MAINZ, Germany, April 8, 2022 — BioNTech SE (Nasdaq: BNTX) today announced that it is one of the companies in Germany to be granted a pandemic preparedness contract by the Federal Republic of Germany. The framework agreement is aimed at pandemic preparedness including manufacturing and supply of mRNA vaccines in emergency situations in Germany.
“There is growing evidence that viral pandemics will continue to pose a public health challenge for years. This contract with the German government will ensure significant supply of vaccine doses to address potential public health threats by 2027,” said Sean Marett, Chief Business and Chief Commercial Officer at BioNTech…
CanSinoBIO
News – Website not responding at inquiry
CIGB
Latest News – No new digest announcements identified
Cinagen
Recent News – No new digest announcements identified
Clover Biopharmaceuticals – China
News – No new digest announcements identified
Curevac [Bayer Ag – Germany]
News – No new digest announcements identified
Gamaleya National Center
Latest News and Events – See Russia below.
IMBCAMS, China
Home – Website not responding at inquiry
Janssen/JNJ
Press Releases
Apr 04, 2022 United States
World Health Organization Updated Emergency Use Listing Recommends Johnson & Johnson COVID-19 Vaccine for Booster Us
:: WHO supports vaccine for use as same-dose and ‘mix-and-match’ booster following a primary mRNA vaccine regimen
:: WHO also recommends extended shelf-life of up to 11 months for Johnson & Johnson COVID-19 vaccine when stored at 2 to 8 degrees Celsius (36 to 46 degrees Fahrenheit)
Medicago
Media – No new digest announcements identified
Moderna
Press Releases
07 April, 2022
IAVI and Moderna Partner to Tackle Broad Global Health Priorities Using mRNA for Vaccines and Antibodies
Nanogen
News – No new digest announcements identified
Novavax
Press Releases
Novavax and Serum Institute of India Receive Emergency Use Authorization for Novavax’ COVID-19 Vaccine in Thailand
Apr 8, 2022
Pfizer
Recent Press Releases
04.07.2022
Pfizer to Acquire ReViral and Its Respiratory Syncytial Virus Therapeutic Candidates
:: Proposed acquisition strengthens Pfizer’s capabilities in infectious disease research and development with a complementary strategy to help improve patient outcomes through treatment for respiratory syncytial virus (RSV) infections and prevent illness through vaccination
:: Expands Pfizer’s innovative anti-infective pipeline and utilizes the company’s R&D, manufacturing and commercialization expertise with the goal of addressing a significant unmet need for RSV treatments
R-Pharm
https://rpharm-us.com/index.php
[No news or media page identified]
Sanofi Pasteur
Press Releases – No new digest announcements identified
Serum Institute of India
NEWS & ANNOUNCEMENTS – No new digest announcements identified
Shifa Pharmed [Iran]
http://shafapharmed.com/
No news page identified.
Sinopharm/WIBPBIBP
News – No new digest announcements identified
Sinovac
Press Releases – No new digest announcements identified
Vector State Research Centre of Viralogy and Biotechnology
Home – No new digest announcements identified
WestVac Biopharma
Media – No new digest announcements identified
Zhifei Longcom, China
[Anhui Zhifei Longcom Biologic Pharmacy Co., Ltd.]
[No website identified]
::::::
GSK
Press releases for media – No new digest announcements identified
Merck
News releases
Expansion of Elkton, Virginia Manufacturing Facility to Further Increase Merck’s HPV Vaccine Supply and Support Broader and Equitable Access
April 04, 2022
KENILWORTH, N.J.–(BUSINESS WIRE)– Merck (NYSE: MRK), known as MSD outside the United States and Canada, reaffirmed its commitment to enable broad equitable access to the company’s HPV vaccines. To support this, the company has invested significantly in manufacturing, and recently expanded its vaccines manufacturing facility located in Elkton, VA, completing the construction of 120,000 square feet and adding 150 new jobs at the site to further increase capacity and supply of the company’s human papillomavirus (HPV) vaccines, following regulatory reviews and approvals…
Novartis
News – No new digest announcements identified
SK Biosciences
Press releases –
SK bioscience Submits Post Approval Change Application of Protein-Based COVID-19 Vaccine for Adolescent Authorization to KMFDS
2022. 04. 07
Valneva
Press Releases – No new digest announcements identified
UNICEF COVID-19 Vaccine Market Dashboard :: Agreements Table Accessed 09 Apr 2022
An overview of information collected from publicly announced bilateral and multilateral supply agreements [no new agreements since 10/22/2021 reported]

COVID-19 Global Targets and Progress Tracker – IMF
The COVID-19 Global Targets and Progress Tracker presents a consolidated view of the progress towards global COVID-19 targets, barriers in access to COVID-19 tools, and delivery of donor pledges.

The global targets presented in the Tracker are based on an alignment of the targets identified in the IMF Pandemic Proposal, ACT-A Strategic Plan & Budget, and the US-hosted Global C19 Summit, and as such have been reaffirmed by multilateral institutions and global leaders. We will continue to enhance the tracker as we improve our data collection efforts.
Global Dashboard on COVID-19 Vaccine Equity
The Dashboard is a joint initiative of UNDP, WHO and the University of Oxford with cooperation across the UN system, anchored in the SDG 3 Global Action Plan for Healthy Lives and Well-being for All.
Dashboard on Vaccine Equity [accessed 09 Apr 2022]: https://data.undp.org/vaccine-equity/
See also visualization on Vaccine Access and Vaccine Affordability

Global COVID-19 Access Tracker
https://www.covid19globaltracker.org/
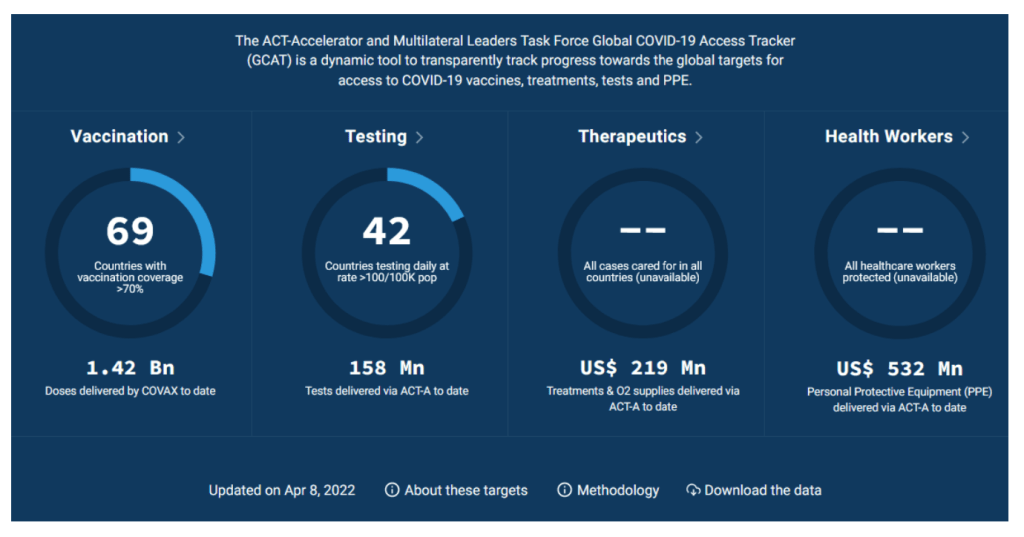
Duke – Launch and Scale Speedometer
The Race for Global COVID-19 Vaccine Equity
A flurry of nearly 200 COVID-19 vaccine candidates are moving forward through the development and clinical trials processes at unprecedented speed; more than ten candidates are already in Phase 3 large-scale trials and several have received emergency or limited authorization. Our team has aggregated and analyzed publicly available data to track the flow of procurement and manufacturing and better understand global equity challenges. We developed a data framework of relevant variables and conducted desk research of publicly available information to identify COVID vaccine candidates and status, deals and ongoing negotiations for procurement and manufacturing, COVID burden by country, and allocation and distribution plans. We have also conducted interviews with public officials in key countries to better understand the context and challenges facing vaccine allocation and distribution
[accessed 24 July 2021]
See our COVID Vaccine Purchases research
See our COVID Vaccine Manufacturing research
See our COVID Vaccine Donations & Exports research

COVID Vaccines – OCHA:: HDX
COVID-19 Data Explorer: Global Humanitarian Operations
COVID-19 Vaccine Roll-out
09 Apr 2022 | COVAX (WHO,GAVI,CEPI), UNDESA, Press Reports | DATA
Global COVID-19 Figures: 494M total confirmed cases; 6.2M total confirmed deaths
Global vaccines administered: 11.4B
Number of Countries: 28
COVAX Allocations Round 4-9 (Number of Doses): 170M
COVAX Delivered (Number of Doses): 280M
Other Delivered (Number of Doses): 260M
Total Delivered (Number of Doses): 540M
Total Administered (Number of Doses): 390M
Multilateral Leaders Task Force on COVID-19 [IMF, World Bank Group, WHO, WTO]
https://data.covid19taskforce.com/data
A global effort to help developing countries access and deliver COVID-19 vaccines, testing, and therapeutics, as they work to end the pandemic and boost economic recovery.
The International Monetary Fund, World Bank Group, World Health Organization and World Trade Organization have joined forces to accelerate access to COVID-19 vaccines, therapeutics and diagnostics by leveraging multilateral finance and trade solutions, particularly in low- and middle-income countries.
Website accessed 09 Apr 2022: https://data.covid19taskforce.com/data The global view below is complemented by country-specific dashboards here.

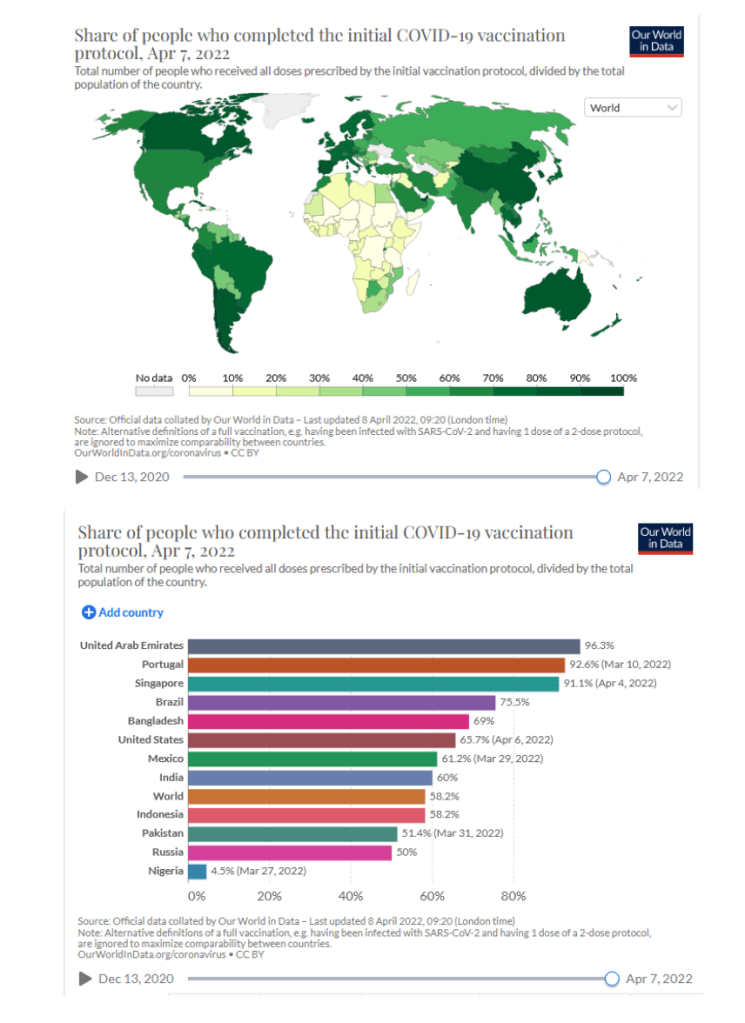
Our World in Data
Coronavirus (COVID-19) Vaccinations [Accessed 09 Apr 2022]
64.7% of the world population has received at least one dose of a COVID-19 vaccine.
11.37 billion doses have been administered globally, and 13.32 million are now administered each day.
Only 14.8% of people in low-income countries have received at least one dose.
U.S.: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
HHS
News
Remarks by HHS Secretary Becerra during a joint press conference on World Health Day with the World Health Organization Director-General Dr. Tedros Adhanom Ghebreyesus
April 7, 2022 | Speech
Remarks by Secretary Xavier Becerra at White House COVID-19 Briefing on New Federal Plan to Accelerate Support for People Experiencing Long-Term Effects of COVID-19
April 5, 2022 | Speech
BARDA – U.S. Department of HHS [to 09 Apr 2022]
https://aspr.hhs.gov/newsroom/Pages/NewsRoomHome.aspx
News
No new digest content identified.
::::::
FDA
Press Announcements
No new digest content identified.
Regulatory Actions
Emergency Use Authorization for Vaccines to Prevent COVID-19 Guidance for Industry – March 2022
Final – March 31, 2022
Docket Number: FDA-2020-D-1137
Issued by: Center for Biologics Evaluation and Research
PDF: https://www.fda.gov/media/142749/download
FDA plays a critical role in protecting the United States (U.S.) from threats such as emerging infectious diseases, including the Coronavirus Disease 2019 (COVID-19) pandemic. FDA is committed to providing timely guidance to support response efforts to this pandemic.
FDA is issuing this guidance to provide sponsors of requests for Emergency Use Authorization (EUA) for COVID-19 vaccines with recommendations regarding the data and information needed to support the issuance of an EUA under section 564 of the FD&C Act (21 U.S.C. 360bbb-3) for an investigational vaccine to prevent COVID-19 for the duration of the COVID-19 public health emergency. This document supersedes the guidance of the same title issued in May 2021 (which superseded the guidance of the same title issued October 2020 and reissued February 2021).
Vaccines and Related Biological Products Advisory Committee– FDA
https://www.fda.gov/advisory-committees/blood-vaccines-and-other-biologics/vaccines-and-related-biological-products-advisory-committee
Calendar
Vaccines and Related Biological Products Advisory Committee Meeting April 6, 2022 Announcement – 04/06/2022
The committee met in open session to discuss considerations for use of COVID-19 vaccine booster doses and the process for COVID-19 vaccine strain selection to address current and emerging variants.
[No meeting materials posted as of 09 Apr 2022]
::::::
White House [U.S.] [to 09 Apr 2022]
Briefing Room – Selected Major COVID Announcements
Statement by White House COVID-19 Response Coordinator Jeff Zients on American Lives Saved by the COVID-19 Vaccination Program
April 08, 2022 • Statements and Releases
A new study out today from the Commonwealth Fund shows that President Biden’s relentless efforts to get Americans vaccinated saved millions of American lives. Our vaccination campaign saved 2.2 million American lives, prevented 17 million hospitalizations, prevented 66 million COVID-19 cases, and avoided $900 billion in health care costs….
Statement from President Joe Biden for World Health Day 2022
April 07, 2022 • Statements and Releases
FACT SHEET: The Biden Administration’s Commitment to Global Health in the FY 2023 President’s Budget
April 07, 2022 • Statements and Releases
Press Briefing by White House COVID-19 Response Team and HHS Public Health Officials | April 5, 2022
April 05, 2022 • Press Briefings
Statement by Press Secretary Jen Psaki on COVID-19 Funding Proposal
April 05, 2022 • Statements and Releases
Memorandum on Addressing the Long-Term Effects of COVID-19
April 05, 2022 • Presidential Actions
FACT SHEET: The Biden Administration Accelerates Whole-of-Government Effort to Prevent, Detect, and Treat Long COVID
April 05, 2022 • Statements and Releases
U.S. Department of State [to 09 Apr 2022]
https://www.state.gov/coronavirus/releases/
Press Statement – COVID Context
No new digest content identified.
USAID [to 09 Apr 2022]
https://www.usaid.gov/news-information/press-releases
Selected Press Releases, Statements, Announcements
On the Urgent Need for Additional Global COVID-19 Resources
April 4, 2022
Statement by Administrator Samantha Power
Throughout the COVID-19 pandemic, Congress has consistently come together in an inspiring show of bipartisan support to fund our response. The $10 billion agreement announced this afternoon, while helping to fund our immediate domestic needs like treatments and tests, unfortunately leaves out the resources we need to power our global COVID response—an effort that is critical to preventing the emergence of new deadly variants and moving the world beyond this pandemic.
Europe: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
European Medicines Agency
News & Press Releases
Press Releases
News: ECDC and EMA issue advice on fourth doses of mRNA COVID-19 vaccines (new)
Last updated: 06/04/2022
The European Centre for Disease Prevention and Control (ECDC) and EMA’s COVID-19 task force (ETF) have concluded that it is too early to consider using a fourth dose of mRNA COVID-19 vaccines (Pfizer’s Comirnaty and Moderna’s Spikevax) in the general population.
However, both agencies agreed that a fourth dose (or second booster) can be given to adults 80 years of age and above after reviewing data on the higher risk of severe COVID-19 in this age group and the protection provided by a fourth dose.
ECDC and EMA also noted that there is currently no clear evidence in the EU that vaccine protection against severe disease is waning substantially in adults with normal immune systems aged 60 to 79 years and thus no clear evidence to support the immediate use of a fourth dose.
Authorities will continue to monitor data to determine if there is an increasing risk of severe illness among those who are vaccinated…
::::::
European Centre for Disease Prevention and Control
https://www.ecdc.europa.eu/en
Latest Updates [Selected]
[No new digest content identified]
::::::
Accessed 09 Apr 2022
https://vaccinetracker.ecdc.europa.eu/public/extensions/COVID-19/vaccine-tracker.html#uptake-tab

::::::
European Commission
https://ec.europa.eu/commission/presscorner/home/en
Latest [Selected]
Press release 9 April 2022
Stand Up For Ukraine: 9.1 billion euros pledged in support of internally displaced and refugees
The “Stand Up for Ukraine” global pledging event and campaign has raised 9.1 billion euros for people fleeing the Russian invasion, inside Ukraine and abroad, including €1 billion from the European Commission.
Press release 8 April 2022
Ukraine: EU agrees fifth package of restrictive measures against Russia
The European Commission welcomes today’s agreement by the Council to adopt a fifth package of restrictive measures against Putin’s regime in response to its brutal aggression against Ukraine and its people.
Press release 6 April 2022
Food crisis: the EU takes action to support Africa’s Sahel and Lake Chad regions
With the aggravation of the food security and nutrition due to Russia’s invasion of Ukraine, the EU is today reinforcing its political and financial commitment to partner countries in Africa. A total of €554 million in 2022 will be targeted at increasing food security in Sahel and Lake Chad.
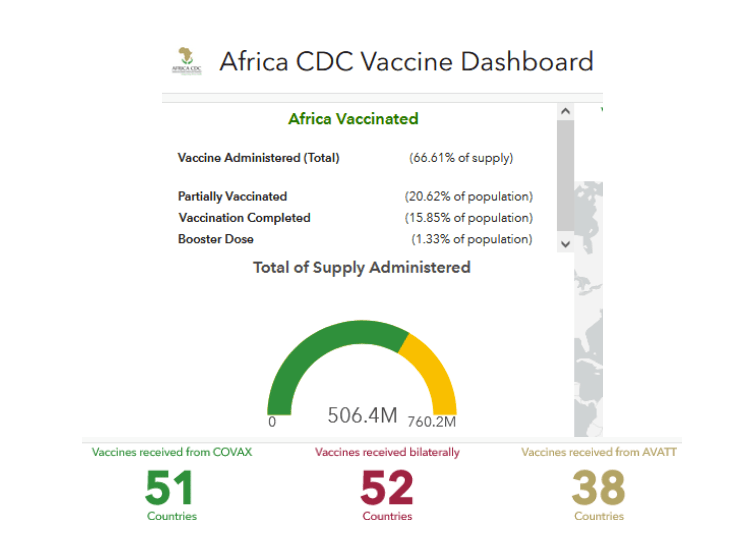
Africa: COVID-19 – Announcements/Regulatory Actions/Deployment
https://africacdc.org/covid-19-vaccination/
Russia: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
Russia: Sputnik V – “the first registered COVID-19 vaccine”
https://sputnikvaccine.com/newsroom/pressreleases/
Press Releases
Latest Updates [Selected]
[No new digest content identified]
India: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment

Ministry of Health and Family Welfare
https://www.mohfw.gov.in/
Government of India – Press Information Bureau
Latest Press Releases
COVID – 19 Vaccination Update – Day 449
:: India’s cumulative vaccination coverage crosses 185.68 Crore
:: More than 12 lakh Vaccine doses administered today till 7 pm
:: More than 2.21 Crore Vaccine Doses administered to 12-14 age group so far
Posted On: 09 APR 2022 8:14PM by PIB Delhi
Indian Council for Medical Research (ICMR)
https://www.icmr.gov.in/media.html
Press Releases
No new digest content identified.
China: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
[We did not identify official announcements about Shanghai confirming the reports below; See China CDC below for additional announcements]
Shanghai widens COVID testing as other Chinese cities impose curbs
Reuters
April 8, 2022
Most large Chinese cities keeping COVID restrictions in place
Food supply issues persist in the locked-down city
Senior epidemiologist calls for “thunderous” action to cut transmission
SHANGHAI, April 8 (Reuters) – Shanghai on Friday announced a record 21,000 new cases and a third consecutive day of COVID testing as a lockdown of its 26 million people showed no sign of easing and other Chinese cities tightened curbs – even in places with no recent infections.
Beijing authorities intervened in Shanghai after its failure to isolate COVID by locking the city down in stages, and insists that the country stick to its zero-tolerance policy to prevent its medical system from breaking down.
Authorities across China, which have mostly managed to keep COVID at bay for the last two years, are stepping up coronavirus control measures, including movement restrictions, mass testing and new quarantine centres.
Cities that sprang into action this week include Zhengzhou, in central Henan province, which on Thursday said it would test all 12.6 million residents after finding a few asymptomatic cases…
Many Shanghai residents say Covid lockdown measures have caused food shortages.
The authorities have locked down the city of 26 million in a bid to contain China’s worst outbreak since the pandemic began.
New York Times, April 8, 2022
By Joy Dong

Emergencies
POLIO
Public Health Emergency of International Concern (PHEIC)
https://polioeradication.org/polio-today/polio-now/this-week/
Polio this week as of 06 April 2022
:: The GPEI has released an updated draft version of the Standard Operating Procedures (SOPs) to offer guidance to any country that detects any type of poliovirus outbreak or event, to respond in a timely and effective manner. This version of the SOPs builds on the prior versions developed since 2015 and takes into account the key developments, lessons learnt and availability of new tools since
:: This week, the Strategic Advisory Group of Experts on immunization (SAGE) is convening in Geneva, Switzerland. On polio eradication, the group will review the global current epidemiology, updated data on novel oral polio vaccine type 2 (nOPV2) use, and recommendations on initial planning for OPV cessation.
:: On 26 April, the programme will launch the new investment case for the GPEI’s Strategy: Delivering on a Promise. This one-hour, virtual event, “Investing in the Promise of a Polio-free World”, will feature live remarks from WHO Director-General Dr Tedros Adhanom Ghebreyesus and UNICEF Executive Director Catherine Russell, as well as the stories of health workers and Ministers of Health working with the polio programme around the world. Please register here to learn more about the financial and political support needed to end polio for good.
Summary of new WPV and cVDPV viruses this week (AFP cases and ES positives):
– DR Congo: seven cVDPV2 cases
– Egypt: one cVDPV2 positive environmental sample
– Nigeria: six cVDPV2 cases and seven positive environmental samples
::::::
::::::
Disease Outbreak News (DONs)
Latest WHO Disease Outbreak News (DONs), providing information on confirmed acute public health events or potential events of concern.
7 April 2022 | Middle East respiratory syndrome coronavirus (MERS-CoV) – Saudi Arabia
Summary
This is the bi-annual update on Middle East Respiratory Syndrome Coronavirus (MERS-CoV) infections reported to WHO from the Kingdom of Saudi Arabia. Between 1 August 2021 to 28 February 2022, six additional cases of MERS-CoV infections, including four associated deaths were reported to WHO from the Kingdom of Saudi Arabia. These cases were reported from Riyadh (four cases), Eastern (one case), and Taif (one case) regions. Based on the current situation and available information, WHO re-emphasizes the importance of strong surveillance by all Member States for acute respiratory infections, including MERS-CoV, and careful review of any unusual patterns.
::::::
::::::
WHO/OCHA Emergencies
Health emergencies list – WHO
“The health emergencies list details the disease outbreaks, disasters and humanitarian crises where WHO plays an essential role in supporting countries to respond to and recover from emergencies with public health consequences.”
Afghanistan crisis [Last apparent update: 29 March 2022]
Crisis in Northern Ethiopia [Last apparent update: 1 June 2021]
Ebola outbreak, Democratic Republic of the Congo, 2021 [Last apparent update: 17 Aug 2021]
Ebola outbreak outbreak, N’Zerekore, Guinea, 2021 [Last apparent update: 17 Aug 2021]
Coronavirus disease (COVID-19) pandemic [See COVID above]
Ebola outbreak, Equateur Province, Democratic Republic of the Congo, 2020
[Last apparent update: 17 Aug 2021]
Ebola outbreak, North Kivu, Ituri, Democratic Republic of the Congo, 2018 – 2020
[Last apparent update: 17 Aug 2021]
Ebola outbreak, Democratic Republic of the Congo, 2018 [Last apparent update: 24 July 2018]
Yemen crisis [Last apparent update: 12 February 2021]
Syria crisis [Last apparent update: 18 June 2021]
Somalia crisis [Last apparent update: 24 March 2018]
Nigeria crisis [Last apparent update: 1 Oct 2021]
Ebola outbreak, Democratic Republic of the Congo, 2017 [Last apparent update: 17 Aug 2021]
Zika virus disease outbreak, 2015-2016 [Last apparent update: 24 Jan 2020]
Ebola outbreak: West Africa, 2014-2016 [Last apparent update: 17 Aug 2021]
Iraq crisis [Last apparent update: 9 Jan 2008]
South Sudan crisis [Last apparent update: 23 Sep 2020]
Avian influenza A (H7N9) virus outbreak [Last apparent update: 13 September 2021]
Middle East respiratory syndrome (MERS-CoV) outbreak [Last apparent update: 8 July 2019]
Influenza A (H1N1) virus, 2009-2010 pandemic [Last apparent update: 10 Aug 2010]
::::::
UN OCHA – Current Emergencies
Current Corporate Emergencies
Afghanistan
No new updates identified.
Northern Ethiopia
Ethiopia – Northern Ethiopia Humanitarian Update Situation Report, 7 Apr 2022
HIGHLIGHTS
Only 8 per cent of cash needs transferred to Tigray for humanitarian operations since 12 July 2021.
To date, about 15,000 people have been relocated to Sabacare 4 displacement site in Mekelle, reaching its full capacity.
245 schools in Afar remain closed, or 20 percent of the schools in the region, forcing more than 53,000 students out of school. Additional 5 schools are hosting displaced people.
An additional 10 mobile health and nutrition teams were deployed to Amhara to scale-up the response, bringing the total number of teams operating in the region to 88.
30 mobile health and nutrition teams are operating in conflict affected areas in Afar, reaching more than 8,000 people with health care during the week.
Ukraine
Ukraine: Humanitarian Impact Situation Report (As of 12:00 p.m. (EET) on 8 April 2022)
::::::
::::::
WHO & Regional Offices [to 09 Apr 2022]
https://www.who.int/news
News [Selected]
7 April 2022
Departmental news
Open to all, anytime, anywhere: Free online courses bring vaccine knowledge to the global community
6 April 2022
News release
WHO urges accelerated action to protect human health and combat the climate crisis at a time of heightened conflict and fragility
4 April 2022
Departmental news
WHO announces updates on new TB antigen-based skin tests for the diagnosis of TB infection
4 April 2022
News release
Billions of people still breathe unhealthy air: new WHO data
4 April 2022
Departmental news
Health and the environment
2 April 2022
Departmental news
Suspension of supply of COVID-19 vaccine (COVAXIN®)
Today, WHO confirmed the suspension of supply of Covaxin (Bharat Biotech) through UN procurement agencies, and recommended that countries using the vaccine take action as appropriate.
The suspension is in response to the outcome of a WHO inspection on 14–22 March 2022, and the need to conduct process and facility upgrade to address recently identified deficiencies in good manufacturing practices (GMP).
Bharat Biotech has committed to addressing the GMP deficiencies and is developing a corrective and preventive action plan for submission to the Drugs Controller General of India (DCGI) and WHO. In the interim and as a precautionary measure, the company has indicated that it will suspend production of Covaxin for export. As a consequence, supply will be interrupted for the foreseeable future.
The risk assessment to date does not indicate change in the risk–benefit ratio. The data, available to WHO, indicate the vaccine is effective and no safety concerns exist…
1 April 2022
Departmental news
Parliaments promote women’s, children’s and adolescents’ health in the time of COVID-19
::::::
WHO Director General Speeches [selected]
https://www.who.int/director-general/speeches
Selected
7 April 2022
Speech
WHO Director-General’s opening remarks at the World Health Day press briefing – 7 April 2022
::::::
WHO Regional Offices
Selected Press Releases, Announcements
WHO African Region
:: 08 April 2022 Managing COVID-19 waste in Africa
:: 07 April 2022 Over two-thirds of Africans exposed to virus which causes COVID-19: WHO study
:: 06 April 2022 Africa faces rising climate-linked health emergencies
:: 01 April 2022 Major antibiotic donation moves African countries closer to defeating yaws
WHO Region of the Americas PAHO
:: On World Health Day, PAHO Director calls for concerted action to safeguard the health of the planet and its people
Washington, D.C., April 7, 2021 (PAHO) – After two years of the COVID-19 pandemic with millions of lives lost to the virus, Pan American Health Organization Director (PAHO) Carissa F. Etienne, marked World Health Day by calling for the urgent protection of the environment and highlighted how the health of the planet is linked to our own. “The so-…
:: World Health Day 2022
World Health Day is celebrated every year on 7 April to commemorate the anniversary of the founding of the World Health Organization (WHO) in 1948. Each year for this date, a theme is chosen that highlights an area of priority concern for WHO. In the face of the current pandemic, a polluted planet, and an increasing incidence of diseases,…
:: Americas at risk of further COVID-19 wave as cases surge in other regions, PAHO Director warns
Vaccination campaigns must focus on protecting the most vulnerable. Washington D.C. 6 April 2022 (PAHO) – As cases of COVID-19 surge in Europe and East Asia due to Omicron BA.2, the Pan American Health Organization (PAHO) Director Carissa F. Etienne has urged countries to remain vigilant and urgently close vaccination gaps. While COVID cases…
WHO South-East Asia Region SEARO
No new digest content identified.
WHO European Region EURO
:: WHO records 100th attack on health care in Ukraine 08-04-2022
:: In solidarity with the health workers of Ukraine this World Health Day 07-04-2022
:: Health on Earth: a healthy planet benefits us all – World Health Day 2022 07-04-2022
:: Ukraine: Crisis averted for now – WHO, PEPFAR and partners ensure supply of antiretroviral drugs for people living with HIV 06-04-2022
:: WHO Regional Director for Europe visits Kazakhstan, strengthens primary health care collaboration 06-04-2022
WHO Eastern Mediterranean Region EMRO
No new digest content identified.
WHO Western Pacific Region
:: 7 April 2022 Countries in the Western Pacific on the frontlines of the climate crisis, says WHO
:: 7 April 2022 Regional Director’s remarks at the virtual press conference on 7 April 2022
::::::
WHO Events
https://www.who.int/news-room/events/1
[Selected]
Public hearings regarding a new international instrument on pandemic preparedness and response
12 – 13 April 2022
In a consensus decision aimed at protecting the world from future infectious diseases crises, in December 2021 the World Health Assembly agreed to kickstart a global process by establishing an intergovernmental negotiating body (INB) to draft and negotiate a convention, agreement or other international instrument under the Constitution of the World Health Organization to strengthen pandemic prevention, preparedness and response.
Dr Tedros Adhanom Ghebreyesus, WHO Director-General, said the decision by the World Health Assembly was historic in nature, vital in its mission, and represented a once-in-a-generation opportunity to strengthen the global health architecture to protect and promote the well-being of all people.
As part of this historic decision, the World Health Assembly requested the Director-General to hold public hearings, in line with standard WHO practice, to support the work of the INB.
Registration and additional information on the public hearings is available at https://inb.who.int
::::::
New WHO Publications
https://www.who.int/publications/i
Selected Titles
7 April 2022
Report of the first meeting of the WHO Technical Advisory Group on Diabetes: virtual meeting, 29-30 September…
7 April 2022
Emergency in Ukraine: external situation report #6, published 7 April 2022: reporting period: 31 March–6…
6 April 2022
Sharing and reuse of health-related data for research purposes: WHO policy and implementation guidance
Overview
The purpose of this document is to clarify the policy and practice on the reuse and onward sharing for research purposes of health data collected under the auspices of WHO technical programmes. This covers use in both emergency and non-emergency situations and complements the following: the Policy on use and sharing of data collected in Member States by the World Health Organization (WHO) outside the context of public health emergencies; the Policy statement on data sharing by the World Health Organization in the context of public health emergencies; and the Joint statement on public disclosure of results from clinical trials. This policy covers the reuse of health data for research purposes. Its scope includes research data generated by research undertaken directly by WHO, or funded by WHO, as well as the use of other health data for research purposes.
This document sets out the objectives of this WHO policy and the obligations of WHO staff and researchers funded by WHO. The following section of this document entitled Guidance on the implementation of the WHO policy on the sharing and reuse of health-related data for research purposes provides further references and resources to assist in the development of a data management and sharing plan that is in alignment with the vision of this policy.
6 April 2022
Clinical care of severe acute respiratory infections – Tool kit
6 April 2022
Strategic framework for collaboration on antimicrobial resistance
5 April 2022
Injection safety in the context of coronavirus disease (COVID-19) vaccination: Addendum to policy brief,…
4 April 2022
Stakeholder consultative meeting on prevention and management of childhood pneumonia and diarrhoea: report,…
4 April 2022
Rapid communication: TB antigen-based skin tests for the diagnosis of TB infection
4 April 2022
WHO Drug Information – Volume 36, No. 1
3 April 2022
HIV module: Global competency and outcomes framework for universal health coverage
3 April 2022
Global competency framework for universal health coverage
3 April 2022
Global Competency and Outcomes Framework for Universal Health Coverage
1 April 2022
Compendium of WHO and other UN guidance on health and environment, 2022 update
CDC/ACIP [U.S.] [to 09 Apr 2022]
http://www.cdc.gov/media/index.html
https://www.cdc.gov/vaccines/acip/index.html
Latest News Releases, Announcements [Selected]
No new digest content identified.
ACIP Meetings
Next ACIP Meeting
April 20, 2022 11:00am – 4:00pm ET.
This meeting is virtual. No registration is required to watch the webcasts.
Webcast Link
No Agenda posted at inquiry
MMWR Weekly: Current Volume (71) April 8, 2022 / No. 14
PDF of this issue
Cardiac Complications After SARS-CoV-2 Infection and mRNA COVID-19 Vaccination — PCORnet, United States, January 2021–January 2022
Notes From the Field: SARS-CoV-2 Omicron Variant Infection in 10 Persons Within 90 Days of Previous SARS-CoV-2 Delta Variant Infection — Four States, October 2021–January 2022
Coronavirus Disease 2019 (COVID-19)– CDC
Approximately 25 announcements/reports/data summaries.
:: 4/8/22 EARLY RELEASE: COVID-19 Mortality and Vaccine Coverage — Hong Kong Special Administrative Region, China, January 6, 2022–March 21, 2022
:: 4/8/22 Overall US COVID-19 Vaccine Distribution and Administration Update as of Fri, 08 Apr 2022 06:00:00 EST
Africa CDC [to 09 Apr 2022]
http://www.africacdc.org/
News
Fifteen “EPI-Biostat Fellows” commence Master’s training at the Kinshasa School of Public Health (KSPH)
8 April 2022
Editor’s Note:
Careful readers will note that the number and range of organizations now monitored in our Announcements section below has grown as the impacts of the pandemic have spread across global economies, supply chains and programmatic activity of multilateral agencies and INGOs.
Airfinity [to 09 Apr 2022]
https://www.airfinity.com/insights
INSIGHTS & COMPANY NEWS
No new digest content identified.
Paul G. Allen Frontiers Group [to 09 Apr 2022]
https://alleninstitute.org/news-press/
News
No new digest content identified.
BMGF – Gates Foundation [to 09 Apr 2022]
https://www.gatesfoundation.org/ideas/media-center
Press Releases and Statements
No new digest content identified.
Bill & Melinda Gates Medical Research Institute [to 09 Apr 2022]
The Bill & Melinda Gates Medical Research Institute is a non-profit biotech organization. Our mission is to develop products to fight malaria, tuberculosis, and diarrheal diseases—three major causes of mortality, poverty, and inequality in developing countries. The world has unprecedented scientific tools at its disposal; now is the time to use them to save the lives of the world’s poorest people
No new digest content identified.
News
No new digest content identified.
Center for Vaccine Ethics and Policy – GE2P2 Global Foundation [to 09 Apr 2022]
https://centerforvaccineethicsandpolicy.net/
:: Past weekly editions and posting of all segments of Vaccines and Global Health: The Week in Review are available here.
:: Informed Consent: A Monthly Review – April 2022is now posted here
:: Patients, Politics, Pressure :: Pharma Stays Steady [Mostly] In Russia Sanctions Context
Observations at 30 March 2022 David R Curry
CEPI – Coalition for Epidemic Preparedness Innovations [to 09 Apr 2022]
CEPI invites vaccine developers and manufacturers to join global outbreak response network
CEPI funding will support trial of UB-612 vaccine candidate as a ‘mix-and-match’ booster dose.
06 Apr 2022
DARPA – Defense Advanced Research Projects Agency [U.S.] [to 09 Apr 2022
News
No new digest content identified.
Duke Global Health Innovation Center [to 09 Apr 2022]
No new digest content identified.
The European & Developing Countries Clinical Trials Partnership (EDCTP) aims to accelerate the development of new or improved drugs, vaccines, microbicides and diagnostics against HIV/AIDS, tuberculosis and malaria as well as other poverty-related and neglected infectious diseases in sub-Saharan Africa, with a focus on phase II and III clinical trials
News
07 April 2022
07 April 2022
Emory Vaccine Center [to 09 Apr 2022]
http://www.vaccines.emory.edu/
Vaccine Center News
No new digest content identified.
European Vaccine Initiative [to 09 Apr 2022]
Latest News, Events
No new digest content identified.
Evidence Aid [to 09 Apr 2022]
Evidence Aid aims to save lives and livelihoods in disasters by providing decision-makers with the best available evidence and by championing its use.
No new digest content identified.
Fondation Merieux [to 09 Apr 2022]
http://www.fondation-merieux.org/
No new digest content identified.
News Releases
8 April 2022
World leaders commit US$ 4.8 billion to help Break COVID Now
[See COVID above for detail]
7 April 2022
Nepal introduces Typhoid vaccine into routine immunisation across the country
:: The Ministry of Health and Population, Gavi, the Vaccine Alliance, UNICEF, WHO and other partners are collaborating on the nationwide typhoid vaccine introduction including a catch-up campaign, which seeks to vaccinate all children from the age of 15 months to 15 years to protect them from typhoid fever.
:: The campaign aims to reach 95% coverage of nearly 7.5 million children with the typhoid conjugate vaccine (TCV) and simultaneously identify ‘zero-dose’ children and those that have missed other routine vaccines.
7 April 2022
New Gavi Risk Sharing Partnership with MedAccess and the Open Society Foundations to help meet country demand for COVID-19 vaccines
:: Gavi, MedAccess and the Open Society Foundations announce a new partnership to create an innovative Risk Sharing Facility to support the COVAX Cost-Sharing Mechanism.
:: The partnership aims to build on intensive efforts by Gavi to extend its suite of innovative financing instruments to help donors and countries stretch their available resources as the pandemic shifts and to protect against future shocks. The Facility’s instruments also represent a pathway and toolkit to address future global health crises.
:: This new partnership comes as Gavi seeks to raise additional funds to support COVAX. April 8 will see Germany, Indonesia and Senegal co-host the 2022 Gavi COVAX AMC Summit, where governments, international organisations, civil society and the private sector will come together to renew their commitment to global vaccination.
6 April 2022
Indonesia and Senegal to co-host 2022 Gavi COVAX AMC Summit, together with Germany
https://www.ghitfund.org/newsroom/press
Press Releases
No new digest content identified.
https://www.theglobalfund.org/en/news/
News & Stories
No new digest content identified.
Global Research Collaboration for Infectious Disease Preparedness [GloPID-R] [to 09 Apr 2022]
News
No new digest content identified
Hilleman Laboratories [to 09 Apr 2022]
News & Insights
No new digest content identified
HHMI – Howard Hughes Medical Institute [to 09 Apr 2022]
Press Room
No new digest content identified.
Human Vaccines Project [to 09 Apr 2022]
http://www.humanvaccinesproject.org/
News
No new digest content identified.
IAVI [to 09 Apr 2022]
Collaboration to target bacterial and viral pathogens including HIV, SARS-CoV-2, antimicrobial-resistant pathogens, and tuberculosis
NEW YORK, NY & CAMBRIDGE, MA — APRIL 7, 2022 — Moderna, Inc. (Nasdaq: MRNA), a biotechnology company pioneering messenger RNA (mRNA) therapeutics and vaccines, and the nonprofit scientific research organization IAVI today announced a new collaboration to employ mRNA technology to meet the challenge of a range of global health threats. These diseases — HIV/AIDS, tuberculosis (TB), antimicrobial-resistant enteric infections, and COVID-19 — are estimated to have caused at least 95 million new infections and more than 4 million deaths in 2020 alone. The collaboration combines the power of Moderna’s mRNA platform and IAVI’s expertise in discovery and product development to advance vaccines and antibodies designed to be globally accessible, especially in low-income countries where the targeted diseases have high incidence and prevalence….
ICRC [to 09 Apr 2022]
https://www.icrc.org/en/whats-new
What’s New [Selected]
The use of force in law enforcement operations
States often face situations in which their officials have to use force to maintain or restore public security, law and order in armed conflicts or situations of violence that do not meet the threshold of applicability of international humanitarian l
06-04-2022 | Legal factsheet
What is international humanitarian law?
International humanitarian law (IHL) is a set of rules that seeks, for humanitarian reasons, to limit the effects of armed conflict.
06-04-2022 | Legal factsheet
International Coalition of Medicines Regulatory Authorities [ICMRA]
http://www.icmra.info/drupal/en/news
Selected Statements, Press Releases, Research
No new digest content identified.
IFFIm
Press Releases/Announcements
IFFIm “essential” to continued financing for the COVAX AMC
05 Apr 2022
… IFFIm’s established, in-demand vaccine bonds have already accelerated funding from donors to aid the fight against COVID-19. So far in this pandemic, over US$ 1.23 billion in new pledges have been committed to IFFIm for the COVAX AMC. IFFIm has frontloaded US$ 975 raised on capital markets backed by those donations in 2021.
…By harnessing its frontloading capability, IFFIm can be a valuable financial tool helping Gavi and donors to respond as efficiently and effectively as possible to upcoming global health challenges without any immediate impact on donors’ fiscal or official development assistance (ODA) positions.
“IFFIm has a crucial capability – bringing pledged money forward in time and putting it to work immediately. IFFIm was created 15 years ago to accelerate childhood vaccination, and was hugely successful at it. Now it’s an essential part of funding the fight to break COVID,” said Ken Lay, IFFIm Board Chair. “Working with Gavi and the World Bank, we expect it can do even more.”
IFRC [to 09 Apr 2022]
http://media.ifrc.org/ifrc/news/press-releases/
People affected by crises everywhere deserve same strong support provided to Ukraine response
Geneva, 6 April 2022 – The International Federation of Red Cross and Red Crescent Societies (IFRC) calls for solidarity with people affected by crises everywhere as the response to the conflict in Ukraine shows the way.
The support from people and donors around the world demonstrates what is possible and illustrates the good practice essential in any humanitarian crisis. At the same time, the massive differences depending on where a crisis occurs and who it affects have been thrown into stark relief…
Institut Pasteur [to 09 Apr 2022]
https://www.pasteur.fr/en/press-area
No new digest content identified.
IOM / International Organization for Migration [to 09 Apr 2022]
http://www.iom.int/press-room/press-releases
News – Selected
News
08 Apr 2022
15 Million People Face Humanitarian Crisis Due to Drought in the Horn of Africa
News
05 Apr 2022
7.1 Million People Displaced by the War in Ukraine: IOM Survey
ISC / International Science Council [to 09 Apr 2022]
https://council.science/current/
ISC is a non-governmental organization with a unique global membership that brings together 40 international scientific Unions and Associations and over 140 national and regional scientific organizations including Academies and Research Councils.
No new digest content identified.
IVAC [to 09 Apr 2022]
World Immunization Week 2022: Webinars
April 2022
During World Immunization Week 2022 the International Vaccine Access Center, along with global immunization and indigenous health partners, will host a three webinars. Registration is free, and we encourage all to sign up and save these dates on their calendars. Enabling Equitable Access to COVID-19 Vaccines for Indigenous Populations When: April 26, 2022 from 6:00 […]
IVI [to 09 Apr 2022]
No new digest content identified.
Johns Hopkins Center for Health Security [to 09 Apr 2022]
https://www.centerforhealthsecurity.org/news/center-news/
Center News
Johns Hopkins Center for Health Security Co-Hosts United States–India Strategic Dialogue on Biosecurity to Discuss Lessons from the Second Year of National Pandemic Responses
April 7, 2022
MSF/Médecins Sans Frontières [to 09 Apr 2022]
Latest [Selected Announcements]
No new digest content identified.
National Academy of Medicine – USA [to 09 Apr 2022]
News
No new digest content identified.
National Academy of Sciences – USA [to 09 Apr 2022]
http://www.nasonline.org/news-and-multimedia/
News
No new digest content identified.
National Vaccine Program Office – U.S. HHS [to 09 Apr 2022]
https://www.hhs.gov/vaccines/about/index.html
Upcoming Meetings/Latest Updates
No new digest content identified.
http://www.nih.gov/news-events/news-releases
No new digest content identified.
OECD [to 09 Apr 2022]
http://www.oecd.org/newsroom/publicationsdocuments/bydate/
Newsroom/Library
No new digest content identified.
PATH [to 09 Apr 2022]
https://www.path.org/media-center/
Press Releases
No new digest content identified.
Sabin Vaccine Institute [to 09 Apr 2022]
http://www.sabin.org/updates/pressreleases
Statements and Press Releases
No new digest content identified.
UNAIDS [to 09 Apr 2022]
Selected Press Releases/Reports/Statements
7 April 2022
7 April 2022
Urgently needed HIV services are supporting Ukrainian refugees in the Republic of Moldova
6 April 2022
United Kingdom parliamentary group visits UNAIDS to strengthen collaboration
6 April 2022
Young people’s monitoring of progress towards 2025 targets begins second phase
UNHCR Office of the United Nations High Commissioner for Refugees [to 09 Apr 2022]
http://www.unhcr.org/en-us/media-centre.htmlS
Selected News Releases, Announcements
UNHCR redoubles its aid inside Ukraine and the region
This is a summary of what was said by UNHCR spokesperson Matthew Saltmarsh – to whom quoted text may be attributed – at today’s press briefing at the Palais des Nations in Geneva.
08 April 2022
https://www.unicef.org/media/press-releases
Press Releases, News Notes, Statements [Selected]
Featured News
07 April 2022
On World Health Day, Unitaid rallies around the “Our Planet, our Health” campaign
Geneva – Unitaid joins partners to observe World Health Day 2022 today, reaffirming its strong commitment to contribute to keep humans and the planet healthy.
Climate change has been identified as the single biggest health threat of the 21st century, already harming the world’s most vulnerable people and menacing to undermine decades of progress in global health…
Vaccine Equity Cooperative [nee Initiative] [to 09 Apr 2022]
News
No new digest content identified.
Vaccination Acceptance & Demand Initiative [Sabin) [to 09 Apr 2022]
https://www.vaccineacceptance.org/
Announcements, Blog
No new digest content identified.
Vaccine Confidence Project [to 09 Apr 2022]
http://www.vaccineconfidence.org/
News, Research and Reports
No new digest content identified.
Vaccine Education Center – Children’s Hospital of Philadelphia [to 09 Apr 2022]
http://www.chop.edu/centers-programs/vaccine-education-center
No new digest content identified.
Wellcome Trust [to 09 Apr 2022]
News. Opinion, Reports
No new digest content identified.
The Wistar Institute [to 09 Apr 2022]
https://www.wistar.org/news/press-releases
No new digest content identified.
WFPHA: World Federation of Public Health Associations [to 09 Apr 2022]
Latest News – Blog
No new digest content identified.
World Bank [to 09 Apr 2022]
http://www.worldbank.org/en/news/all
Date: April 08, 2022 Type: Statement
World Customs Organization – WCO [to 09 Apr 2022]
Latest News
08 April 2022
Ethics and Transparency: A commitment for all
With the aim of exchanging best practices and motivating Customs Administrations to promote ethics through WCO tools, the Webinar “Ethics and Transparency – A Commitment for All” was organized by the Private Sector Consultative Group and the WCO Regional Vice-Chair for the Americas and the Caribbean…
World Organisation for Animal Health (OIE) [to 09 Apr 2022]
https://www.oie.int/en/media/news/
Press Releases, Statements
Acting for better aquatic animal health and welfare worldwide
3 April 2022 France, Paris
The World Organisation for Animal Health (OIE) joins the World Aquatic Animal Day celebrations by raising awareness on the need to maintain sustainable aquatic ecosystems to ensure a better future for all.
Over 50 million people worldwide depend on fisheries and aquaculture for their livelihoods. However, aquatic animal diseases can jeopardise the economic and environmental sustainability of this source of income. For instance, on a global scale, disease outbreaks cost the aquaculture industry over US$6 billion per year…
WTO – World Trade Organisation [to 09 Apr 2022]
http://www.wto.org/english/news_e/news_e.htm
WTO News and Events [Selected]
Members welcome COVID-19 trade-related measures report, agree on future experience sharing
31 March 2022
Members welcomed the latest update on export restrictions and prohibitions and trade-easing measures relating to the COVID-19 pandemic, which was presented by the WTO Secretariat at a meeting of the Committee on Market Access on 30-31 March. The Committee also heard about the first experience-sharing session on COVID-19-related goods, where members sought to better understand how they define lists of “essential goods” to combat the pandemic and the challenges they face with tariff classification.
DDG Ellard: WTO is essential to the rules-based international order
31 March 2022
In her address to the Steering Committee of the Inter-Parliamentary Union on 31 March, Deputy Director-General Angela Ellard provided an overview of the state of negotiations in the run-up to the 12th Ministerial Conference (MC12). She also highlighted the importance of the rules based international order and multilateralism, particularly in light of the war in Ukraine, and called on parliamentarians to help “make the world safer and more prosperous through trade”. The full text of her remarks is below.
::::::
ARM [Alliance for Regenerative Medicine] [to 09 Apr 2022]
Selected Press Releases
No new digest content identified.
BIO [to 09 Apr 2022]
https://www.bio.org/press-releases
Press Releases, Letters, Testimony, Comments [Selected]
No new digest content identified.
DCVMN – Developing Country Vaccine Manufacturers Network [to 09 Apr 2022]
No new digest content identified.
ICBA – International Council of Biotechnology Associations [to 09 Apr 2022]
https://internationalbiotech.org/news/
News
No new digest content identified.
http://www.ifpma.org/resources/news-releases/
Selected Press Releases, Statements, Publications
Largest pharmaceutical industry-backed collective venture fund tackling antimicrobial resistance announces first portfolio investments
April 4, 2022, Geneva – The international trade body representing the biopharmaceutical industry (IFPMA) today welcomes the announcement of the AMR Action Fund’s first portfolio investments. The Fund is the world’s largest public-private partnership investing in biotech companies that are developing antibiotics, involving more than 20 leading biopharmaceutical companies who raised the majority of the approximately US $1 billion when the Fund was launched in 2020. IFPMA played an instrumental role conceiving the Fund in collaboration with the European Investment Bank, the Wellcome Trust and the World Health Organization.
The announcement marks an important milestone for the Fund, which has as its goal to invest in the development of between two and four new antibiotics by 2030. New antibiotics are critical to helping stem the rise of antimicrobial resistance (AMR), which is a leading cause of death worldwide. At least 1.27 million deaths per year are directly attributable to infections that have grown resistant to existing antibiotics, with an additional 4.95 million deaths associated with resistant infections…
[See AMR Action Fund announcements in Perspectives above]
International Generic and Biosimilar Medicines Association [IGBA]
https://www.igbamedicines.org/
News
No new digest content identified.
International Alliance of Patients’ Organizations – IAPO [to 09 Apr 2022]
https://www.iapo.org.uk/news/topic/6
Press and media [Selected]
No new digest content identified.
PhRMA [to 09 Apr 2022]
Latest News [Selected]
No new digest content identified.
Journal Watch
Vaccines and Global Health: The Week in Review continues its weekly scanning of key peer-reviewed journals to identify and cite articles, commentary and editorials, books reviews and other content supporting our focu-s on vaccine ethics and policy. Journal Watch is not intended to be exhaustive, but indicative of themes and issues the Center is actively tracking. We selectively provide full text of some editorial and comment articles that are specifically relevant to our work. Successful access to some of the links provided may require subscription or other access arrangement unique to the publisher.
If you would like to suggest other journal titles to include in this service, please contact David Curry at: david.r.curry@centerforvaccineethicsandpolicy.org
AMA Journal of Ethics
Volume 24, Number 4: E249-341 Apr 2022
https://journalofethics.ama-assn.org/issue/health-equity-us-latinx-communities
Health Equity in US Latinx Communities
Latinx communities in the US include over 60 million people with a plurality of political beliefs, cultural practices, and wealth. This diversity is also expressed in population health data: some health indicators suggest advantages of Latinx community membership while other data demonstrate inequitable disease burden and maldistribution of environmental and occupational risk. Legacies of colonial conquest of the Americas persist in discrimination and marginalization today and are embodied by members of our Latinx communities.
American Journal of Public Health
April 2022 112(52)
http://ajph.aphapublications.org/toc/ajph/current
Supplement 2 2022 = Global Opioid Crisis
BMJ Global Health
April 2022 – Volume 7 – 4
https://gh.bmj.com/content/7/4
Analysis
Ethical considerations in international clinical trial site selection (6 April, 2022)
Jennifer Miller, Joseph Millum
Abstract
New medicines and vaccines are predominantly tested in high-income countries. However, as the COVID-19 pandemic highlighted, the populations who can benefit from these interventions are not limited to these wealthier regions. One-third of novel Food and Drug Administration approved drugs, sponsored by large companies, treat infectious diseases like tuberculosis and HIV, which disproportionately affect low-income and middle-income countries (LMICs). The medicines for non-communicable diseases (NCDs) are also relevant to LMIC health needs, as over three-quarters of deaths from NCDs occur in LMICs. There are concerns clinical trial data may not extrapolate across geographical regions, as product effectiveness can vary substantially by region. The pentavalent rotavirus vaccine, for example, had markedly lower efficacy in LMICs. Efficacy variations have also been found for other vaccines and drugs. We argue there are strong ethical arguments for remedying some of this uneven distribution of clinical trial sites by geography and income. Chief among them, is that these disparities can impede equitable access to the benefits of clinical research, such as representation in the evidence base generated to guide prescribing and use of medicines and vaccines. We suggest trial site locations should be made more transparent and for later stage trials their selection should be informed by the global distribution of disease burden targeted by an experimental product. Countries with high prevalence, incidence, severity or infection transmission rates for targeted diseases should have real opportunities to engage in and enrol their populations in trials for novel medicines and vaccines.
BMC Health Services Research
http://www.biomedcentral.com/bmchealthservres/content
(Accessed 09 Apr 2022)
Examining the unit costs of COVID-19 vaccine delivery in Kenya
Vaccines are considered the path out of the COVID-19 pandemic. The government of Kenya is implementing a phased strategy to vaccinate the Kenyan population, initially targeting populations at high risk of seve…
Authors: Stacey Orangi, Angela Kairu, Anthony Ngatia, John Ojal and Edwine Barasa
Citation: BMC Health Services Research 2022 22:439
Content type: Research Published on: 4 April 2022