Vaccine
Volume 39, Issue 49 Pages 7117-7220 (3 December 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/49
Discussion Full text access
COVID-19 from a polio perspective
Robert Hill
Pages 7117-7118
Vaccine
Volume 39, Issue 49 Pages 7117-7220 (3 December 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/49
Discussion Full text access
COVID-19 from a polio perspective
Robert Hill
Pages 7117-7118
Vaccine
Volume 39, Issue 49 Pages 7117-7220 (3 December 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/49
Discussion Full text access
SARS-CoV-2 vaccine for domestic and captive animals: An effort to counter COVID-19 pandemic at the human-animal interface
Khan Sharun, Ruchi Tiwari, AbdulRahman A. Saied, Kuldeep Dhama
Pages 7119-7122
Vaccine
Volume 39, Issue 49 Pages 7117-7220 (3 December 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/49
Discussion Full text access
The influence of interval between doses on response to vaccines
Charlene M.C. Rodrigues, Stanley A. Plotkin
Pages 7123-7127
Vaccine
Volume 39, Issue 49 Pages 7117-7220 (3 December 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/49
Discussion Full text access
COVAX no fault compensation program for COVID-19 vaccine injuries in 92 low and middle income countries
Anne Mazur, Stefanie Benitez, Stéphanie Chuffart-Finsterwald, Rafael La Rotta, Lee M. Hampton
Pages 7128-7130
Vaccine
Volume 39, Issue 49 Pages 7117-7220 (3 December 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/49
Research article Full text access
Social inequalities in COVID-19 vaccine acceptance and uptake for children and adolescents in Montreal, Canada
Britt McKinnon, Caroline Quach, Ève Dubé, Cat Tuong Nguyen, Kate Zinszer
Pages 7140-7145
Vaccine
Volume 39, Issue 49 Pages 7117-7220 (3 December 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/49
Research article Abstract only
Mandatory immunization and vaccine injury support programs: A survey of 28 GNN countries
Shawn H.E. Harmon, David E. Faour, Noni E. MacDonald
Pages 7153-7157
Vaccine
Volume 39, Issue 49 Pages 7117-7220 (3 December 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/49
Advancing vaccine-product innovations for LMICs: Updates from the Vaccine Innovation Prioritisation Strategy (VIPS)
Discussion Full text access
A global collaboration to advance vaccine product innovations – The Vaccine Innovation Prioritisation Strategy
Debra Kristensen, Birgitte Giersing, Julian Hickling, Fatima Kazi, … Marion Menozzi-Arnaud
Pages 7191-7194
Vaccine
Volume 39, Issue 49 Pages 7117-7220 (3 December 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/49
Research article Open access
Vaccine innovation prioritisation strategy: Findings from three country-stakeholder consultations on vaccine product innovations
Mercy Mvundura, Collrane Frivold, Anna Janik Osborne, Priyanka Soni, … Debra Kristensen
Pages 7195-7207
Vaccine
Volume 39, Issue 49 Pages 7117-7220 (3 December 2021)
https://www.sciencedirect.com/journal/vaccine/vol/39/issue/49
Review article Open access
Strategies for vaccine-product innovation: Creating an enabling environment for product development to uptake in low- and middle-income countries
Birgitte Giersing, Natasha Shah, Debra Kristensen, Jean-Pierre Amorij, … Marion Menozzi-Arnaud
Pages 7208-7219
Pre-Print Servers
Gates Open Research
https://gatesopenresearch.org/browse/articles
[Accessed 11 Dec 2021]
Research Article metrics AWAITING PEER REVIEW
Do differentiated service delivery models for HIV treatment in sub-Saharan Africa save money? Synthesis of evidence from field studies conducted in sub-Saharan Africa in 2017-2019 [version 1; peer review: awaiting peer review]
Sydney Rosen, Brooke Nichols, Teresa Guthrie, Mariet Benade, Salome Kuchukhidze, Lawrence Long
Peer Reviewers Invited
Funder – NIH
PUBLISHED 08 Dec 2021
medRxiv
https://www.medrxiv.org/content/about-medrxiv
medRxiv is a free online archive and distribution server for complete but unpublished manuscripts (preprints) in the medical, clinical, and related health sciences. Preprints are preliminary reports of work that have not been certified by peer review. They should not be relied on to guide clinical practice or health-related behavior and should not be reported in news media as established information. medRxiv is for the distribution of preprints – complete but unpublished manuscripts – that describe human health research conducted, analyzed, and interpreted according to scientific principles…
[Accessed 11 Dec 2021]
Selected Content
SARS-CoV-2 Omicron has extensive but incomplete escape of Pfizer BNT162b2 elicited neutralization and requires ACE2 for infection
Sandile Cele, Laurelle Jackson, Khadija Khan, David S Khoury, Thandeka Moyo-Gwete, Houriiyah Tegally, Cathrine Scheepers, Daniel Amoako, Farina Karim, Mallory Bernstein, Gila Lustig, Derseree Archary, Muneerah Smith, Yashica Ganga, Zesuliwe Jule, Kajal Reedoy, James Emmanuel San, Shi-Hsia Hwa, Jennifer Giandhari, Jonathan Blackburn, Bernadett I Gosnell, Salim Abdool Karim, Willem Hanekom, NGS-SA, COMMIT-KZN Team, Anne von Gottberg, Jinal Bhiman, Richard Lessells, Mahomed-Yunus S Moosa, Miles Philip Davenport, Tulio de Oliveira, Penny L. Moore, Alex Sigal
medRxiv 2021.12.08.21267417; doi: https://doi.org/10.1101/2021.12.08.21267417
The prevalence of adaptive immunity to COVID-19 and reinfection after recovery, a comprehensive systematic review and meta-analysis
Tawanda Chivese, Joshua Matizanadzo, Omran Musa, George Hindy, Luis Furuya-Kanamori, Nazmul Islam, Rafal Al-Shebly, Rana Shalaby, Mohammad Habibullah, Talal Al-Marwani, Rizeq F Hourani, Ahmed D Nawaz, Mohammad Haider, Mohamed M Emara, Farhan Cyprian, Suhail A. R. Doi
medRxiv 2021.09.03.21263103; doi: https://doi.org/10.1101/2021.09.03.21263103
Screening and vaccination against COVID-19 to minimize school closure
Elisabetta Colosi, Giulia Bassignana, Diego A Contreras, Canelle Poirier, Pierre-Yves Boëlle, Simon Cauchemez, Yazdan Yazdanpanah, Bruno Lina, Arnaud Fontanet, Alain Barrat, Vittoria Colizza
medRxiv 2021.08.15.21261243; doi: https://doi.org/10.1101/2021.08.15.21261243
Estimates of reduced vaccine effectiveness against hospitalization, infection, transmission and symptomatic disease of a new SARS-CoV-2 variant, Omicron (B.1.1.529), using neutralizing antibody titers
Billy J Gardner, A. Marm Kilpatrick
medRxiv 2021.12.10.21267594; doi: https://doi.org/10.1101/2021.12.10.21267594
Knowledge, attitudes, and practices (KAP) towards COVID-19: A quick online cross-sectional survey among Tanzanian residents.
sima rugarabamu, Aisha Byanaku, Mariam Ibrahim
medRxiv 2020.04.26.20080820; doi: https://doi.org/10.1101/2020.04.26.20080820
Surveillance of COVID-19 vaccine effectiveness – a real-time case-control study in southern Sweden
Jonas Björk, Carl Bonander, Mahnaz Moghaddassi, Magnus Rasmussen, Ulf Malmqvist, Fredrik Kahn, Malin Inghammar
medRxiv 2021.12.09.21267515; doi: https://doi.org/10.1101/2021.12.09.21267515
Trust in the research community predicts intent to comply with COVID-19 prevention measures: An analysis of a large-scale international survey dataset
Hyemin Han
medRxiv 2021.12.08.21267486; doi: https://doi.org/10.1101/2021.12.08.21267486
Vaccine hesitancy for COVID-19 explored in a phenomic study of 259 socio-cognitive-behavioural measures in the UK-REACH study of 12,431 UK healthcare workers
I Chris McManus, Katherine Woolf, Christopher A Martin, Laura B Nellums, Anna L Guyatt, Carl Melbourne, Luke Bryant, Amit Gupta, Catherine John, Martin D Tobin, Sue Carr, Sandra Simpson, Bindu Gregary, Avinash Aujayeb, Stephen Zingwe, Rubina Reza, Laura J Gray, Kamlesh Khunti, Manish Pareek
medRxiv 2021.12.08.21267421; doi: https://doi.org/10.1101/2021.12.08.21267421
Vaccine hesitancy for COVID-19 explored in a phenomic study of 259 socio-cognitive-behavioural measures in the UK-REACH study of 12,431 UK healthcare workers
I Chris McManus, Katherine Woolf, Christopher A Martin, Laura B Nellums, Anna L Guyatt, Carl Melbourne, Luke Bryant, Amit Gupta, Catherine John, Martin D Tobin, Sue Carr, Sandra Simpson, Bindu Gregary, Avinash Aujayeb, Stephen Zingwe, Rubina Reza, Laura J Gray, Kamlesh Khunti, Manish Pareek
medRxiv 2021.12.08.21267421; doi: https://doi.org/10.1101/2021.12.08.21267421
Global disparities in SARS-CoV-2 genomic surveillance
Anderson F. Brito, Elizaveta Semenova, Gytis Dudas, Gabriel W. Hassler, Chaney C. Kalinich, Moritz U.G. Kraemer, Joses Ho, Houriiyah Tegally, George Githinji, Charles N. Agoti, Lucy E. Matkin, Charles Whittaker, Danish Covid-19 Genome Consortium, COVID-19 Impact Project, Network for Genomic Surveillance in South Africa (NGS-SA), GISAID core curation team, Benjamin P Howden, Vitali Sintchenko, Neta S. Zuckerman, Orna Mor, Heather M Blankenship, Tulio de Oliveira, Raymond T. P. Lin, Marilda Mendonça Siqueira, Paola Cristina Resende, Ana Tereza R. Vasconcelos, Fernando R. Spilki, Renato Santana Aguiar, Ivailo Alexiev, Ivan N. Ivanov, Ivva Philipova, Christine V. F. Carrington, Nikita S. D. Sahadeo, Céline Gurry, Sebastian Maurer-Stroh, Dhamari Naidoo, Karin J von Eije, Mark D. Perkins, Maria van Kerkhove, Sarah C. Hill, Ester C. Sabino, Oliver G. Pybus, Christopher Dye, Samir Bhatt, Seth Flaxman, Marc A. Suchard, Nathan D. Grubaugh, Guy Baele, Nuno R. Faria
medRxiv 2021.08.21.21262393; doi: https://doi.org/10.1101/2021.08.21.21262393
Abstract
Genomic sequencing provides critical information to track the evolution and spread of SARS-CoV-2, optimize molecular tests, treatments and vaccines, and guide public health responses. To investigate the spatiotemporal heterogeneity in the global SARS-CoV-2 genomic surveillance, we estimated the impact of sequencing intensity and turnaround times (TAT) on variant detection in 167 countries. Most countries submit genomes >21 days after sample collection, and 77% of low and middle income countries sequenced <0.5% of their cases. We found that sequencing at least 0.5% of the cases, with a TAT <21 days, could be a benchmark for SARS-CoV-2 genomic surveillance efforts. Socioeconomic inequalities substantially impact our ability to quickly detect SARS-CoV-2 variants, and undermine the global pandemic preparedness. The challenge of limited vaccine supplies: impact of prior infection on anti-spike IgG antibody trajectories after a single COVID-19 vaccination
Jia Wei, Philippa C. Matthews, Nicole Stoesser, Ian Diamond, Ruth Studley, Emma Rourke, Duncan Cook, John I Bell, John N Newton, Jeremy Farrar, Alison Howarth, Brian D. Marsden, Sarah Hoosdally, E Yvonne Jones, David I Stuart, Derrick W. Crook, Tim E. A. Peto, A. Sarah Walker, David W. Eyre, Koen B. Pouwels, the COVID-19 Infection Survey team
medRxiv 2021.12.08.21267353; doi: https://doi.org/10.1101/2021.12.08.21267353
Long COVID Citizen Scientists – Developing a needs-based research agenda by persons affected by Long COVID
Sarah Ziegler, Alessia Raineri, Vasileios Nittas, Natalie Rangelov, Fabian Vollrath, Chantal Britt, Milo A Puhan
medRxiv 2021.12.08.21267181; doi: https://doi.org/10.1101/2021.12.08.21267181
COVID-19 Vaccination Mandates and Vaccine Uptake
Alexander Karaivanov, Dongwoo Kim, Shih En Lu, Hitoshi Shigeoka
medRxiv 2021.10.21.21265355; doi: https://doi.org/10.1101/2021.10.21.21265355
Abstract
We evaluate the impact of government mandated proof of vaccination requirements for access to public venues and non-essential businesses on COVID-19 vaccine uptake. We find that the announcement of a mandate is associated with a rapid and significant surge in new vaccinations (more than 60% increase in weekly first doses) using the variation in the timing of these measures across Canadian provinces in a difference-in-differences approach. Time-series analysis for each province and for France, Italy and Germany corroborates this finding, and we estimate cumulative gains of up to 5 percentage points in provincial vaccination rates and 790,000 or more first doses for Canada as a whole as of October 31, 2021 (5 to 13 weeks after the provincial mandate announcements). We also find large vaccination gains in France (3 to 5 mln first doses), Italy (around 6 mln) and Germany (around 3.5 mln) 11 to 16 weeks after the proof of vaccination mandate announcements.
COVID-19 infections post-vaccination by HIV status in the United States
Sally B. Coburn, Elizabeth Humes, Raynell Lang, Cameron Stewart, Brenna C Hogan, Kelly A. Gebo, Sonia Napravnik, Jessie K. Edwards, Lindsay E. Browne, Lesley S. Park, Amy C. Justice, Kirsha Gordon, Michael A. Horberg, Julia M. Certa, Eric Watson, Celeena R Jefferson, Michael Silverberg, Jacek Skarbinski, Wendy A Leyden, Carolyn F. Williams, Keri N. Althoff, for the Corona-Infectious-Virus Epidemiology Team (CIVETs) of the NA-ACCORD of IeDEA
medRxiv 2021.12.02.21267182; doi: https://doi.org/10.1101/2021.12.02.21267182
Wellcome Open Research [to 11 Dec 2021]
https://wellcomeopenresearch.org/browse/articles
[Accessed 11 Dec 2021]
Wellcome Open Research provides all Wellcome researchers with a place to rapidly publish any results they think are worth sharing. All articles benefit from rapid publication, transparent peer review and editorial guidance on making all source data openly available.
Research Article metrics
Revised
Estimating the number of undetected COVID-19 cases among travellers from mainland China [version 3; peer review: 3 approved]
Sangeeta Bhatia, Natsuko Imai, Gina Cuomo-Dannenburg, Marc Baguelin, Adhiratha Boonyasiri, Anne Cori, Zulma Cucunubá, Ilaria Dorigatti, Rich FitzJohn, Han Fu, Katy Gaythorpe, Azra Ghani, Arran Hamlet, Wes Hinsley, Daniel Laydon, Gemma Nedjati-Gilani, Lucy Okell, Steven Riley, Hayley Thompson, Sabine van Elsland, Erik Volz, Haowei Wang, Yuanrong Wang, Charles Whittaker, Xiaoyue Xi, Christl A. Donnelly, Neil M. Ferguson
Peer Reviewers Jeremy Bingham and Juliet R.C. Pulliam; Sebastian Funk; Hannah E. Clapham
Funders
Wellcome Trust
European and Developing Countries Clinical Trials Partnership
National Institute for Health Research
Department for International Development, UK Government
Medical Research Council
LATEST VERSION PUBLISHED 06 Dec 2021
Think Tanks
Brookings [to 11 Dec 2021]
http://www.brookings.edu/
Accessed 11 Dec 2021
Up Front
What we can learn from Israel’s COVID-19 vaccination of children
Oren Heller, Yaniv Shlomo, and Michal Grinstein-Weiss
Friday, December 10, 2021
A study conducted by the Social Policy Institute at Washington University in St. Louis shows that among surveyed parents in Israel, only 37 percent intended to vaccinate their child (aged 5-11) against COVID-19 when the option became available, while 23 percent of parents were undecided and 40 percent had no intention to vaccinate their children.
The study was conducted November 3-8, 2021, among a representative sample of Israeli parents of children aged 5-11 (N = 912)…
Center for Global Development [to 11 Dec 2021]
http://www.cgdev.org/page/press-center
Accessed 11 Dec 2021
Applications of Machine Learning and Advanced Analytics in Pandemic Preparedness and Response: Translating Models into Policy Action
Event
12/15/21
The Updated International Health Regulations Need Better Rules for Travel Restrictions
December 9, 2021
The last week has demonstrated the level of security theatre that has often been involved with travel bans put in place to reduce the spread of COVID-19. But heavy travel restrictions have played a role in the strategies of some of the countries that have most successfully battled the pandemic. As part of an effort to strengthen the International Health Regulations, the World Health Organization (WHO) should propose approaches that will limit travel restrictions to where they are most likely to be effective and design them to be minimally disruptive.
Charles Kenny
We Should Prepare for an Omicron COVID-19 Wave by Prioritising Cost-Effective Essential Emergency Critical Care Now
December 6, 2021
Hiral Anil Shah et al.
Chatham House [to 11 Dec 2021]
https://www.chathamhouse.org/
Accessed 11 Dec 2021
Covid’s solidarity failure
Pledges of ‘vaccines for everyone’ may have gone unfulfilled but the case for universal health reforms is gaining ground, writes Rob Yates
The World Today
3 December 2021
CSIS
https://www.csis.org/
Accessed 11 Dec 2021
Upcoming Event
The Road to the 2022 Summit of the Americas: Vaccine Diplomacy and Engagement
December 14, 2021
Kaiser Family Foundation
https://www.kff.org/search/?post_type=press-release
Accessed 11 Dec 2021
December 10, 2021 News Release
Health Employment Continues Slow Recovery Since the Beginning of the Pandemic
Unlike past recessions, the health sector saw a big drop in employment in early 2020 similar to other sectors as the COVID-19 pandemic shut down much of the nation’s economy and remains below expected employment levels through November 2021, a new KFF chart collection shows. The chart collection takes a…
December 9, 2021 News Release
Half of Parents of Adolescents 12-17 Say Their Child Has Gotten a COVID-19 Vaccine, though Uptake Has Slowed; 16% of Parents of 5-11 Year-olds Say Their Child Has Gotten a Vaccine
COVID-19 vaccine uptake among adolescents ages 12-17 has slowed after an initial wave of enthusiasm over the summer, with half (49%) of parents saying their adolescent has received at least one dose, a new KFF COVID-19 Vaccine Monitor report reveals. The share is little changed since earlier in the fall.…
December 8, 2021 News Release
Following an Early Period of High Demand, Vaccination for Children Ages 5-11 Has Significantly Slowed
As of December 5, 16.7% of 5-11 year-olds had received at least one COVID-19 vaccine dose with 4.3% of children reaching full vaccination, according to a new KFF analysis. After a short period of high demand, the rate of new vaccinations slowed significantly leading into the Thanksgiving holiday and has…
ODI [Overseas Development Institute] [to 11 Dec 2021]
https://odi.org/en/publications/
Publications
[No new digest content identified]
Vaccines and Global Health: The Week in Review is a weekly digest summarizing news, events, announcements, peer-reviewed articles and research in the global vaccine ethics and policy space. Content is aggregated from key governmental, NGO, international organization and industry sources, key peer-reviewed journals, and other media channels. This summary proceeds from the broad base of themes and issues monitored by the Center for Vaccine Ethics & Policy in its work: it is not intended to be exhaustive in its coverage. You are viewing the blog version of our weekly digest, typically comprised of between 30 and 40 posts below all dated with the current issue date
.– Request an Email Summary: Vaccines and Global Health : The Week in Review is published as a single email summary, scheduled for release each Saturday evening before midnight (EDT in the U.S.). If you would like to receive the email version, please send your request to david.r.curry@centerforvaccineethicsandpolicy.org.
– pdf version: A pdf of the current issue is available here:
– blog edition: comprised of the approx. 35+ entries posted below.
– Twitter: Readers can also follow developments on twitter: @vaxethicspolicy.
.
– Links: We endeavor to test each link as we incorporate it into any post, but recognize that some links may become “stale” as publications and websites reorganize content over time. We apologize in advance for any links that may not be operative. We believe the contextual information in a given post should allow retrieval, but please contact us as above for assistance if necessary.
Support this knowledge-sharing service: Your financial support helps us cover our costs and to address a current shortfall in our annual operating budget. Click here to donate and thank you in advance for your contribution.
.
David R. Curry, MS
Executive Director
Center for Vaccine Ethics and Policy
Gavi Board approves malaria vaccine programme funding, COVAX 2022 strategic approach and measures to maintain, restore and strengthen routine immunisation
A new malaria vaccination programme has been approved, providing Gavi-eligible countries in sub-Saharan Africa a powerful new tool with which to fight malaria
In 2022, Gavi, through COVAX, will continue to help countries meet COVID-19 vaccination targets, focusing mostly on lower-income countries and those most in need including support for delivery at scale
Gavi will invest US$ 250 million over 2022-2026 in its partnership with India; US$ 53 million over 2022-2025 will be channelled towards supporting the availability of diagnostic tests for yellow fever, cholera, typhoid, meningococcus, measles, and rubella
José Manuel Barroso, Board Chair: “I am greatly heartened that the Gavi Board has endorsed decisions that allow progress to be made on all fronts: for leveraging the latest innovations to tackle malaria, for COVAX to support national COVID-19 vaccination strategies, and for maintaining, restoring and strengthening routine immunisation programmes – including targeting the increasing numbers of zero dose children in the world today.”
Geneva, 3 December 2021 – The Board of Gavi, the Vaccine Alliance this week approved a number of measures aimed at tackling malaria, driving equitable global access to COVID-19 vaccines and maintaining, restoring and strengthening routine immunisation in 2022.
During the three-day meeting, the Board approved a new malaria vaccination programme to support the introduction, procurement and delivery of the malaria vaccine to Gavi-eligible countries in sub-Saharan Africa. An initial investment of US$ 155.7 million for 2022-2025 will initiate the implementation of this additional tool in the fight against malaria alongside currently recommended malaria control interventions, which could save tens of thousands of lives annually and drive down child mortality in Africa.
The Board also approved the Vaccine Alliance’s strategic direction and role in COVAX. In 2022, Gavi, through COVAX, will continue building the foundations of COVID-19 protection with a focus on lower-income countries and the most in need; deploying a flexible portfolio of COVID-19 vaccines to manage uncertainty and anticipate risk; and providing support for delivery at-scale.
“Events of 2021, illustrated most recently by the emergence of the omicron variant, have highlighted the danger of leaving COVID-19 to flourish unchecked in large parts of the world, but also of the vital importance of maintaining routine life-saving immunisation programmes that have been hit so hard by the pandemic”, said José Manuel Barroso, Chair of the Gavi Board. “I am greatly heartened that the Gavi Board has endorsed decisions that allow progress to be made on all fronts: for leveraging the latest innovations to tackle malaria, for COVAX to support national COVID-19 vaccination strategies, and for maintaining, restoring and strengthening routine immunisation programmes – including targeting the increasing numbers of zero dose children in the world today.”
Malaria vaccine programme
The Board’s historical approval of a malaria vaccine programme and financing will provide a critical new tool for African countries in the fight against malaria. With an initial investment of approximately US$ 155.7 million for 2022-2025, the Board noted that a successful malaria vaccine programme should support deliberate and intensive coordination between malaria control and immunisation programmes at global and country levels to ensure most impactful deployment of the vaccine alongside other interventions.
The Board also recognised the opportunity for integration and strengthening of primary healthcare given the wide acceptance of the vaccine among caregivers and healthcare workers, as was seen during the Malaria Vaccine Implementation Programme (MVIP). Child immunisation provides a powerful platform to reach vulnerable children, including those who are unreached with bednets or other existing prevention measures, and can help advance the equity agenda. Finally, the Board noted opportunities for next generation vaccines and a need for market-shaping efforts to support the development of a healthy malaria vaccine market.
“Today marks an important milestone in our fight against malaria,” said Dr Seth Berkley, CEO of Gavi, the Vaccine Alliance. “This decision by the Gavi Board to finance a new malaria vaccination programme for countries in sub-Saharan Africa could save tens of thousands of lives annually in Africa. The vaccine is an important additional tool to control malaria in Africa, alongside other interventions, such as routine use of insecticide-treated bed nets, indoor spraying with insecticides, malaria chemoprevention, and timely testing and treatment. We look forward to working with global malaria stakeholders to make sure that countries are able to implement this additional tool in the fight against malaria.”
After considerable gains in the past two decades, progress in malaria control has stalled and new tools are urgently needed to get back on track. More than 260,000 African children under the age of five die from malaria annually, and six Gavi-eligible countries account for 50% of global mortality. Africa continues to bear the heaviest malaria burden and African children are at highest risk of dying of malaria, especially at a time when the COVID-19 pandemic threatens disruptions to immunisation programmes.
Following the decision by the Gavi Board, a series of critical next steps will need to take place before the vaccine is rolled-out to Gavi-supported countries. This includes technical guidance on the use of the vaccine alongside other malaria interventions, procurement of the vaccine, and the opening of the funding window to allow applications from Gavi-supported countries. Additionally, countries will need to make decisions on the rollout of malaria vaccine, taking in consideration such factors as the public health priority of the disease, evidence of disease burden, and the optimal mix of malaria interventions tailored to the local context.
COVAX’s 2022 strategic approach
The Board approved the Vaccine Alliance’s strategic direction for 2022 to support 91 lower-income countries towards achieving their individual COVID-19 vaccination coverage ambitions in view of the WHO Global Vaccination Target of 70% by mid-2022 and taking into account sources of supply beyond COVAX. COVAX has supplied over 80% of all vaccines shipped to low-income countries and has secured access to enough additional doses to protect around 40% of each countries’ population by early next year.
Subject to funding availability for the COVAX AMC, the COVAX Facility’s approach to procurement of COVID-19 vaccines for 2022, devised to provide maximum flexibility in the event of new evidence appearing or markets evolving, was also approved by the Gavi Board. The core element of the approach, the Pandemic Vaccine Pool, will enable COVAX to manage demand- and supply-side risks, as well as provide doses to the countries that are most reliant on COVAX, including vulnerable countries, countries where current rates of coverage are the lowest and others that need certainty of supply in order to be able to plan successfully.
The COVAX 2022 strategic approach also recognises the difference in countries’ capacity to roll-out vaccines and the fact that many partners are providing support to countries to scale up delivery. The Alliance’s role in the scale up of COVID-19 vaccination in 2022 will focus on more systematic collaboration with other funders at global, regional and country levels; more targeted financial support and enhanced technical support to AMC-eligible countries; as well as enhanced monitoring of delivery progress and risks.
In order to strengthen COVAX’s processes from supply allocation to last mile delivery, the Board recognised the need to establish an enhanced and unified COVAX delivery coordination structure, agreeing to set up a temporary Steering Committee with delegated authority over delivery-related strategy and decisions of the COVAX Facility. The Committee will also oversee COVID-19 vaccination delivery support provided by COVAX, including alignment on allocation strategy, country absorptive capacity and funding. Recognising the important role that each Vaccine Alliance member needs to play in this coordination structure, it also requested that the new structure work with existing and additional partners and countries to rapidly accelerate vaccination delivery, including through campaigns, mass vaccination and hard-to-reach populations while safeguarding routine immunisation. The enhanced coordination structure will report on delivery progress to the Gavi Board through the temporary Steering Committee on a regular basis.
The Board also delegated to Gavi, at the guidance of the new COVAX coordination structure, the authority to allot current and future COVID-19 delivery funding without requiring independent review given the emergency context and urgent country needs, while utilising existing programmatic and fiduciary risk mitigation mechanisms.
The Board also noted the importance of COVAX’s cost-sharing mechanism as an important voluntary mechanism for countries to access additional doses and approved its extension until December 2023.
Disease surveillance and diagnostics
As part of Gavi’s efforts to restore, maintain and strengthen routine immunisation, the Board approved US$ 32 million in funding in 2022-2025 for costs related to the procurement and distribution of yellow fever, cholera, typhoid, meningococcus, measles, and rubella diagnostic test kits, reagents, supplies, and equipment. This market purchase commitment will put incentives in place for improved commercially available diagnostics. Additionally, US$ 21 million was approved to ensure that any test kits procured with Gavi funding are accurate, reliable, and useful for vaccine programmes’ efforts to use vaccines in the right places and times.
Fit-for-purpose diagnostic tools are critical for countries’ abilities to plan and implement targeted vaccination programmes. Making such diagnostic tools available to countries will help make sure that Gavi investments in these vaccine programmes, which are currently projected at over US$ 1.6 billion during 2021-2025, will be more efficient, effective, and equitable.
The Board also approved:
The renewed Gavi’s strategic partnership with India and the associated investment of US$ 250 million for 2022-2026, which will aim to further decrease the number of zero-dose and under-immunised children and expand full immunisation coverage in the next five years, as well as provide catalytic support for the introduction of Human papillomavirus (HPV) and Typhoid conjugate vaccines (TCV) in India.
The Gavi’s private sector engagement strategy which is expected to play a critical role in supporting the strategic priorities of Gavi 5.0 and COVAX related objectives, such as the zero-dose agenda, Covid vaccine delivery, innovation from the private sector and immunisation systems strengthening.
The Risk & Assurance Report 2021, which shows that Gavi’s overall risk profile remains elevated due to the ongoing uncertain environment, Gavi’s ambitious strategy and the unprecedented mission of the COVAX Facility. As per Gavi’s updated Risk Appetite Statement, approved by the Board in its June 2021 meeting, these risks are worth taking, but Gavi continues to actively monitor and mitigate them to the extent possible.
The Financial Forecast for Gavi 5.0 for the 2021-2025 strategic period and the Financial Forecast for Gavi COVAX AMC for the 2020-2022 period.
Finally, the Gavi Board gave a warm send-off to Maty Dia, Abdoulaye Sabre Fadoul, Gilbert Mokoki, David Sidwell and Joan Valadou, who have all made an important contribution to Gavi’s efforts to accelerate the introduction of new vaccines and improve routine immunisation. The Board also welcomed new members, including Bernhard Braune, Silvia Lutcuta, Bvudzai Magadrize, Charlemagne Marie Ragnag-Néwendé Ouedraogo, Jan Paehler, Anne Schuchat and Rafael Vilasanjuan.
WHO Pandemic Ethics & Policy Summit
6 December 2021
Equitable access, solidarity, and global health justice: Bridging the gap between ethics and decision-making in pandemics
From resource allocation and priority-setting, access to vaccines, vaccine mandates, lockdowns, travel restrictions, public health surveillance, and obligations to conduct clinical trials, the COVID-19 pandemic has raised profound ethical challenges on an unprecedented global scale.
At the same time, and also in an unprecedented manner, ethical values like equity, fairness, solidarity, and trust have figured prominently in global political discussions. Despite an abundance of guidance, the question remains: how much progress, if any, have we made in achieving these values, in advancing towards global health justice? Much has been said about the importance of evidence-informed decision-making, but what have we achieved in terms of ‘Ethics-informed decision-making’? This Summit provides a forum to discuss the translation of ethics into policy making in COVID-19; where we have succeeded and failed; what are the major challenges and what steps need to be taken to improve ethics informed public health decision-making for pandemic preparedness and response.
The Summit is organized by the WHO Heath Ethics and Governance Unit in collaboration with the WHO’s Working Group on Ethics & COVID-19 and the ACT-Accelerator Ethics & Governance Working Group.
Confirmed speakers include: Thalia Arawi, Caesar Atuire, Alena Buyx, Ezekiel Emanuel, Ruth Faden, Anthony Fauci, Christine Grady, Fatima Hassan, Sharon Kaur, Ilona Kickbusch, Florencia Luna, Roli Mathur, Peter Singer, Jerome Singh, Soumya Swaminathan, Beatriz Thomé, Ross Upshur, Effy Vayena.
Register Here: https://who.zoom.us/webinar/register/WN_hjp0eY19RDawdZfj1EML8Q
::::::
Extraordinary meeting of the Strategic Advisory Group of Experts on Immunization (SAGE) – 7 December 2021
7 December 2021
This extraordinary virtual meeting for the Strategic Advisory Group of Experts on Immunization (SAGE) will take place on Tuesday 7 December 2021 to propose recommendations to WHO on the use of COVID-19 vaccine(s).
Agenda: https://cdn.who.int/media/docs/default-source/immunization/sage/2021/december/sage-agenda-7dect2021-virtual-draft-rev26nov.pdf?sfvrsn=f2b71e9f_10
World Health Assembly – Special Session
WHASS2: Special session of the World Health Assembly referred to in decision WHA74(16)
29 November – 1 December 2021
Geneva, Switzerland | Virtual event
WHASS2 Documents [at 4 Dec 2021]
:: Statements submitted by Member States and other participants at the Second special session of the World Health Assembly.
:: Statements by non-State actors in official relations with WHO at the WHO governing bodies meetings
Decisions
The World Together: Establishment of an intergovernmental negotiating body to strengthen pandemic prevention, preparedness and response
SSA2(5) 1 December 2021 :: 2 pages
The Second special session of the World Health Assembly,
:: Recalling resolution WHA74.7 and decision WHA74(16), and welcoming the report of the Member States Working Group on Strengthening WHO Preparedness and Response to Health Emergencies (WGPR),1
:: Expressing appreciation for the ongoing work of the WGPR under resolution WHA 74.7, including to identify the tools to implement the recommendations that fall under the technical work of WHO and further develop proposals to strengthen the International Health Regulations (IHR (2005)) including potential targeted IHR (2005) amendments, and elements that may most effectively be addressed in other venues;
:: Acknowledging the need to address gaps in preventing, preparing for, and responding to health emergencies, including in development and distribution of, and unhindered, timely and equitable access to, medical countermeasures such as vaccines, therapeutics and diagnostics, as well as strengthening health systems and their resilience with a view to achieving UHC;
:: Emphasizing the need for a comprehensive and coherent approach to strengthen the global health architecture, and recognizing the commitment of Member States to develop a new instrument for pandemic prevention, preparedness and response with a whole-of-government and whole-of-society approach, prioritizing the need for equity;
:: Stressing that Member States should guide their efforts to develop such an instrument by the principle of solidarity with all people and countries, that should frame practical actions to deal with both causes and consequences of pandemics and other health emergencies.
1. DECIDES:
(1) to establish, in accordance with Rule 41 of its Rules of Procedure, an intergovernmental negotiating body open to all Member States and Associate Members2 (the “INB”) to draft and negotiate a WHO convention, agreement or other international instrument on pandemic prevention, preparedness and response, with a view to adoption under Article 19, or under other provisions of the WHO Constitution as may be deemed appropriate by the INB;
(2) that the first meeting of the INB shall be held no later than 1 March 2022, in order to elect two co-chairs, reflecting a balance of developed and developing countries, and four vice-chairs, one from each of the six WHO regions, and to define and agree on its working methods and timelines, consistent with this decision and based on the principles of inclusiveness, transparency, efficiency, Member State leadership and consensus;
(3) that as part of its working methods, the INB shall determine an inclusive Member State led process, to be facilitated by the co-chairs and vice-chairs, to first identify the substantive elements of the instrument and to then begin the development of a working draft to be presented, on the basis of progress achieved, for the consideration of the INB at its second meeting, to be held no later than 1 August 2022, at the end of which the INB will identify the provision of the WHO Constitution under which the instrument should be adopted in line with paragraph 1(1);
(4) that the process referred to in paragraph 1(3) should be informed by evidence and should take into account the discussions and outcomes of the Member States Working Group on Strengthening WHO Preparedness and Response to Health Emergencies, considering the need for coherence and complementarity between the process of developing the new instrument and the ongoing work under resolution WHA74.7, particularly with regard to implementation and strengthening of the IHR (2005);
(5) that the INB shall submit its outcome for consideration by the Seventy-seventh World Health Assembly [2024], with a progress report to the Seventy-sixth World Health Assembly;
2. REQUESTS the Director-General to support the INB by:
(1) convening its first meeting no later than 1 March 2022, and subsequent meetings at the request of the co-chairs as frequently as necessary;
(2) holding public hearings, in line with standard WHO practice, prior to the second meeting of the INB to inform its deliberations;
(3) facilitating the participation, to the extent the INB so decides, in accordance with relevant Rules of Procedure and resolutions and decisions of the Health Assembly, of representatives of organizations of the United Nations system and other intergovernmental organizations with which WHO has established effective relations, Observers, representatives of non-State actors in official relations with WHO, and of other relevant stakeholders and experts as decided by the INB, recognizing the importance of broad engagement to ensure a successful outcome;
(4) providing the INB with the necessary services and facilities for the performance of its work, including complete, relevant and timely information and advice.
Fifth plenary meeting, 1 December 2021 SSA2/SR/5
::::::
WHO Director-General’s closing remarks at the Special Session of the World Health Assembly –
Speech
01 December 2021
[Excerpts]
…I thank you for the overwhelming support expressed by Member States for the need for a new accord on pandemic preparedness and response.
I welcome the decision you have adopted today, to establish an intergovernmental negotiating body to draft and negotiate a WHO convention, agreement or other international instrument on pandemic prevention, preparedness and response, with a view to adoption under Article 19, or under other provisions of the WHO Constitution as may be deemed appropriate by the intergovernmental negotiating body.
I welcome your commitment to an inclusive, transparent and efficient process, led by Member States and based on consensus.
I welcome your commitment to hold the first meeting of the INB no later than the first of March 2022, and to submit its outcome for consideration to the World Health Assembly in 2024.
And I give you my commitment that the Secretariat will support this process…
I call on all Member States to choose to achieve our targets of vaccinating 40% of the population of every country by the end of this year, and 70% by the middle of next year.
I call on those countries that have already reached 70% to choose to swap your vaccine delivery schedules with COVAX and AVAT, as Switzerland has just done;
I call on all Member States to choose to remove every barrier to scaling up vaccine production, by sharing technology and know-how, and by supporting a waiver of intellectual property rights.
I call on those countries that have promised to donate vaccines to choose to make good on those promises, as urgently as possible.
I call on Member States to choose to fully fund the ACT Accelerator, which needs 23.4 billion U.S. dollars over the next 12 months to get tests, treatments and vaccines to where they are needed most.
I call on every Member State to choose a comprehensive, tailored, layered combination of proven public health and social measures to reduce transmission, reduce the pressure on your health systems, and save lives.
I call on all Member States to focus on strengthening and optimizing clinical pathways, from primary to intensive care, ensuring the right patient get the right care at the right time, and that health workers are supported and protected while doing their life-saving work.
I call on all Member States to intensify and target risk communication, strengthen community engagement, empowerment and support, addressing community concerns, combatting misinformation and building trust.
I call on every Member State to choose to increase surveillance, testing, sequencing and reporting, and to refrain from penalizing countries that do, in compliance with the International Health Regulations.
And I call on all Member States to choose to commit to strengthening your WHO, especially by supporting the proposals of the Working Group on Sustainable Financing.
We are one humanity. We have one planet. We have one health. And we have one WHO. Your WHO.
1 Document SSA2/3.
2 And regional economic integration organizations as appropriate.
WTO – TRIPS Waiver Status
DG calls on members to agree on pandemic response, fisheries subsidies by end-February
2 December 2021
Following last week’s eleventh-hour decision by WTO members to postpone the 12th Ministerial Conference, negotiations are continuing in Geneva, and Director-General Ngozi Okonjo-Iweala today (2 December) urged delegation heads to redouble their efforts to bridge differences. She called on them to conclude agreements on the WTO system’s response to pandemics as well as on curbing harmful fisheries subsidies by the end of February 2022 to pave the way for approval by ministers. “Seven billion people are waiting for us on TRIPS and pandemic response. And 260 million people are waiting for us on fisheries subsidies,” she said.
Speaking to a meeting of delegation heads from all WTO members, the Director-General said the decision to postpone MC12 was “as disappointing as it was necessary”. In addition to the potential health risks from the Omicron variant, travel restrictions introduced in response to its emergence had made it impossible for all ministers to participate on an equitable basis at the conference, which had been set to run from 30 November to 3 December in Geneva.
Just because the ministerial had been postponed did not mean that negotiations had stopped, the Director-General emphasised. She welcomed the ongoing meetings among ambassadors and experts to try to keep bridging gaps they had narrowed over the course of weeks of nearly round-the-clock preparatory negotiations and encouraged them to continue. The challenge for them now was to “recover from last week’s unexpected setback to deliver results for the people we serve”.
DG Okonjo-Iweala said she had spoken to several ministers in recent days, and they all wanted negotiations in Geneva to continue — and to deliver results.
She pointed to a joint letter that the officers of MC12 — Conference Chair Timur Suleimenov, President’s Envoy for International Trade of Kazakhstan, and Vice-Chairs Dan Tehan, Minister for Trade, Tourism and Investment of Australia; Jerome Xavier Walcott, Minister for Foreign Affairs and Foreign Trade of Barbados; and Harriet Ntabazi, Minister of State for Trade of Uganda — had sent to ministers from all WTO members, asking them to empower Geneva representatives to resolve outstanding differences so that ministers can reconvene in March to adopt agreements. That letter called for prioritizing outcomes on pandemic response, including a “sensible solution” on the proposed intellectual property waiver, given the global health situation, as well as on fisheries subsidies, where recent convergence had put a deal in the long-running negotiations within reach.
“If we were ready to make progress this week, we can finish pandemic response and fisheries subsidies by the end of February, and know where we are on agriculture,” the Director-General said.
“The pandemic is raging — people are dying globally in numbers similar to April 2020. The public expects a response from WTO members,” she added. “The new Omicron variant has reminded us once again of the urgency of achieving equitable access to vaccines in every country in the world.”
“It underscores how important it is for us to converge on the WTO’s contribution to achieving this goal, including the TRIPS dimension. I strongly believe that a comprehensive response to the pandemic must include intellectual property,” she said, urging all sides to come together to find a compromise.
Noting that it was not clear whether the epidemiological situation would permit ministers to gather in March, the Director-General said Geneva representatives should seek to prepare clean texts, or at most texts with one outstanding issue to be resolved, for ministers. This would permit them to finalize or bless an outcome even if they have to do so remotely.
“Let us aim to finish pandemic response and fisheries by end February. And let us agree that ministers should be able to finalise them without physical presence. Seven billion people are waiting for us on TRIPS and pandemic response. And 260 million people are waiting for us on fisheries subsidies,” she said.
She urged delegations to advance where possible in the days remaining before the WTO’s winter break and to return early in January with “guidance and instructions for getting us to the finish line”.
Joint Statement on Dose Donations of COVID-19 Vaccines to African Countries
A joint statement from the African Vaccine Acquisition Trust (AVAT), the Africa Centres for Disease Control and Prevention (Africa CDC) and COVAX
29 November 2021| Addis Ababa| Geneva.
Building on lessons learned from our collective experience with dose donations over the past several months, the African Vaccine Acquisition Trust (AVAT), the Africa Centres for Disease Control and Prevention (Africa CDC) and COVAX wish to draw the attention of the international community to the situation of donations of COVID-19 vaccines to Africa, and other COVAX participating economies, particularly those supported by the Gavi COVAX Advance Market Commitment (AMC).
AVAT and COVAX complement each other’s efforts to support African countries to meet their immunisation targets, recognising the global goal of immunising 70% of the African population. Dose donations have been an important source of supply while other sources are stepping up, but the quality of donations needs to improve.
AVAT and COVAX are focused on accelerating access to and rollout of COVID-19 vaccines in Africa. Together we are rapidly expanding supply to the continent, and providing countries with the support to be able to utilise the doses they receive. To date, over 90 million donated doses have been delivered to the continent via COVAX and AVAT and millions more via bilateral arrangements.
However, the majority of the donations to-date have been ad hoc, provided with little notice and short shelf lives. This has made it extremely challenging for countries to plan vaccination campaigns and increase absorptive capacity. To achieve higher coverage rates across the continent, and for donations to be a sustainable source of supply that can complement supply from AVAT and COVAX purchase agreements, this trend must change.
Countries need predictable and reliable supply. Having to plan at short notice and ensure uptake of doses with short shelf lives exponentially magnifies the logistical burden on health systems that are already stretched. Furthermore, ad hoc supply of this kind utilizes capacity – human resources, infrastructure, cold chain – that could be directed towards long-term successful and sustainable rollout. It also dramatically increases the risks of expiry once doses with already short shelf-lives arrive in country, which may have long-term repercussions for vaccine confidence.
Donations to COVAX, AVAT, and African countries must be made in a way that allows countries to effectively mobilise domestic resources in support of rollout and enables long-term planning to increase coverage rates.
We call on the international community, particularly donors and manufacturers, to commit to this effort by adhering to the following standards, beginning from 1 January 2022:
Quantity and predictability: Donor countries should endeavour to release donated doses in large volumes and in a predictable manner, to reduce transaction costs. We acknowledge and welcome the progress being made in this area, but note that the frequency of exceptions to this approach places increased burden on countries, AVAT and COVAX.
Earmarking: These doses should be unearmarked for greatest effectiveness and to support long-term planning. Earmarking makes it far more difficult to allocate supply based on equity, and to account for specific countries’ absorptive capacity. It also increases the risk that short shelf-life donations utilise countries’ cold chain capacity – capacity that is then unavailable when AVAT or COVAX are allocating doses with longer shelf lives under their own purchase agreements.
Shelf life: As a default, donated doses should have a minimum of 10 weeks shelf life when they arrive in-country, with limited exceptions only where recipient countries indicate willingness and ability to absorb doses with shorter shelf lives.
Early notice: Recipient countries need to be made aware of the availability of donated doses not less than 4 weeks before their tentative arrival in-country.
Response times: All stakeholders should seek to provide rapid response on essential information. This includes essential supply information from manufacturers (total volumes available for donation, shelf life, manufacturing site), confirmation of donation offer from donors, and acceptance/refusal of allocations from countries. Last minute information can further complicate processes, increasing transaction costs, reducing available shelf life and increasing risk of expiry.
Ancillaries: The majority of donations to-date do not include the necessary vaccination supplies such as syringes and diluent, nor do they cover freight costs – meaning these have to be sourced separately – leading to additional costs, complexity and delay. Donated doses should be accompanied with all essential ancillaries to ensure rapid allocation and absorption…
Coronavirus [COVID-19] – WHO
Public Health Emergency of International Concern (PHEIC)
https://www.who.int/emergencies/diseases/novel-coronavirus-2019
Weekly Epidemiological and Operational updates
Last update: 3 Dec 2021
Confirmed cases :: 263 563 622 [259 502 031 week ago]
Confirmed deaths :: 5 232 562 [5 183 003 week ago]
Vaccine doses administered: 7 864 123 038 [7 702 859 718 week ago]
::::::
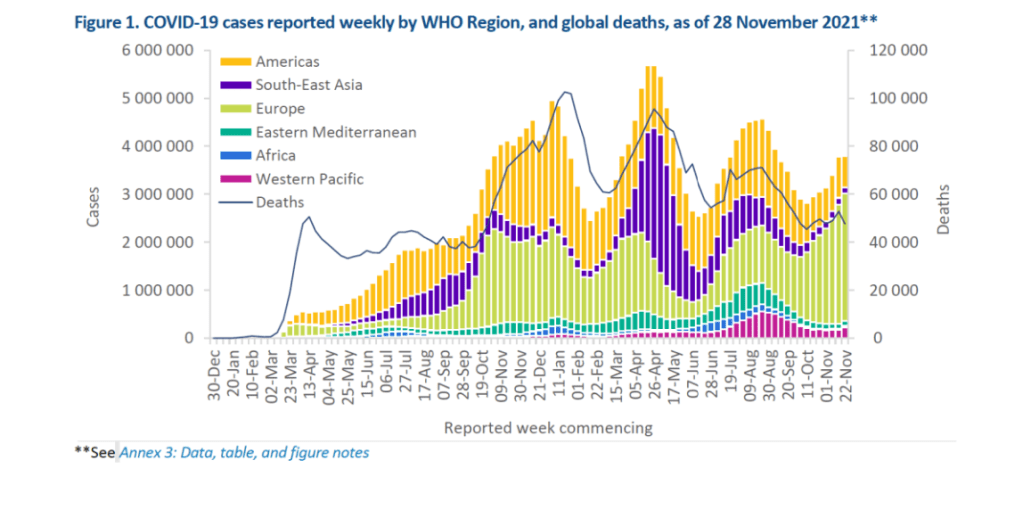
Weekly epidemiological update on COVID-19 – 30 November 2021
WHO
Overview
Globally, weekly COVID-19 case incidence plateaued this week, with nearly 3.8 million confirmed new cases reported during the week of 22-28 November 2021, similar to the previous week’s figures. However, new weekly cases increased in three of the six WHO regions. While global weekly deaths decreased by 10% in the past seven days as compared to the previous week, with over 47 500 new deaths reported, an increase in weekly deaths was reported in two of the six regions. As of 28 November, over 260 million confirmed cases and nearly 5.2 million deaths have been reported globally.
In this edition, we provide:
-Details on the newly designated SARS-CoV-2 Variant of Concern (VOC) Omicron (B.1.1.529)
-Updates on the geographic distribution of VOCs
-A summary of phenotypic characteristics (transmissibility, disease severity, risk of reinfection, and impacts on diagnostics and vaccine performance) of VOCs based on available studies.
WHO Director General Speeches [selected]
https://www.who.int/director-general/speeches
Selected
1 December 2021
Speech
WHO Director-General’s closing remarks at the Special Session of the World Health Assembly – 01 December 2021
[See WHASS above for detail]
1 December 2021
Speech
WHO Director-General’s opening remarks for World AIDS Day 2021 – 1 December 2021
1 December 2021
Speech
WHO Director-General’s opening remarks at the media briefing on COVID-19 – 1 December 2021
Today, WHO’s Member States decided to embark on the process of drafting and negotiating a new convention, agreement or other international instrument on pandemic prevention, preparedness and response.
The emergence of the Omicron variant has understandably captured global attention. At least 23 countries from five of six WHO regions have now reported cases of Omicron, and we expect that number to grow.
We call on all countries to take rational, proportional risk-reduction measures, in keeping with the International Health Regulations.
We must not forget that we are already dealing with a highly transmissible, dangerous variant – the Delta variant, which currently accounts for almost all cases globally. Globally, we have a toxic mix of low vaccine coverage, and very low testing – a recipe for breeding and amplifying variants.
As we mark World AIDS Day, we are reminded that more than 40 years into the global AIDS epidemic, we still have no vaccine and no cure for this disease. Two years into the COVID-19 pandemic, we have not one but many vaccines, and many other effective tools.
30 November 2021
Speech
WHO Director-General’s panel remarks – Trends for a Changing World: COVID-19 – the Importance of universal health coverage and primary health care
30 November 2021
Remarks
WHO Director-General’s opening remarks at the Member State Information Session on the Omicron variant of SARS-CoV-2
29 November 2021
Speech
WHO Director-General’s opening remarks at the Special Session of the World Health Assembly – 29 November 2021
::::::
::::::
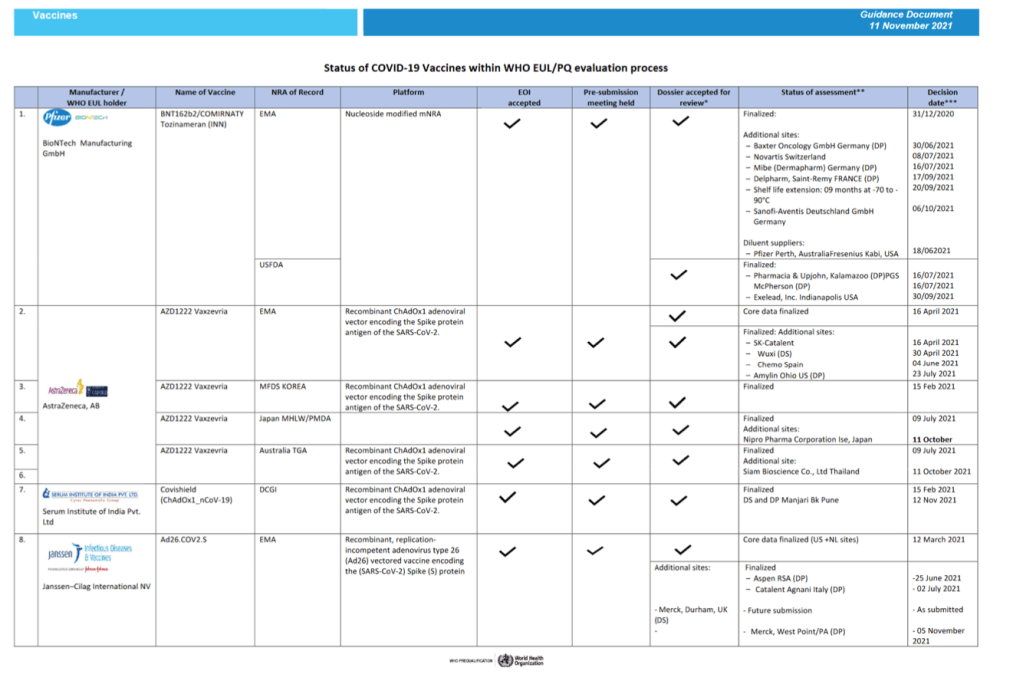
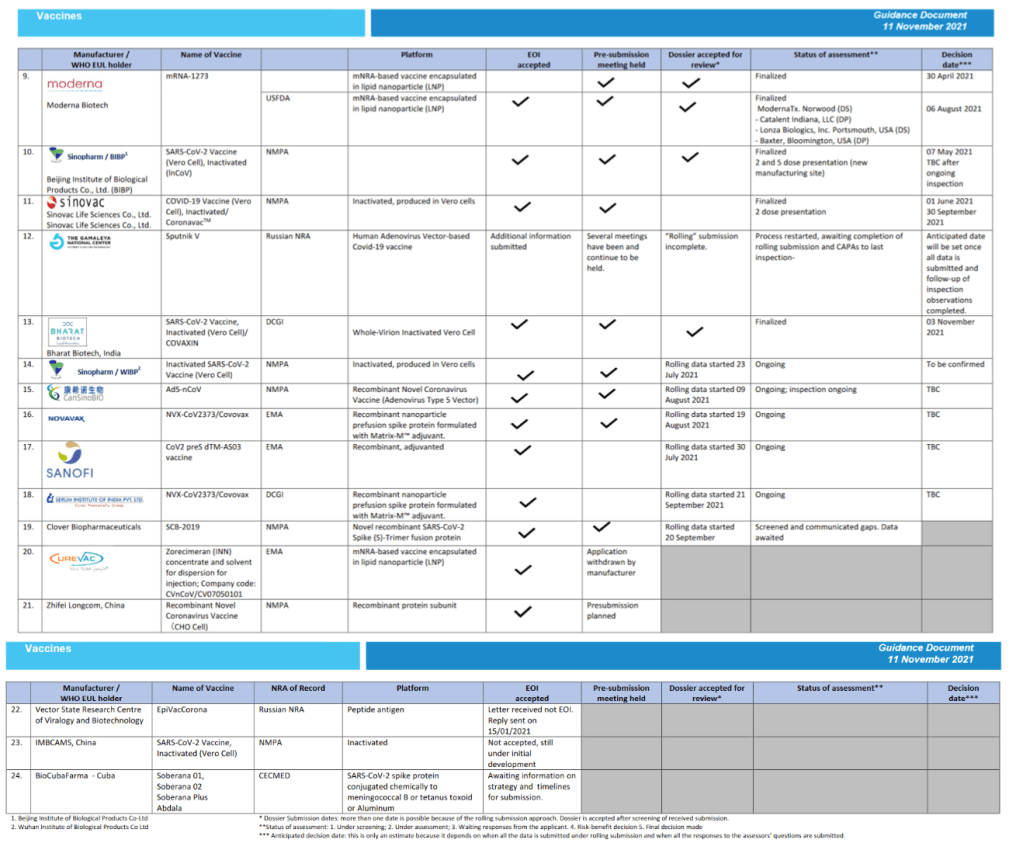
Status of COVID-19 Vaccines within WHO EUL/PQ evaluation process 11 November 2021
For 24 vaccine candidates, presents Manufacturer, Name of Vaccine, NRA of Record, Platform, EOI Accepted Status, Pre-submission Meeting Held Status, Dossier Accepted for Review, Status of Assessment; Anticipated/Completed Decision Date
[Full scale view available at title link above]
COVID Vaccine Developer/Manufacturer Announcements
[relevant press releases/announcement from organizations from WHO EUL/PQ listing above]
AstraZeneca
Press Releases – No new digest announcements identified
Bharat Biotech
Press Releases – No new digest announcements identified
BioCubaFarma – Cuba
Últimas Noticias – [Website not responding at inquiry; receiving 403-Forbidden]
CanSinoBIO
News – [Website not responding at inquiry]
Clover Biopharmaceuticals – China
News – No new digest announcements identified
Curevac [Bayer Ag – Germany]
News – No new digest announcements identified
Gamaleya National Center
Latest News and Events – No new digest announcements identified [See Russia/RFID below]
IMBCAMS, China
Home – No new digest announcements identified
Janssen/JNJ
Press Releases
Dec 03, 2021
United States
Janssen to Showcase Progress on Respiratory Syncytial Virus (RSV) Vaccine Candidate at ESWI 2021
Nov 29, 2021
United States
Johnson & Johnson to Evaluate Its COVID-19 Vaccine Against New Omicron COVID-19 Variant
Since the emergence of the COVID-19 pandemic, Johnson & Johnson (the Company) has been closely monitoring newly emerging COVID-19 variants. In collaboration with academic groups in South Africa and around the world, the Company has been evaluating the effectiveness of its COVID-19 vaccine across variants, including now the new and rapidly spreading Omicron variant. The Company is testing blood serum from participants in completed and ongoing booster studies to look for neutralizing activity against the Omicron variant. In addition, the Company is pursuing an Omicron-specific variant vaccine and will progress it as needed…
Moderna
Press Releases
December 1, 2021
Moderna Announces Supply Agreement with the UK for Additional 60 Million Doses of Moderna’s COVID-19 Vaccine in 2022 and 2023
Agreement includes 29 million doses for delivery in 2022 and 31 million doses for delivery in 2023
Novavax
Press Releases
Novavax Statement on Omicron Variant Response
Dec 2, 2021
Novavax is rapidly responding to the emergence of the latest potential threat of the SARS-CoV-2 Omicron (B.1.1.529) variant of concern (VoC). The company is executing a two-pronged variant strategy.
First, Novavax is evaluating its vaccine against the Omicron variant, as the company has done for previous variants including Alpha, Beta and Delta. Second, Novavax has initiated development of an Omicron-specific vaccine construct…
Pfizer
Recent Press Releases – No new digest announcements identified
Sanofi Pasteur
Press Releases
Vaccines Investor Event: Sanofi reiterates confidence in strong growth outlook and showcases pipeline of innovative vaccine candidates
December 01 2021
Broad pipeline of innovative vaccines, with a total of 10 candidates planned to advance into the clinic by 2025, six of which will leverage the recently established mRNA Center of Excellence
Serum Institute of India
NEWS & ANNOUNCEMENTS – No new digest announcements identified
Sinopharm/WIBPBIBP
News – No new digest announcements identified
Sinovac
Press Releases
Instituto Butantan and SINOVAC will hold ‘CoronaVac® Symposium’ international event about efficacy and security of CoronaVac®
2021/11/2
Vector State Research Centre of Viralogy and Biotechnology
Home – No new digest announcements identified
Zhifei Longcom, China
[Anhui Zhifei Longcom Biologic Pharmacy Co., Ltd.]
[No website identified]
::::::
GSK
Press releases for media
02 December 2021
GSK welcomes Gavi decision to fund the roll out of malaria vaccines for children
Gavi to provide funding for procurement and introduction of malaria vaccines into child immunisation programmes in Gavi eligible countries.
02 December 2021
GSK and the University of Oxford launch new Oxford-GSK Institute to harness advanced technology and unravel mechanisms of disease
:: Major new collaboration aims to deepen understanding of complex diseases such as Alzheimer’s and Parkinson’s, and increase drug discovery and development success rates
:: New Institute to be based at Oxford’s Nuffield Department of Medicine
02 December 2021
Preclinical data demonstrate sotrovimab retains activity against key Omicron mutations, new SARS-CoV-2 variant
:: Data to be confirmed by further in vitro pseudo-virus testing
30 November 2021
GSK appoints Phil Dormitzer M.D., Ph.D., as Global Head of Vaccines R&D
Phil Dormitzer M.D., Ph.D., will join the company as Global Head of Vaccines R&D on 3 December 2021
Merck
News releases
U.S. FDA Accepts for Priority Review the Supplemental Biologics License Application for Merck’s VAXNEUVANCE™ (Pneumococcal 15-valent Conjugate Vaccine) for Use in Infants and Children
December 3, 2021
Novartis
News – No new digest announcements identified
SK Biosciences
Press releases – No new digest announcements identified
Valneva
Press Releases
December 3, 2021
Valneva Comments on COV-Boost Clinical Trial Data
December 2, 2021
Valneva Confirms Initiation of Rolling Review with EMA and Provides Updates on its COVID-19 Vaccine Program VLA2001
November 29, 2021
Valneva and IDT Biologika Announce Collaboration for Production of Inactivated COVID-19 Vaccine VLA2001
UNICEF COVID-19 Vaccine Market Dashboard :: Agreements Table Accessed 4 Dec 2021
An overview of information collected from publicly announced bilateral and multilateral supply agreements [no new agreements since 10/22/2021 reported]

IMF-WHO Vaccine Supply Forecast Dashboard
Updated on 22 November 2021
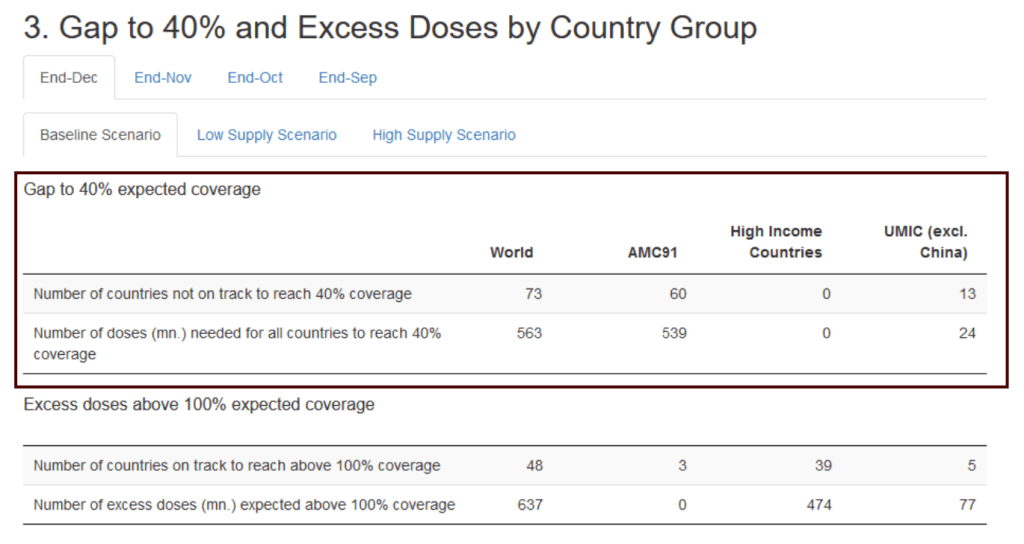
Many countries are not on track to meet the 40% vaccine coverage target. To make progress towards this target, we must identify: (1) Where and in which countries are the gaps?, and (2) How can gaps the be addressed?
The IMF-WHO COVID-19 Vaccine Supply Forecast Dashboard estimates expected month vaccine supply until the end of 2021 by country, product and channel. The tracker builds on the work of the IMF Staff Discussion Note – A Proposal to End the COVID-19 Pandemic (Agarwal and Gopinath, 2021).
Duke – Launch and Scale Speedometer
The Race for Global COVID-19 Vaccine Equity
A flurry of nearly 200 COVID-19 vaccine candidates are moving forward through the development and clinical trials processes at unprecedented speed; more than ten candidates are already in Phase 3 large-scale trials and several have received emergency or limited authorization. Our team has aggregated and analyzed publicly available data to track the flow of procurement and manufacturing and better understand global equity challenges. We developed a data framework of relevant variables and conducted desk research of publicly available information to identify COVID vaccine candidates and status, deals and ongoing negotiations for procurement and manufacturing, COVID burden by country, and allocation and distribution plans. We have also conducted interviews with public officials in key countries to better understand the context and challenges facing vaccine allocation and distribution
[accessed 24 July 2021]
See our COVID Vaccine Purchases research
See our COVID Vaccine Manufacturing research
See our COVID Vaccine Donations & Exports research
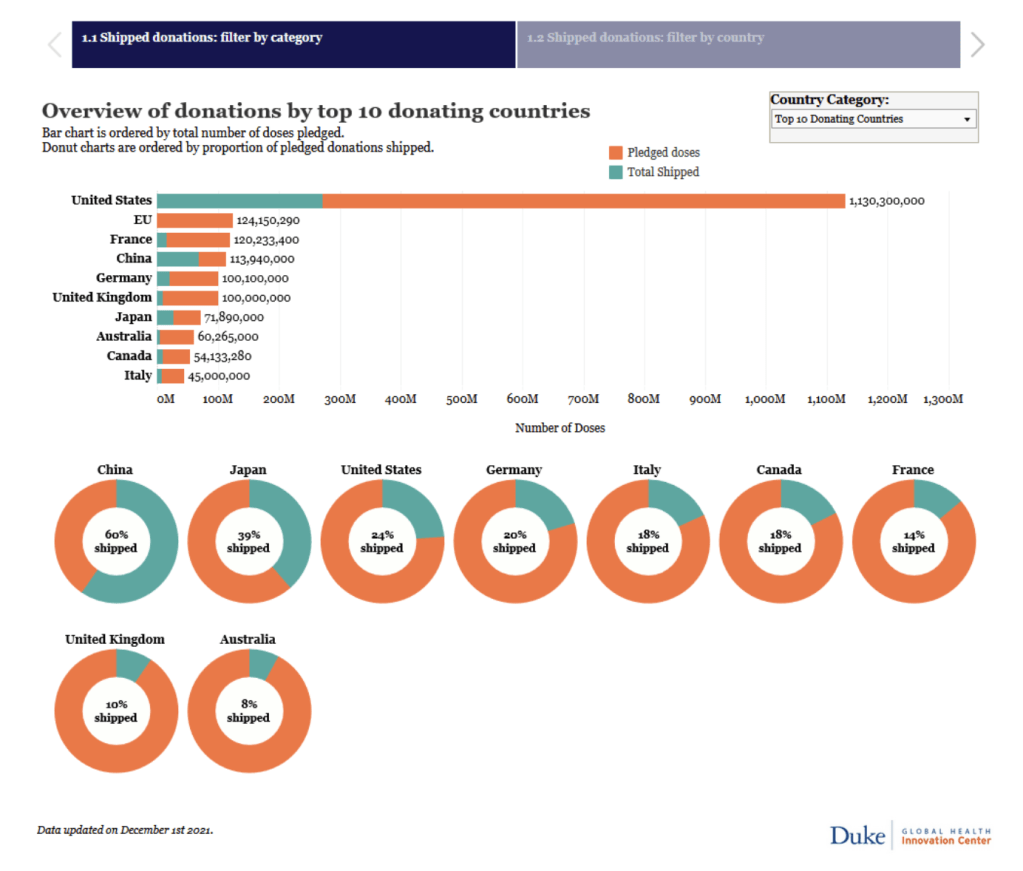
Global Dashboard on COVID-19 Vaccine Equity
The Dashboard is a joint initiative of UNDP, WHO and the University of Oxford with cooperation across the UN system, anchored in the SDG 3 Global Action Plan for Healthy Lives and Well-being for All.
Dashboard on Vaccine Equity [accessed 4 Dec 2021]: https://data.undp.org/vaccine-equity/
See also visualization on Vaccine Access and Vaccine Affordability
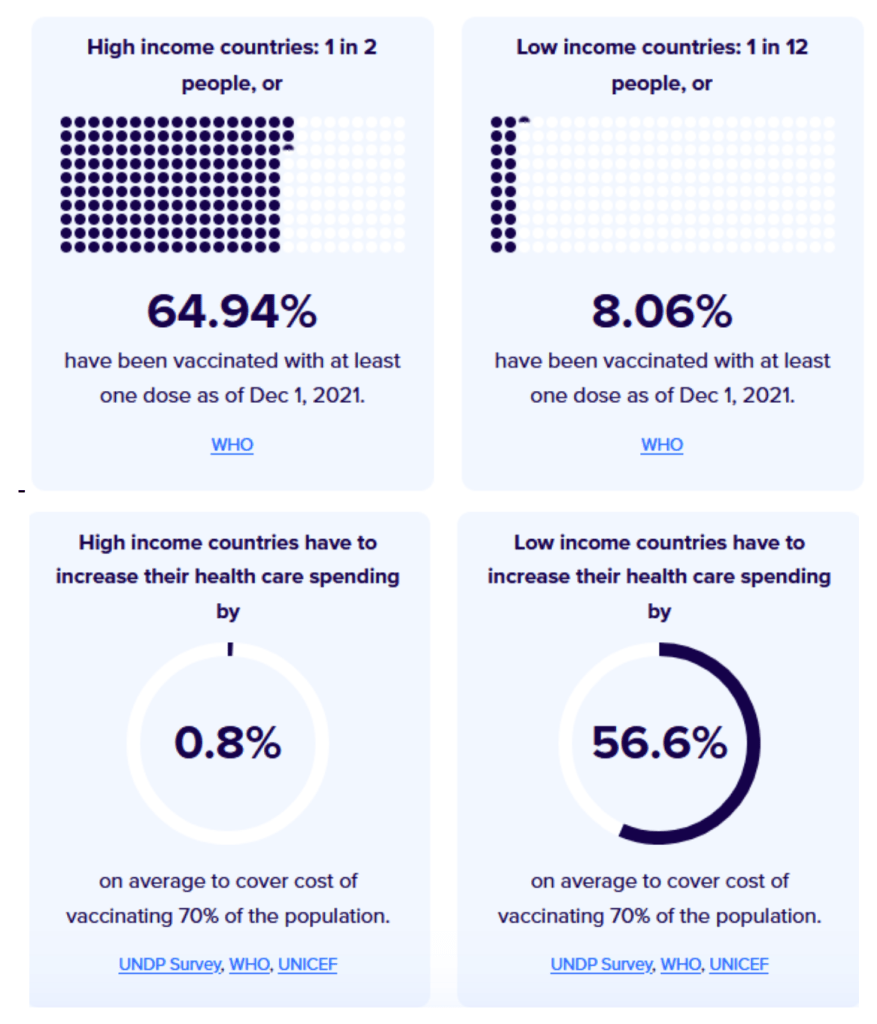
COVID Vaccines – OCHA:: HDX
COVID-19 Data Explorer: Global Humanitarian Operations
COVID-19 Vaccine Roll-out
4 Dec 2021 | COVAX (WHO,GAVI,CEPI), UNDESA, Press Reports | DATA
Global COVID-19 Figures: 263M total confirmed cases; 5.2M total confirmed deaths
Global vaccines administered: 8.078B
Number of Countries: 30 [30 week ago]
COVAX Allocations Round 4-9 (Number of Doses): 220M [220M week ago]
COVAX Delivered (Number of Doses): 180M [170M week ago]
Other Delivered (Number of Doses): 300M [280M week ago]
Total Delivered (Number of Doses): 480M [450M week ago]
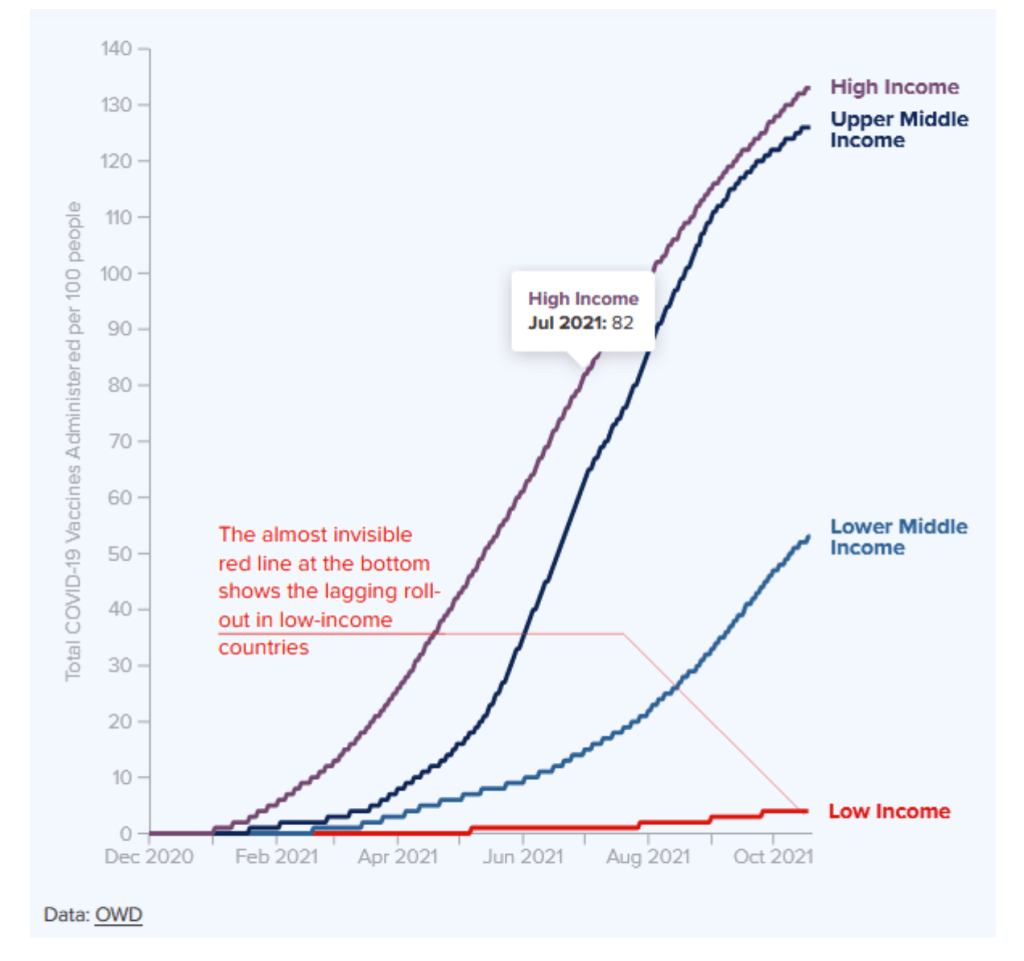
Multilateral Leaders Task Force on COVID-19 [IMF, World Bank Group, WHO, WTO]
https://data.covid19taskforce.com/data
A global effort to help developing countries access and deliver COVID-19 vaccines, testing, and therapeutics, as they work to end the pandemic and boost economic recovery.
The International Monetary Fund, World Bank Group, World Health Organization and World Trade Organization have joined forces to accelerate access to COVID-19 vaccines, therapeutics and diagnostics by leveraging multilateral finance and trade solutions, particularly in low- and middle-income countries.
Website accessed 4 Dec 2021: https://data.covid19taskforce.com/data The global view below is complemented by country-specific dashboards here.
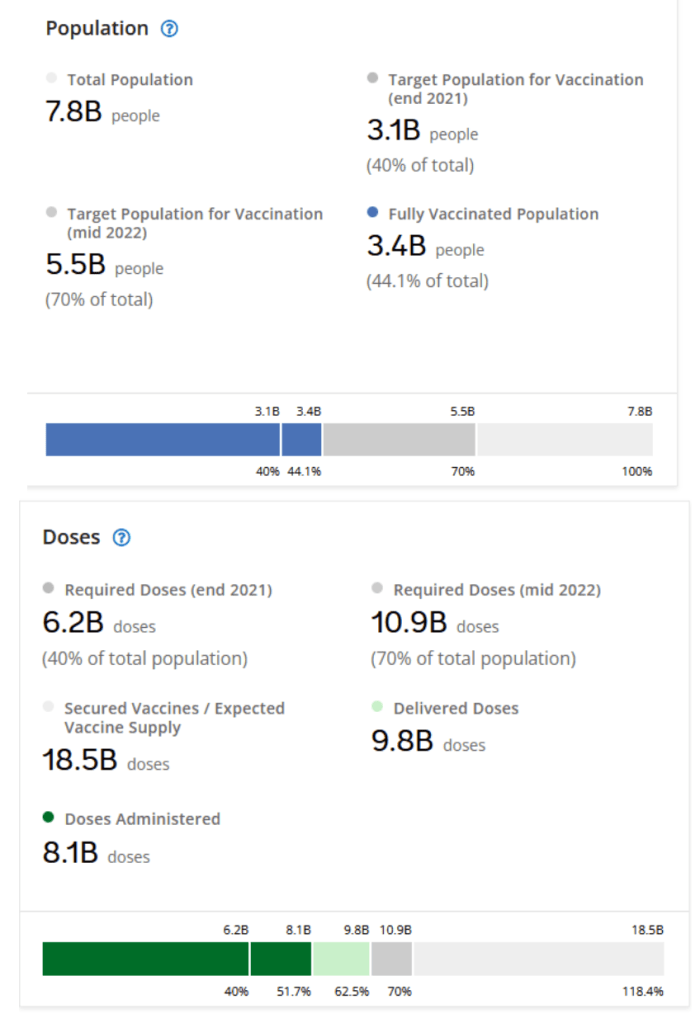
Our World in Data
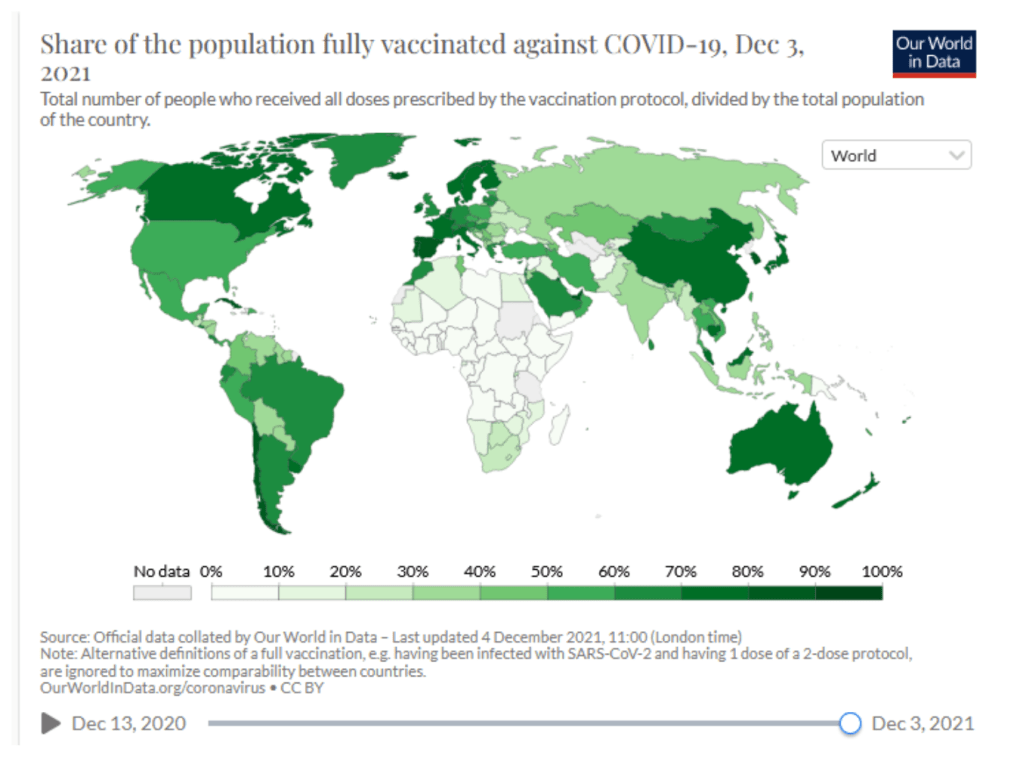
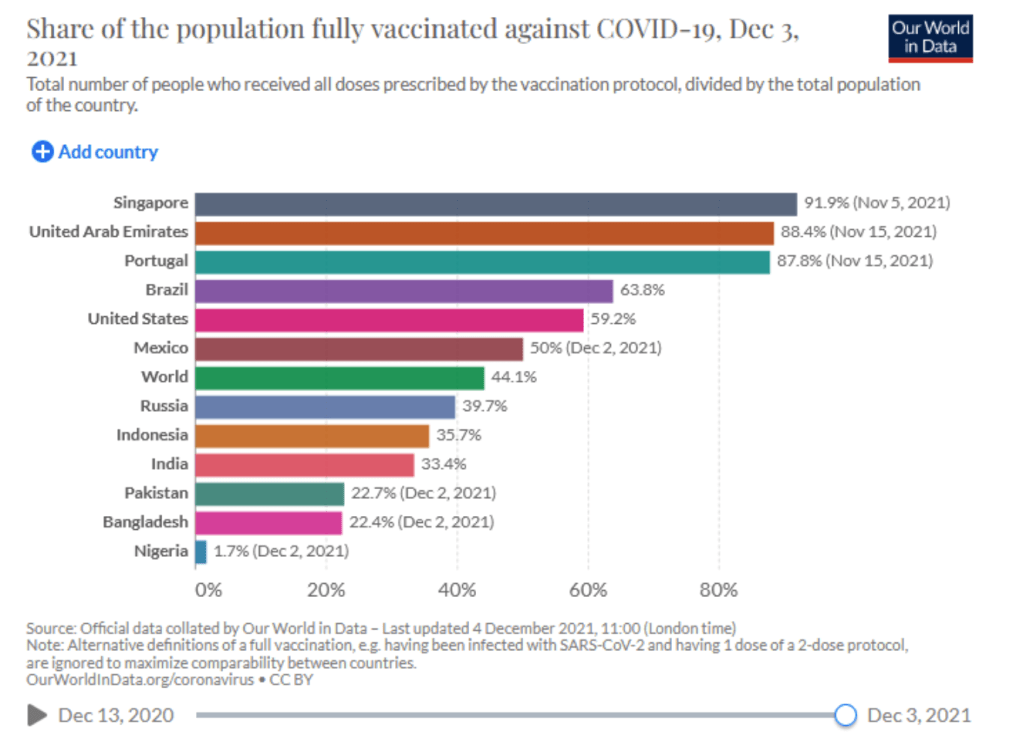
Coronavirus (COVID-19) Vaccinations [Accessed 4 Dec 2021]
:: 54.9% of the world population has received at least one dose of a COVID-19 vaccine.
:: 8.14 billion doses have been administered globally, and 34.41 million are now administered each day.
:: Only 6.2% of people in low-income countries have received at least one dose.
U.S.: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
FDA
Press Announcements
December 3, 2021 – FDA Expands Authorization of Two Monoclonal Antibodies for Treatment and Post-Exposure Prevention of COVID-19 to Younger Pediatric Patients, Including Newborns
November 30, 2021 – Coronavirus (COVID-19) Update: November 30, 2021
Vaccines and Related Biological Products Advisory Committee– FDA
https://www.fda.gov/advisory-committees/blood-vaccines-and-other-biologics/vaccines-and-related-biological-products-advisory-committee
No meetings on calendar
::::::
White House [U.S.]
Briefing Room – Selected Major COVID Announcements
Press Briefing by White House COVID-19 Response Team and Public Health Officials
December 03, 2021 Press Briefings
Remarks by President Biden on the COVID-19 Winter Plan
December 02, 2021 Speeches and Remarks
Background Press Call by Senior Administration Officials on New Actions to Protect Americans Against the Delta and Omicron Variants This Winter
December 02, 2021 Press Briefings
President Biden Announces New Actions to Protect Americans Against the Delta and Omicron Variants as We Battle COVID-19 this Winter
December 02, 2021 Statements and Releases
Statement from White House COVID-19 Response Coordinator Jeff Zients on First Case of the Omicron COVID-19 Variant in the United States
December 01, 2021• Statements and Releases
Press Briefing by White House COVID-19 Response Team and Public Health Officials
November 30, 2021 • Press Briefings
Readout of President Biden’s Meeting with COVID-19 Response Team on the Omicron Variant
November 28, 2021 Statements and Releases
U.S. Department of State
https://www.state.gov/coronavirus/releases/
Media Notes
Press Statement
Gayle Smith’s Service as the Coordinator for Global COVID-19 Response and Health Security
Antony J. Blinken November 30, 2021
Europe: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
European Medicines Agency
News & Press Releases
News: ICMRA and WHO map out flexibilities used by regulators to respond to the COVID-19 pandemic (new)
Last updated: 03/12/2021
[See Milestones above for detail]
News: Meeting highlights from the Pharmacovigilance Risk Assessment Committee (PRAC) 29 November – 2 December 2021 (new)
PRAC, Last updated: 03/12/2021
PRAC update on risk of myocarditis and pericarditis with mRNA vaccines
News: EMA starts rolling review of Valneva’s COVID-19 vaccine (VLA2001) (new)
CHMP, Last updated: 02/12/2021
News: Increase in manufacturing capacity for COVID-19 vaccine from AstraZeneca (new)
CHMP, Last updated: 01/12/2021
::::::
European Centre for Disease Prevention and Control
https://www.ecdc.europa.eu/en
Latest Updates
Publication & Data
Epidemiological update: Omicron variant of concern (VOC) – data as of 4 December 2021 (12.00)
Epidemiological update
4 Dec 2021
As of 4 December and since 3 December 2021, 58 additional SARS-CoV-2 Omicron variant of concern (VOC) cases have been confirmed in the European Union and European Economic Area (EU/EEA), contributing to an overall total of 167 confirmed cases so far.
Cases have been reported by 17 countries: Austria (10), Belgium (6), Czechia (1), Denmark (18), Finland (1), France (12, including 2 in Reunion), Germany (15), Greece (1), Iceland (7), Ireland (1), Italy (9), Luxembourg (1), the Netherlands (18), Norway (19), Portugal (34), Spain (7) and Sweden (7) according to information from public sources. One new EU/EEA country (Luxembourg) has reported the Omicron variant and a number of probable cases are currently under investigation in several countries.
The majority of confirmed cases have a history of travel to countries in Africa, with some having taken connecting flights at other locations between Africa and Europe. Several EU/EEA countries (Belgium, Germany, Spain) detected cases without an epidemiological link to areas where community transmission of the Omicron variant is documented or presumed. This indicates that undetected community transmission could be ongoing in these countries. Finland reported cases with travel history within the EU/EEA (Sweden, Denmark).
All cases for which there is available information on severity were either asymptomatic or mild. No deaths have been reported among these cases so far. These figures should be assessed with caution as the number of confirmed cases is too low to understand if the disease clinical spectrum of Omicron differs from that of previously detected variants.
::::::
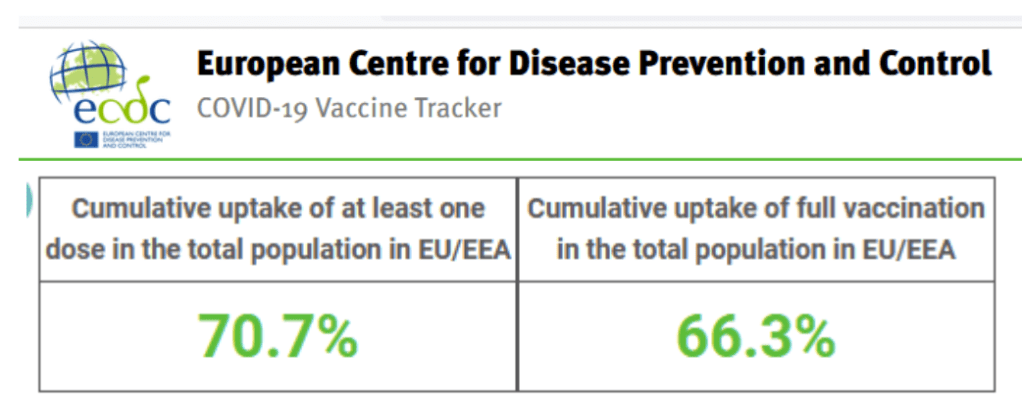
Accessed 4 Dec 2021
https://vaccinetracker.ecdc.europa.eu/public/extensions/COVID-19/vaccine-tracker.html#uptake-tab
European Commission
https://ec.europa.eu/commission/presscorner/home/en
Statement 1 December 2021
Statement by President von der Leyen on facing current and new COVID-19 challenges
Press release 1 December 2021
Commission reiterates calls to step up vaccination, rapid deployment of boosters, vigilance and rapid reaction to Omicron variant
The Commission is today putting forward a common and coordinated EU approach to address effectively the challenges from the resurgence of COVID-19 in many Member States this autumn.
There are rapidly rising case numbers and a renewed pressure on hospitals, which calls for urgent and determined action. The new potential threat from the Omicron variant is adding to these concerns, and underlines the importance of tackling the pandemic to progress towards long-term health security, both in the EU and globally…
Press release 30 November 2021
European Health Union: Commission welcomes agreement on a stronger European Centre for Disease Prevention and Control
Last night, the European Parliament and the Council reached an agreement on a reinforced role for the European Centre for Disease Prevention and Control (ECDC). The revised ECDC mandate will allow the agency to take a stronger role in supporting the EU and its Member States in the prevention and control of communicable disease threats and improve European preparedness for future health challenges…
Statement 26 November 2021
Press statement by President von der Leyen on the new COVID variant
Commission President von der Leyen has given a press statement on the new COVID variant.
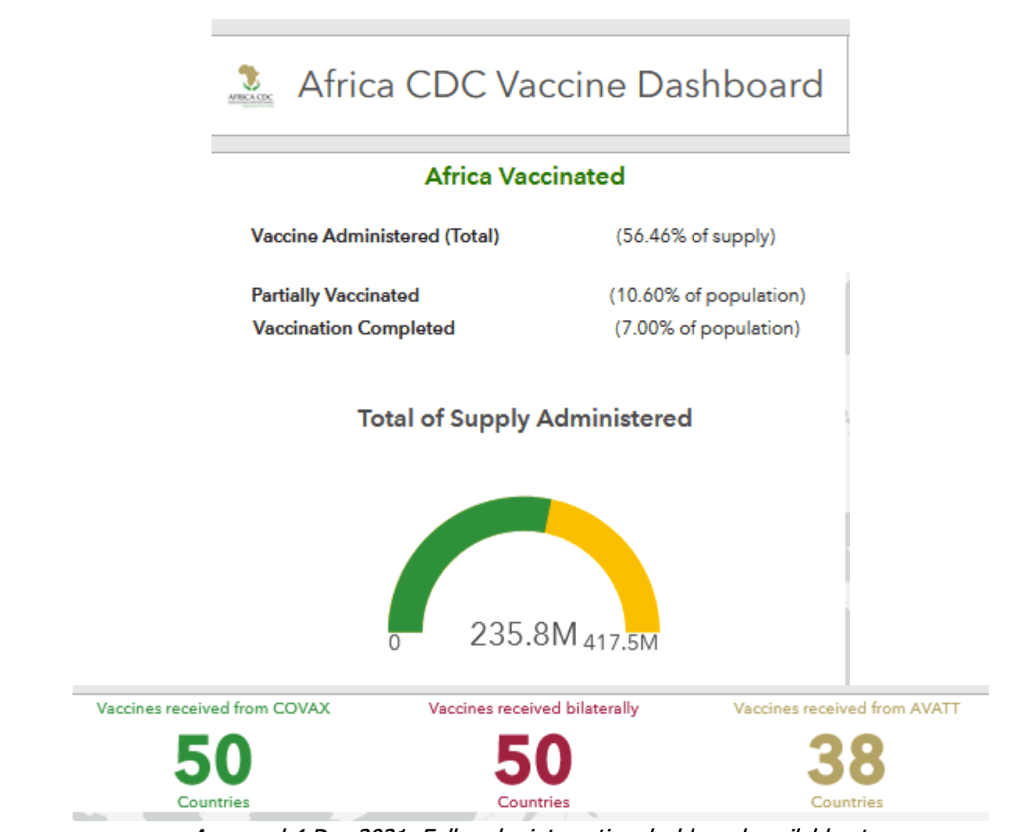
Africa: COVID-19 – Announcements/Regulatory Actions/Deployment
Accessed 4 Dec 2021. Full scale, interactive dashboard available at:
https://africacdc.org/covid-19-vaccination/
Russia: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
Russia: Sputnik V – “the first registered COVID-19 vaccine”
https://sputnikvaccine.com/newsroom/pressreleases/
Press Releases
RDIF signs agreements with partners in Vietnam to expand production of the Russian Sputnik V vaccine
Press release, 01.12.2021
The one-shot Sputnik Light vaccine authorized in UAE as a universal booster shot
Press release, 30.11.2021
Statement of RDIF and the Gamaleya Institute on Omicron variant of COVID
Press release, 29.11.2021
“…The Gamaleya Institute believes both Sputnik V and Sputnik Light will neutralize the latest Omicron variant and has started the necessary studies.
Nonetheless the Gamaleya Institute, based on existing protocols of immediately developing vaccine versions for variants of concern, has already begun developing the new version of Sputnik vaccine adapted to Omicron. In an unlikely case such modification is needed, the new Sputnik Omicron version can be ready for mass-scale production in 45 days. Several hundred million Sputnik Omicron boosters can be provided to international markets already by Feb 20, 2022 with over 3 billion doses available in 2022.”
India: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
Ministry of Health and Family Welfare
https://www.mohfw.gov.in/
03.12.2021 FAQs on SARS-CoV-2 Variant-Omicron
Government of India – Press Information Bureau
Latest Press Releases
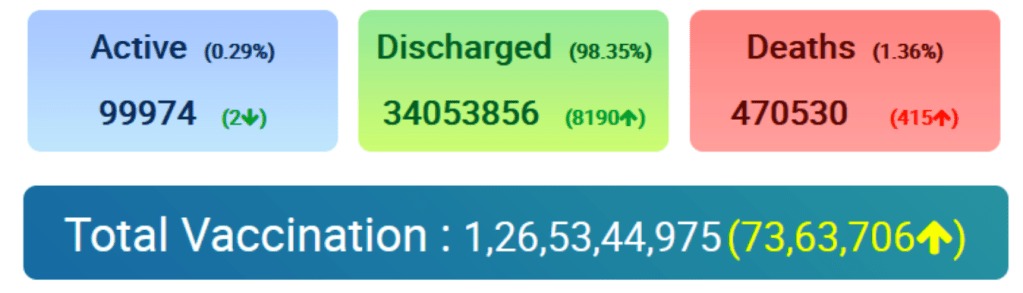
COVID-19 Vaccination Update – Day 323
:: India’s cumulative vaccination coverage crosses 127 crore landmark milestone
:: More than 93 lakh Vaccine doses administered today till 7 pm
Posted On: 04 DEC 2021 8:09PM by PIB Delhi
Indian Council for Medical Research (ICMR)
https://www.icmr.gov.in/media.html
Press Releases
No new digest content identified.
China: COVID-19 Vaccines – Announcements/Regulatory Actions/Deployment
National Health Commission of the People’s Republic of China [to 4 Dec 2021]
http://en.nhc.gov.cn/
News
Dec 4: Daily briefing on novel coronavirus cases in China
On Dec 3, 31 provincial-level regions and the Xinjiang Production and Construction Corps on the Chinese mainland reported 90 new cases of confirmed infections.
China has provided over 1.8b COVID-19 vaccine doses to international community
2021-12-03
BEIJING – To date, China has provided over 1.8 billion doses of COVID-19 vaccines to more than 120 countries and international organizations.
China has delivered on its promise to make vaccines global public goods, and made continued efforts to improve vaccine accessibility and affordability in developing countries.
China has pledged to strive to provide a total of 2 billion vaccine doses to the world by the end of this year, and said that in addition to donating $100 million to COVAX, it would donate 100 million vaccine doses to other developing countries in the course of this year.
As of Nov 12, China has supplied more than 70 million vaccine doses to COVAX and is conducting joint vaccine production with 19 developing countries…
China pushing development of vaccine against Omicron
2021-12-03
China is rapidly advancing development of COVID-19 vaccines targeting the Omicron variant, a health official said during an interview with China Central Television on Thursday.
Zheng Zhongwei, a National Health Commission official who heads China’s COVID-19 vaccine development task force, said preliminary analysis suggests that the new strain is more transmissible than the Delta variant, but it is too early to determine if the Omicron variant would diminish the efficacy of existing vaccines.
“We think the majority of vaccines remain effective,” he said. “Despite the possibility of some degree of immunity escape, our initial finding is that they still have marked effects on preventing severe diseases and deaths.”…
China sticks to its strategy in COVID-19 response: official
2021-12-02
BEIJING — A health official has emphasized the importance of committing to the current strategy in China to clear COVID-19 infections in a timely manner as highly contagious variants of the virus continue to rage around the world.
Since mid-October, the latest COVID-19 resurgence in China has spread to more than 20 provincial-level regions. Most regions managed to contain the outbreak within an incubation period of about 14 days, said Ma Xiaowei, head of the National Health Commission, in a recent interview with Xinhua.
“We have been thinking, especially during the process of coping with the spread of the Delta variant, how to control the epidemic with more effective measures, lower costs and at faster speeds to minimize its impact on economic and social development and people’s lives,” Ma said.
He added that the government has also noticed that the epidemic response in some countries, which have not implemented strict epidemic control measures, has not been satisfactory. There have been relapses that severely impacted people’s lives, health and the economy and society overall.
As a result, China has been focusing on improving epidemic prevention and early detection, he noted…
Emergencies
POLIO
Public Health Emergency of International Concern (PHEIC)
https://polioeradication.org/polio-today/polio-now/this-week/
Polio this week as of 01 December 2021
Summary of new WPV and cVDPV viruses this week (AFP cases and ES positives):
Pakistan: one WPV1 positive environmental sample
Central African Republic: one cVDPV2 positive environmental sample
Nigeria: seven cVDPV2 positive environmental samples
Uganda: one cVDPV2 positive environmental sample
::::::
::::::
WHO/OCHA Emergencies
Health emergencies list – WHO
“The health emergencies list details the disease outbreaks, disasters and humanitarian crises where WHO plays an essential role in supporting countries to respond to and recover from emergencies with public health consequences.”
Afghanistan crisis [Last apparent update: 18 Oct 2021]
Crisis in Northern Ethiopia [Last apparent update: 1 June 2021]
Ebola outbreak, Democratic Republic of the Congo, 2021 [Last apparent update: 17 Aug 2021]
Ebola outbreak outbreak, N’Zerekore, Guinea, 2021 [Last apparent update: 17 Aug 2021]
Coronavirus disease (COVID-19) pandemic [See COVID above]
Ebola outbreak, Equateur Province, Democratic Republic of the Congo, 2020
[Last apparent update: 17 Aug 2021]
Ebola outbreak, North Kivu, Ituri, Democratic Republic of the Congo, 2018 – 2020
[Last apparent update: 17 Aug 2021]
Ebola outbreak, Democratic Republic of the Congo, 2018 [Last apparent update: 24 July 2018]
Yemen crisis [Last apparent update: 12 February 2021]
Syria crisis [Last apparent update: 18 June 2021]
Somalia crisis [Last apparent update: 24 March 2018]
Nigeria crisis [Last apparent update: 1 Oct 2021]
Ebola outbreak, Democratic Republic of the Congo, 2017 [Last apparent update: 17 Aug 2021]
Zika virus disease outbreak, 2015-2016 [Last apparent update: 24 Jan 2020]
Ebola outbreak: West Africa, 2014-2016 [Last apparent update: 17 Aug 2021]
Iraq crisis [Last apparent update: 9 Jan 2008]
South Sudan crisis [Last apparent update: 23 Sep 2020]
Avian influenza A (H7N9) virus outbreak [Last apparent update: 13 September 2021]
Middle East respiratory syndrome (MERS-CoV) outbreak [Last apparent update: 8 July 2019]
Influenza A (H1N1) virus, 2009-2010 pandemic [Last apparent update: 10 Aug 2010]
::::::
UN OCHA – Current Emergencies
Current Corporate Emergencies
Afghanistan
[No new digest content identified]
Northern Ethiopia
Ethiopia – Northern Ethiopia Humanitarian Update Situation Report, 2 Dec 2021
::::::
::::::
WHO & Regional Offices [to 4 Dec 2021]
https://www.who.int/
Selected News, Statements
3 December 2021
Procurement
Request for proposal: Technical support to SAGE executive secretariat
3 December 2021
Departmental news
South-East Asia: Influenza virus sharing in a time of COVID-19
3 December 2021
Departmental news
Global and regional support for the 2021–2022 influenza season
3 December 2021
Departmental news
Critical evidence to strengthen seasonal vaccination in the Americas
2 December 2021
Departmental news
Botswana is first country with severe HIV epidemic to reach key milestone in the elimination of mother-to-child HIV transmission
1 December 2021
Joint News Release
World AIDS Day 2021 – Step up, be bold, end AIDS, end inequalities and end pandemics
1 December 2021
Joint News Release
Tripartite and UNEP support OHHLEP’s definition of “One Health”
1 December 2021
News release
World Health Assembly agrees to launch process to develop historic global accord on pandemic prevention, preparedness and response
1 December 2021
Departmental news
It’s time for action on inequality to end TB and AIDS
30 November 2021
Departmental news
WHO Pandemic Ethics & Policy Summit
29 November 2021
Joint News Release
Joint Statement on Dose Donations of COVID-19 Vaccines to African Countries
::::::
Weekly Epidemiological Record, Vol. 96, No. 48, pp. 585–596 3 December 2021
:: Schistosomiasis and soil-transmitted helminthiases: progress report, 2020
::::::
WHO Regional Offices
Selected Press Releases, Announcements
WHO African Region AFRO
:: Africa steps up Omicron variant detection as COVID-19 cases rise in southern Africa 02 December 2021
:: Botswana is first country with severe HIV epidemic to reach key milestone in the elimination of mother-to-child HIV transmission 02 December 2021
:: WHO stands with African nations and calls for borders to remain open 28 November 2021
WHO Region of the Americas PAHO
No new digest content identified
WHO South-East Asia Region SEARO
:: 1 December 2021 Statement- World AIDS Day
:: 27 November 2021 News release
WHO asks countries in South-East Asia Region to be vigilant as cases surge globally and new Variant of Concern is detected
WHO European Region EURO
:: What you need to know about the new Omicron COVID-19 variant 03-12-2021
:: Greece: civil society reduces barriers to health and education for young people with disabilities during the pandemic 03-12-2021
:: WHO/Europe high-level experts complete mission in Turkmenistan to strengthen health system preparedness 01-12-2021
:: Urgent need to take preventive measures in the fight against COVID-19 the focus of WHO/Europe advocacy campaign 30-11-2021
:: Building trust helps United Kingdom chart a path towards eliminating AIDS by 2030 30-11-2021
WHO Eastern Mediterranean Region EMRO
:: World AIDS Day: Reaching more people with HIV services and reducing inequalities 1 December 2021
WHO Western Pacific Region
:: 3 December 2021 Regional Director’s remarks at the virtual press conference on 3 December 2021
::::::
WHO Events
https://www.who.int/news-room/events/2
[Selected]
Extraordinary meeting of the Strategic Advisory Group of Experts on Immunization (SAGE) – 7 December 2021
7 December 2021
This extraordinary virtual meeting for the Strategic Advisory Group of Experts on Immunization (SAGE) will take place on Tuesday 7 December 2021 to propose recommendations to WHO on the use of COVID-19 vaccine(s).
Agenda: https://cdn.who.int/media/docs/default-source/immunization/sage/2021/december/sage-agenda-7dect2021-virtual-draft-rev26nov.pdf?sfvrsn=f2b71e9f_10
Building stronger, sustainable, equitable societies for pandemic prevention and for navigating the response to the COVID-19 crisis
7 December 2021 12:00 – 13:00 CET
Join us to launch the new WHO evidence brief, “COVID-19 and the social determinants of health and health equity”, and to discuss the implications of the impacts of the social determinants of health equity for pandemic prevention, preparedness, response, and recovery.
* Tuesday 7 December 2021 * 12:00-13:00 Central European Time *
* Join via Zoom * https://who.zoom.us/j/96260096854
Password: Yt@W21bZ
::::::
New WHO Publications
https://www.who.int/publications/i
Selected Titles
1 December 2021
Global vaccine safety blueprint 2.0 (GVSB2.0) 2021-2023
Overview
This Global Vaccines Safety Blueprint 2.0 (GVSB2.0) was developed through a step-wise consensus process to define the strategic priorities and objectives in the area of vaccines safety for 2021 to 2023 and beyond. The overall vision, goals and operating principles for a vaccine safety monitoring system described in the GVSB2.0 are equally relevant for medicines and for building product-agnostic pharmacovigilance systems in countries. Where possible, we need to build common systems and approaches that are smart, product-agnostic, address common needs and advance the principles of work-sharing and reliance to take into consideration competing priorities in resource-limited settings. For example, how we collect and manage data (databases) may not be very different between vaccines and medicines; on the other hand, the principles of benefit risk assessment and how risks are communicated requires specialized and perceptive handling for vaccines.
PDF: https://apps.who.int/iris/rest/bitstreams/1392676/retrieve
1 December 2021
Global vaccine safety blueprint 2.0 background research
30 November 2021
Global progress report on WASH in health care facilities: Fundamentals first
30 November 2021
Framework for countries to achieve an integrated continuum of long-term care
30 November 2021
Approaches to enhance and accelerate study of new drugs for HIV and associated infections in pregnant…
29 November 2021
COVID-19 vaccine trial designs in the context of authorized COVID-19 vaccines and expanding global access: ethical considerations
Overview
Executive Summary
In June 2020, global regulators convened under the auspices of the International Coalition of Medicines Regulatory Authorities (ICMRA) and co-chaired jointly by the European Medicines Agency (EMA) and United States Food and Drug Administration (FDA) reached consensus on the study design requirements for Phase 3 COVID-19 vaccine clinical trials. The ICMRA noted that phase 3 clinical trials should be randomized, double-blinded and controlled with a placebo or active comparators. In September 2020, the World Health Organization (WHO ) advised: “Phase IIB/III efficacy trials should be randomized, double-blinded and placebo controlled.” Since then, multiple COVID-19 vaccines have been authorized worldwide based on interim results of pivotal placebo-controlled efficacy trials, and billions of COVID-19 vaccine doses have been administered under emergency use/conditional marketing authorization or full approval regulatory mechanisms.
In December 2020, a WHO expert group advised that the placebo control arms of COVID-19 vaccine trials should be progressively unblinded as authorized vaccines become available in the community hosting the trial, starting with prioritized groups.
Before a COVID-19 vaccine trial commences enrolment, if one or more authorized/approved COVID-19 vaccine is locally available and the participant meets programmatic eligibility criteria, the study team should advise the participant that they are eligible to receive the authorized vaccine(s). Participants may elect to receive the authorized vaccine at any point in the trial.
The appropriateness of conducting a placebo control trial may depend on whether the candidate vaccine is a prototype vaccine, modified vaccine or next generation vaccine
PDF: https://apps.who.int/iris/rest/bitstreams/1396234/retrieve
26 November 2021
Global guidance on criteria and processes for validation: elimination of mother-to-child transmission of HIV, syphilis and hepatitis B virus
CDC/ACIP [U.S.] [to 4 Dec 2021]
http://www.cdc.gov/media/index.html
https://www.cdc.gov/vaccines/acip/index.html
Latest News Releases, Announcements [Selected]
CDC Tightens Testing Requirement for International Travel to the US to One Day
Friday, December 3, 2021
Public Health Authorities Investigating Additional Confirmed Case of COVID-19 Caused by the Omicron Variant
Thursday, December 2, 2021
First Confirmed Case of Omicron Variant Detected in the United States
Wednesday, December 1, 2021
New CDC Vital Signs Report Reveals a Decade of Continuing HIV Inequities on Eve of World AIDS Day Tuesday, November 30, 2021
CDC Expands COVID-19 Booster Recommendations
Monday, November 29, 2021
ACIP Meetings
No new meetings listed.
MMWR News Synopsis Friday, December 3, 2021
Selected Content
:: Symptoms of Depression, Anxiety, Post-Traumatic Stress Disorder, and Suicidal Ideation Among State, Tribal, Local, and Territorial Public Health Workers During the COVID-19 Pandemic — United States, March–April 2021
:: Vital Signs: HIV Infection, Diagnosis, Treatment, and Prevention Among Gay, Bisexual, and Other Men Who Have Sex with Men — United States, 2010–2019
:: Correction and Republication: Symptoms of Depression, Anxiety, Post-Traumatic Stress Disorder, and Suicidal Ideation Among State, Tribal, Local, and Territorial Public Health Workers During the COVID-19 Pandemic — United States, March–April 2021
Coronavirus Disease 2019 (COVID-19)– CDC
Approximately 25 announcements/reports/data summaries.
:: 3 Dec CDC Tightens Testing Requirement for International Travel to the US to One Day
:: 3 Dec Overall US COVID-19 Vaccine Distribution and Administration Update as of Fri, 03 Dec 2021 06:00:00 EST
Africa CDC [to 4 Dec 2021]
http://www.africacdc.org/
News
Press Releases
Africa Centres for Disease Control and Prevention’s Statement regarding the new SARS-COV-2 virus variant B.1.1.529
26 November 2021
Press Releases
Mastercard Foundation and Africa CDC’s Saving Lives and Livelihoods initiative delivers first tranche of over 15 million vaccines
ADDIS ABABA, ETHIOPIA, 25 November 2021 — The Mastercard Foundation and Africa CDC jointly announced that 15.2 million vaccines purchased under the Saving Lives and Livelihoods initiative are currently being distributed across Africa. Through Saving Lives and Livelihoods, the Mastercard Foundation is purchasing vaccines for more than 65 million people. This is the first tranche of vaccines to be delivered under the initiative. The vaccines will be distributed within countries by UNICEF.
Launched in June 2021, Saving Lives and Livelihoods is now a $1.5 billion partnership that aims to enable vaccination for millions of people, develop a workforce for vaccine manufacturing, and strengthen the Africa CDC’s capacity to oversee a historic vaccination campaign and effectively respond to future outbreaks.
“Timely delivery of these vaccines underscores the effectiveness of the African Vaccine Acquisition Trust. Working with the Africa CDC and multiple organizations, the Mastercard Foundation will support the roll out of vaccinations to millions across the continent. More remains to be done to urgently increase vaccination rates. We call on governments, funders, civil society and others to step forward to save lives and livelihoods in Africa,” said Reeta Roy, President and CEO of the Mastercard Foundation…
China CDC http://www.chinacdc.cn/en/
National Health Commission of the People’s Republic of China [to 4 Dec 2021]
http://en.nhc.gov.cn/
News
Dec 4: Daily briefing on novel coronavirus cases in China
On Dec 3, 31 provincial-level regions and the Xinjiang Production and Construction Corps on the Chinese mainland reported 90 new cases of confirmed infections.
China has provided over 1.8b COVID-19 vaccine doses to international community
2021-12-03
China pushing development of vaccine against Omicron
2021-12-03
China sticks to its strategy in COVID-19 response: official
2021-12-02
National Medical Products Administration – PRC [to 4 Dec 2021]
http://english.nmpa.gov.cn/
News
Over 2.5b COVID-19 vaccine doses administered on Chinese mainland
2021-12-02
Over 2.5 billion COVID-19 vaccine doses had been administered on the Chinese mainland as of Nov 30, data from the National Health Commission showed on Dec 1.
Xi says China will supply Africa with additional 1 bln COVID-19 vaccine doses
2021-11-30
Chinese President Xi Jinping on Monday said China will provide an additional 1 billion doses of COVID-19 vaccines to Africa.
CCDC Weekly – Weekly Reports: Current Volume (3)
2021-12-03 / No. 49 INFLUENZA ISSUE
View PDF of this issue
Editorial: China CDC Weekly’s Second Meeting of the Editorial Board, Advisory Committee, and Editorial Office to Celebrate the Second Anniversary of the Inaugural Issue
Methods and Applications: The Incoming Influenza Season — China, the United Kingdom, and the United States, 2021–2022
Perspectives: Quo Vadis Influenza?
Perspectives: GISAID’s Role in Pandemic Response
Perspectives: A Tale of Two Cities: From Influenza HxNy to SARS-CoV-z
::::::
Organization Announcements
Editor’s Note:
Careful readers will note that the number and range of organizations now monitored in our Announcements section below has grown as the impacts of the pandemic have spread across global economies, supply chains and programmatic activity of multilateral agencies and INGOs.
Paul G. Allen Frontiers Group [to 4 Dec 2021]
https://alleninstitute.org/news-press/
News
$20M awards fund centers’ studies of human brain evolution, animal development
December 2, 2021
Two Allen Discovery Centers enter their next phase of discovery, poised to address large questions about biology
BARDA – U.S. Department of HHS [to 4 Dec 2021]
https://www.phe.gov/about/barda/Pages/default.aspx
News
No new digest content identified.
BMGF – Gates Foundation [to 4 Dec 2021]
https://www.gatesfoundation.org/ideas/media-center
Press Releases and Statements
No new digest content identified.
Bill & Melinda Gates Medical Research Institute [to 4 Dec 2021]
https://www.gatesmri.org/
The Bill & Melinda Gates Medical Research Institute is a non-profit biotech organization. Our mission is to develop products to fight malaria, tuberculosis, and diarrheal diseases—three major causes of mortality, poverty, and inequality in developing countries. The world has unprecedented scientific tools at its disposal; now is the time to use them to save the lives of the world’s poorest people
No new digest content identified.
CARB-X [to 4 Dec 2021]
https://carb-x.org/
News
No new digest content identified.
Center for Vaccine Ethics and Policy – GE2P2 Global Foundation [to 4 Dec 2021]
https://centerforvaccineethicsandpolicy.net/
News/Analysis/Statements
:: Past weekly editions and posting of all segments of Vaccines and Global Health: The Week in Review are available here.
:: Informed Consent: A Monthly Review – December 2021 is now posted here
CEPI – Coalition for Epidemic Preparedness Innovations [to 4 Dec 2021]
http://cepi.net/
Latest News
CEPI and MVC co-fund mix-and-match booster trial of COVID-19 vaccines
Clinical trial will evaluate heterologous and fractional booster doses – or ‘mix-and-match’ booster – combinations of MVC-COV1901 with other COVID-19 vaccines.
02 Dec 2021
Speech made by CEPI CEO, Dr Richard Hatchett, on the emergence of Omicron variant at today’s World Health Assembly special session
Dr Richard Hatchett, CEO of CEPI, addressed today’s special session of the World Health Assembly—on behalf of the Access to COVID-19 Tools Accelerator.
“…South Africa and Botswana’s efficient surveillance, early detection, and warning, and sharing of information have granted the world precious time. Time to implement containment measures and time to prepare. Our industry partners are now intensively investigating whether our current vaccines have been compromised and consulting with regulators and WHO as they develop new ones just in case.
“As you embark on this historic Special Session, I would urge you to keep this example, which is right in front of you, in mind.
“The world is at a crossroads. We know we will face future global infectious disease emergencies and potential pandemic threats. That is a certainty. What happens when these threats materialize, however, is up to us – and in a very tangible sense, up to you.
“We urgently need a global framework or convention on pandemic preparedness and response if we are to have any hope of preventing future pandemics, accelerating the global availability of critical medical countermeasures, and ensuring more just outcomes than we have achieved with COVID…”
DARPA – Defense Advanced Research Projects Agency [to 4 Dec 2021
https://www.darpa.mil/news
News
Our Blog
No new digest content identified.
Duke Global Health Innovation Center [to 4 Dec 2021]
https://dukeghic.org/
Our Blog
No new digest content identified.
EDCTP [to 4 Dec 2021]
http://www.edctp.org/
The European & Developing Countries Clinical Trials Partnership (EDCTP) aims to accelerate the development of new or improved drugs, vaccines, microbicides and diagnostics against HIV/AIDS, tuberculosis and malaria as well as other poverty-related and neglected infectious diseases in sub-Saharan Africa, with a focus on phase II and III clinical trials
News
01 December 2021
World AIDS Day 2021: tackling inequalities
Emory Vaccine Center [to 4 Dec 2021]
http://www.vaccines.emory.edu/
Vaccine Center News
No new digest content identified.
European Vaccine Initiative [to 4 Dec 2021]
http://www.euvaccine.eu/
Latest News News, Events
EVI
World AIDS Day 2021: End inequalities. End AIDS
1 December 2021 What is world AIDS
Fondation Merieux [to 4 Dec 2021]
http://www.fondation-merieux.org/
News, Events
Mérieux Foundation co-organized event
8th Meeting of the GTFCC Working Group on Oral Cholera Vaccine
December 6 – 8, 2021 – Événement virtuel & Centre des Pensières, Veyrier du Lac (France)
Gavi [to 4 Dec 2021]
https://www.gavi.org/
News Releases
3 December 2021
Gavi Board approves malaria vaccine programme funding, COVAX 2022 strategic approach and measures to maintain, restore and strengthen routine immunisation
[See Week in Review above for detail]
2 December 2021
Gavi Board approves funding to support malaria vaccine roll-out in sub-Saharan Africa
2 December 2021
New Zealand kicks off 2022 COVAX fundraising efforts with NZ$ 9 million funding pledge
29 November 2021
Joint Statement on Dose Donations of COVID-19 Vaccines to African Countries
GHIT Fund [to 4 Dec 2021]
https://www.ghitfund.org/newsroom/press
GHIT was set up in 2012 with the aim of developing new tools to tackle infectious diseases that
No new digest content identified.
Global Fund [to 4 Dec 2021]
https://www.theglobalfund.org/en/news/
News & Stories
News
The Next Chapter of Our Fight Against HIV
01 December 2021 by Peter Sands, Executive Director
Current Testing Tools Uncompromised by New COVID-19 Variant of Concern Omicron (B.1.1.529)
29 November 2021
The accuracy of existing molecular (PCR, NAAT) tests appears uncompromised by COVID-19 variant Omicron (B.1.1.529), designated a Variant of Concern by the World Health Organization (WHO) on Friday. Preliminary evidence suggests that this is also true…
Global Research Collaboration for Infectious Disease Preparedness [GloPID-R] [to 4 Dec 2021]
https://www.glopid-r.org/news/
News
No new digest content identified.
Hilleman Laboratories [to 4 Dec 2021]
http://www.hillemanlabs.org/
….No “news” page or tab identified.
HHMI – Howard Hughes Medical Institute [to 4 Dec 2021]
https://www.hhmi.org/news
Press Room
No new digest content identified
Human Vaccines Project [to 4 Dec 2021]
http://www.humanvaccinesproject.org/
News
No new digest content identified
IAVI [to 4 Dec 2021]
https://www.iavi.org/newsroom
Latest News
December 3, 2021
GreenLight (ENVI) and IAVI begin work on Omicron variant-adapted COVID-19 vaccine candidate
BOSTON and NEW YORK – DECEMBER 3, 2021 – GreenLight Biosciences (ENVI) and IAVI, a non-profit scientific research organization, today announced work on a messenger RNA vaccine candidate to tackle the COVID-19 Omicron variant.
“The highly mutated Omicron variant may be partially capable of eluding existing vaccines, so rapid adaptation is an urgent priority,” said GreenLight CEO Andrey Zarur. “We have started lab work already. We will be testing a new candidate vaccine shortly in preclinical models. We have also begun an analysis on how we would proceed in clinical studies in Africa, in the context of our current clinical plans.”
IAVI President and CEO Mark Feinberg said, “While it’s impossible to say right now where Omicron first emerged, we wouldn’t have been able to respond to this threat so quickly had it not been for the skill and openness of the South African scientists who identified the variant and shared the sequence with the world. Now we must focus on bringing solutions forward both to address this new variant and the stark inequities that remain in the global availability of vaccines to prevent COVID-19.”..
November 30, 2021
GreenLight Biosciences (ENVI) partners with IAVI to accelerate COVID-19 vaccine trial in Africa
IAVI’s extensive clinical trial experience will be combined with GreenLight COVID-19 vaccine candidate.
Goal is to broaden and accelerate access to messenger RNA (mRNA) COVID-19 vaccines.
Phase I trial planned for late Q1 2022.
Plan speeds creation of vaccines in Africa, for Africa, and suitable for use in low-income countries globally.
International Coalition of Medicines Regulatory Authorities [ICMRA]
http://www.icmra.info/drupal/en/news
Selected Statements, Press Releases, Research
No new digest content identified.
ICRC [to 4 Dec 2021]
https://www.icrc.org/en/whats-new
Selected News Releases, Statements, Reports
ICRC: Omicron highlights the need to step up global vaccinations, including in conflict zones
Statement from Esperanza Martinez, the head of the COVID-19 Crisis Management Team at the ICRC on vaccine inequity.
02-12-2021 | News release
IFFIm
http://www.iffim.org/
Press Releases/Announcements
No new digest content identified.
IFRC [to 4 Dec 2021]
http://media.ifrc.org/ifrc/news/press-releases/
Selected Press Releases, Announcements
02/12/2021
“Impulsive reactions are an affront to the global solidarity we need to successfully respond to a crisis of this scale.” [WHA]
29/11/2021
“Impulsive and ineffective reactions” to COVID-19’s Omicron variant will send more Africans into poverty
Mohammed Mukhier, IFRC Regional Director for Africa
29/11/2021
“The pandemic has both thrived on inequities and exacerbated them”
Institut Pasteur [to 4 Dec 2021]
https://www.pasteur.fr/en/press-area
Press Documents
Press release
26.11.2021
ComCor study: new results on places of infection with SARS-CoV-2 and analysis of the efficacy of messenger RNA vaccines against the Delta variant
From May 23 to August 13, 2021, the Institut Pasteur, in partnership with the French National Health Insurance Fund (CNAM), Santé publique France and the Ipsos institute, conducted the fourth part of the ComCor epidemiological study on circumstances and places of infection with the SARS-CoV-2 virus in France. The aim of the study was to identify the socio-demographic factors, places visited and behaviors associated with a higher risk of infection with SARS-CoV-2. The study was also used to carry out a real-world analysis of the effect of previous infection with COVID-19 and the efficacy of messenger RNA vaccines against symptomatic infection with the Delta variant. The findings were published in The Lancet Regional Health Europe on November 26, 2021.
IOM / International Organization for Migration [to 4 Dec 2021]
http://www.iom.int/press-room/press-releases
News – Selected
News
01 Dec 2021
IOM’s World Migration Report Shows Global Displacement Rising Despite COVID-19 Mobility Limits
ISC / International Science Council [to 4 Dec 2021]
https://council.science/current/
ISC is a non-governmental organization with a unique global membership that brings together 40 international scientific Unions and Associations and over 140 national and regional scientific organizations including Academies and Research Councils.
News
Press releases
The International Year of Basic Sciences for Sustainable Development proclaimed by the United Nations General Assembly for 2022
03.12.2021
We need more basic sciences to achieve Agenda 2030 and its 17 Sustainable Development Goals. This is the message sent to the world by the United Nations General Assembly on 2 December 2021: Member States approved by consensus the resolution 76/A/L.12 promulgating the year 2022 as the International Year of Basic Sciences for Sustainable Development (IYBSSD2022).
With this resolution, the United Nations General Assembly ‘invites all [its] Member States, organizations of the United Nations system and other global, regional and subregional organizations, as well as other relevant stakeholders, including academia, civil society, inter alia, international and national non-governmental organizations, individuals and the private sector, to observe and raise awareness of the importance of basic sciences for sustainable development, in accordance with national priorities’…
IVAC [to 4 Dec 2021]
https://www.jhsph.edu/research/centers-and-institutes/ivac/index.html
Updates; Events
No new digest content identified.
IVI [to 4 Dec 2021]
http://www.ivi.int/
IVI News & Announcements
No new digest content identified.
Johns Hopkins Center for Health Security [to 4 Dec 2021]
https://www.centerforhealthsecurity.org/news/center-news/
Center News
No new digest content identified.
MSF/Médecins Sans Frontières [to 4 Dec 2021]
http://www.msf.org/
Latest [Selected Announcements]
Ukraine
Volunteers help fellow villagers access healthcare in conflict-affected eastern Ukraine
Project Update 3 Dec 2021
Coronavirus COVID-19 pandemic
In an unequal world, our response to COVID-19 cannot be one size-fits-all
Opinion 3 Dec 2021
Myanmar
Myanmar: Political turmoil threatens HIV care
MSF Asia Pacific. 1 Dec 2021
HIV/AIDS
DRC: People are still dying unnecessarily from HIV
Project Update 1 Dec 2021
World AIDS Day
Why are people living with HIV still dying of AIDS?
Opinion 1 Dec 2021
World Health Assembly
Pandemic preparedness and response: some lessons learnt
Statement 30 Nov 2021
MSF delivered a statement on 30 November 2021 during the World Health Assembly Special Session.
National Academy of Medicine – USA [to 4 Dec 2021]
https://nam.edu/programs/
Selected News/Programs/Events
Expert Paper from the National Academy of Medicine Identifies Lessons Learned and Compelling Needs to Center Patients, Families, and Communities in the Delivery of Health and Health Care After COVID-19
November 29, 2021
WASHINGTON – An individually-authored discussion paper was published in NAM Perspectives today that focuses on the experience and needs of patients, families, and communities, was published on November 29, 2021. Patients, Families, and Communities COVID-19 Impact Assessment: Lessons Learned and Compelling Needs reviews the impact and implications of COVID-19 on patients, families, and communities, offers […]
National Academy of Sciences – USA [to 4 Dec 2021]
http://www.nasonline.org/news-and-multimedia/
News
No new digest content identified.
National Vaccine Program Office – U.S. HHS [to 4 Dec 2021]
https://www.hhs.gov/vaccines/about/index.html
Upcoming Meetings/Latest Updates
No new digest content identified.
NIH [to 4 Dec 2021]
http://www.nih.gov/news-events/news-releases
News Releases
NIH Statement on World AIDS Day 2021
December 1, 2021 — Statement of Anthony S. Fauci, M.D. and Maureen M. Goodenow, Ph.D.
… Although the HIV vaccine field has been marked by disappointing results over the years, finding a safe, effective and durable HIV vaccine remains an NIH priority. Currently, the Phase 3 Mosaico/HPTN 706 HIV vaccine clinical trial is underway in the Americas and Europe with results expected in 2024. Lessons learned from highly effective SARS-CoV-2 vaccines also offer an encouraging path forward for HIV vaccine discovery by providing applications for new vaccine platforms, such as mRNA, and novel strategies for rapidly identifying vaccine targets. Additionally, promising outcomes utilizing bNAbs suggest it may be possible to achieve an HIV vaccine with a high level of efficacy—an almost inconceivable scientific possibility several years ago…
Too many people with HIV fail to achieve durable viral suppression
November 30, 2021 — NIH-funded study estimates global progress toward UNAIDS goal.
Article: WM Han et al. Global estimates of viral suppression in children and adolescents and adults on antiretroviral therapy adjusted for missing viral load measurements: a multiregional, retrospective cohort study in 31 countries(link is external). The Lancet HIV (2021).
OECD [to 4 Dec 2021]
http://www.oecd.org/newsroom/publicationsdocuments/bydate/
Newsroom
Japan: broaden the digital transition to strengthen economic recovery from COVID-19, says OECD
3-December-2021
Rising vaccination rates and a rebound in exports are helping Japan’s economy to recover from the shock caused by COVID-19, although challenges remain. Investing in technology, education and professional training to broaden and accelerate the country’s digital transformation would help to spur productivity growth and reinforce public finances, according to a new OECD report.
PATH [to 4 Dec 2021]
https://www.path.org/media-center/
Press Releases
PATH welcomes commitment by Gavi board to finance wider rollout of the world’s first malaria vaccine, RTS,S
December 2, 2021 by PATH
Today’s important decision enables low- and middle-income countries to consider adding RTS,S as an additional tool to combat malaria
Additional investments will be needed to expand vaccine supply if current demand projections are realized
Together with partners and other stakeholders, PATH is working to help ensure sufficient malaria vaccine supply to meet potential demand
Sabin Vaccine Institute [to 4 Dec 2021]
http://www.sabin.org/updates/pressreleases
Statements and Press Releases
No new digest content identified.
UNAIDS [to 4 Dec 2021]
http://www.unaids.org/en
Selected Press Releases/Reports/Statements
3 December 2021
Strengthening Haiti’s HIV response through community-led monitoring
2 December 2021
Community health workers strengthen HIV and COVID-19 responses
1 December 2021
Letter from Prince Harry to Dr Tedros Adhanom Ghebreyesus and Ms Winnie Byanyima on World AIDS Day
UNHCR Office of the United Nations High Commissioner for Refugees [to 4 Dec 2021]
http://www.unhcr.org/en-us/media-centre.htmlS
Selected News Releases, Announcements
Prolonged COVID-19 pandemic deepens hardship for over 12 million forcibly displaced people with disabilities
3 Dec 2021
At least 12 million people with disabilities are forcibly displaced worldwide, UNHCR, the UN Refugee Agency, estimates, and their already precarious situation is becoming harder as the COVID-19 pandemic drags on.
“Forced displacement disproportionately affects people with disabilities. They are often at higher risk of violence, discrimination, neglect, gender-based violence, exploitation and abuse, face barriers to access basic services, and are often excluded from education and livelihood opportunities,” said Gillian Triggs, UNHCR’s Assistant High Commissioner for Protection.
“While many refugees faced these unacceptable, pre-existing risks of exclusion and discrimination, we fear that the prolonged COVID-19 pandemic is only deepening inequalities and hardship,” she added.
While estimates say over 12 million people with disabilities have been forcibly displaced by persecution, violence and human rights violations worldwide, surveys and assessments suggest the real number may be much higher.
Challenges and risks begin at the moment of flight. In sudden onset disasters, abrupt violence, and active hostilities, people with disabilities are often left behind by family members due to the sudden nature of armed attacks; in extreme cases reported, people were abandoned while chained.
On reaching safety, services and facilities, including assistance programmes and protection, may be inaccessible. People with disabilities may face physical barriers and obstacles to reach, enter or use basic services, or key information may be delivered in formats not accessible to them.
Refugees and internally displaced and stateless people with disabilities were already less likely to access health care, education and employment opportunities, and the global crisis has further compounded this situation.
On the International Day of Persons with Disabilities today, UNHCR is urging national authorities to do more to secure the rights of forcibly displaced and stateless people with disabilities and to counter all forms of discrimination…
UNICEF [to 4 Dec 2021]
https://www.unicef.org/media/press-releases
Press Releases, News Notes, Statements [Selected]
Press release
11/29/2021
Joint Statement on Dose Donations of COVID-19 Vaccines to African Countries
A joint statement from the African Vaccine Acquisition Trust (AVAT), the Africa Centres for Disease Control and Prevention (Africa CDC) and COVAX
[See Milestones above for detail]
Press release
11/29/2021
A child was infected with HIV every two minutes in 2020 – UNICEF
A prolonged COVID-19 pandemic is deepening the inequalities that have long driven the HIV epidemic, UNICEF warns Ahead of World AIDS Day.
Unitaid [to 4 Dec 2021]
https://unitaid.org/
Featured News
30 November 2021
COVID-19 creates new urgency for access to HIV self-testing, says global health agency Unitaid
The number of countries with self-testing policies in place has increased by nearly 15-fold since 2015, with an estimated 192 million self-tests needed to cover low- and middle-income countries by 2025.
Geneva – Disruptions and delays to HIV services caused by the COVID-19 pandemic led to declines in HIV testing and diagnoses in 2020 for the first time in more than two decades. HIV self-testing, which has contributed to a 40% reduction in the number of people who do not know their HIV status since 2015, is now crucial to maintaining access to HIV care for millions of people in the face of new COVID-19 roadblocks.
With more than 37 million people living with HIV worldwide – an estimated 6.1 million of whom do not know their status – receiving a diagnosis is a vital first step in accessing treatment.
Unitaid has led a large-scale effort to create access to HIV treatment and prevention through self-testing, with more than US$100 million invested since 2015. This work has helped reduce prices for self-tests, generate demand, demonstrate implementation pathways, and support scale up across 14 countries in Africa[1], with more recent work in India and Indonesia looking to further expand successful self-testing strategies to more people…
While procurement of HIV self-tests is rising, it is greatly outpaced by the need. By 2025, the number of self-tests procured is projected to fill just 15% of the estimated 192 million tests needed in low- and middle-income countries. And although the funding allocated for self-testing has increased significantly over the past several years, an additional US$104 million is needed to cover the costs of the 29 million self-tests expected to be procured.
Ahead of World AIDS Day, 1 December, Unitaid is calling for urgent funding and scale-up of HIV self-testing to protect progress threatened by COVID-19 and secure pathways to HIV treatment and prevention services for millions of people worldwide.
Vaccine Equity Cooperative [nee Initiative] [to 4 Dec 2021]
https://vaccineequitycooperative.org/news/
News
No new digest content identified.
Vaccination Acceptance & Demand Initiative [Sabin) [to 4 Dec 2021]
https://www.vaccineacceptance.org/
Announcements
No new digest content identified.
Vaccine Confidence Project [to 4 Dec 2021]
http://www.vaccineconfidence.org/
News, Research and Reports
No new digest content identified.
Vaccine Education Center – Children’s Hospital of Philadelphia [to 4 Dec 2021]
http://www.chop.edu/centers-programs/vaccine-education-center
News
No new digest content identified.
Wellcome Trust [to 4 Dec 2021]
https://wellcome.ac.uk/news
News. Opinion, Reports
News
“There isn’t a united global strategy” – we need to ensure vaccine equity to end Covid-19
30 November 2021
As the World Health Organization (WHO) announces a new variant of concern, we find out what people around the world think about Covid-19 vaccine distribution.
News
Public trust in scientists rose during the Covid-19 pandemic
29 November 2021
Insights from the Wellcome Global Monitor – the largest global survey of how people think and feel about science – found levels of trust in scientists have increased since 2018, placing it on par with doctors and nurses…
Key findings
[1] Covid-19 has put science in the public eye like never before. The Wellcome Global Monitor offers insight into how people think and feel about science and major health challenges worldwide.
Key findings include:
[2] The impact of Covid-19 has been uneven around the world. The Wellcome Global Monitor found that the pandemic has had a disproportionate impact on low-income countries and people with low incomes across all countries.
[3] People’s trust in science and scientists has increased. Globally, those who said they trust scientists ‘a lot’ rose from 34% in 2018 to 43% by the end of 2020. These increases were highest among those who indicated they know ‘some’ or ‘not much/nothing at all’ about science.
People are not sure that their governments value scientific advice. World leaders need to listen and show that they share public confidence in the role of science in solving health challenges fairly for everyone.
The Wistar Institute [to 4 Dec 2021]
https://www.wistar.org/news/press-releases
Press Releases
Dec. 2, 2021
Fox Chase Cancer Center, The Wistar Institute Announce Opening of Phase 1 Clinical Trial of Anticancer Drug Candidate
PHILADELPHIA — (Dec. 2, 2021) — Fox Chase Cancer Center has opened an investigator-initiated, phase 1 clinical trial to evaluate the safety and efficacy of gamitrinib in patients with advanced cancer. Gamitrinib is a first-in-class, mitochondrial-targeted inhibitor of the molecular chaperone heat shock protein-90 (HSP90) that works by inhibiting tumor cell metabolism and survival.
Press Release
Nov. 30, 2021
Wistar Scientists Identify Genes Critical to Protecting Ovarian Cancer from the Immune System
Discovery could make immunotherapy more effective for ovarian and other cancers.
WFPHA: World Federation of Public Health Associations [to 4 Dec 2021]
https://www.wfpha.org/
Latest News – Blog
A Vaccine TRIPS Waiver Now: Letter to WTO
Nov 29, 2021
Vaccination can save lives in millions. Massive upscaling of vaccine production is required across the globe with the COVID-19 pandemic continuing and with the prospect of new waves, and new variants in the future.
The World Federation of Public Health Associations, the Sustainable Health Equity Movement and the Global Network for Academic Public Health urge the World Trade Organisation to do everything in its power to achieve agreement to a temporary vaccine Trade-related Intellectual Property Rights waiver at its Ministerial Conference.
We believe the vaccine TRIPS waiver is the single most important action that can be taken to secure full and fair access to vaccine across the world, to give us the best possible chance of ending this pandemic, saving lives, and preventing disease and disability.
Letter
World Bank [to 4 Dec 2021]
http://www.worldbank.org/en/news/all
Selected News, Announcements
An Uneven Recovery: the Impact of COVID-19 on Latin America and the Caribbean
WASHINGTON D.C., November 29, 2021– Employment rates in some Latin American and Caribbean countries have experienced a relative recovery, although in most, rates fall short of pre-pandemic levels. The…
Date: November 29, 2021 Type: Press Release
World Organisation for Animal Health (OIE) [to 4 Dec 2021]
https://www.oie.int/en/media/news/
Press Releases, Statements
Tripartite and UNEP support OHHLEP’s definition of “One Health”
Joint Tripartite (FAO, OIE, WHO) and UNEP Statement
1 Dec 2021
The Food and Agriculture Organization of the United Nations (FAO), the World Organisation for Animal Health (OIE), the United Nations Environment Programme (UNEP) and the World Health Organization (WHO) welcome the newly formed operational definition of One Health from their advisory panel, the One Health High Level Expert Panel (OHHLEP), whose members represent a broad range of disciplines in science and policy-related sectors relevant to One Health from around the world. The four organizations are working together to mainstream One Health so that they are better prepared to prevent, predict, detect, and respond to global health threats and promote sustainable development.
The One Health definition developed by the OHHLEP states:
“One Health is an integrated, unifying approach that aims to sustainably balance and optimize the health of people, animals and ecosystems. It recognizes the health of humans, domestic and wild animals, plants, and the wider environment (including ecosystems) are closely linked and inter-dependent.The approach mobilizes multiple sectors, disciplines and communities at varying levels of society to work together to foster well-being and tackle threats to health and ecosystems, while addressing the collective need for clean water, energy and air, safe and nutritious food, taking action on climate change, and contributing to sustainable development.”
WTO – World Trade Organisation [to 4 Dec 2021]
http://www.wto.org/english/news_e/news_e.htm
WTO News and Events
Negotiations on services domestic regulation conclude successfully in Geneva
2 December 2021
Heads of Geneva delegations from the 67 WTO members participating in the Joint Initiative on Services Domestic Regulation announced on 2 December the successful conclusion of negotiations aimed at slashing administrative costs and creating a more transparent operating environment for service providers hoping to do business in foreign markets.
DG calls on members to agree on pandemic response, fisheries subsidies by end-February
2 December 2021
Following last week’s eleventh-hour decision by WTO members to postpone the 12th Ministerial Conference, negotiations are continuing in Geneva, and Director-General Ngozi Okonjo-Iweala today (2 December) urged delegation heads to redouble their efforts to bridge differences. She called on them to conclude agreements on the WTO system’s response to pandemics as well as on curbing harmful fisheries subsidies by the end of February 2022 to pave the way for approval by ministers. “Seven billion people are waiting for us on TRIPS and pandemic response. And 260 million people are waiting for us on fisheries subsidies,” she said.
[See Milestones above for detail]
::::::
ARM [Alliance for Regenerative Medicine] [to 4 Dec 2021]
https://alliancerm.org/press-releases/
Selected Press Releases
No new digest content identified.
BIO [to 4 Dec 2021]
https://www.bio.org/press-releases
Press Releases, Letters, Testimony, Comments [Selected]
No new digest content identified.
DCVMN – Developing Country Vaccine Manufacturers Network [to 4 Dec 2021]
http://www.dcvmn.org/
News; Upcoming events
No new digest content identified.
ICBA – International Council of Biotechnology Associations [to 4 Dec 2021]
https://internationalbiotech.org/news/
News
No new digest content identified.
IFPMA [to 4 Dec 2021]
http://www.ifpma.org/resources/news-releases/
Selected Press Releases, Statements, Publications
Global pharmaceutical industry leaders meet with UK Prime Minister to discuss leadership in global health
London, 3 December, 2021 – Yesterday, global leaders from the pharmaceutical industry met with the Prime Minister, senior UK Cabinet Ministers and the NHS Chief Executive, to discuss global health and innovation. Their discussions focused on how the industry and UK Government can work together to drive pharmaceutical innovation and rapidly deliver the next generation of medicines to patients worldwide, and deliver on the ambition of the UK Government’s Life Sciences Vision…
Global biopharmaceutical industry early response to Omicron
3 December 2021 – COVID-19, the worst pandemic the world has seen in 100 years, has shown us that we cannot let our guard down. The global biopharmaceutical industry understands the concern caused by the emergence of the Omicron variant of COVID-19 since it was declared a variant of concern by the World Health Organization.
As a science-driven industry addressing some of the world’s biggest healthcare challenges, the biopharmaceutical industry has demonstrated over the last two years how it is uniquely positioned to respond rapidly to COVID-19. Biopharmaceutical companies are bringing the same commitment to innovation in responding to the Omicron variant.
It is early days in terms of understanding Omicron. Together with the broader scientific community, public health and regulatory bodies around the world, our industry will again bring its experience and expertise to bear on new variants such as Omicron.
None of this work would have been possible if the South African and Botswana scientists had not shared the Omicron variant so swiftly through tried and tested platforms such as GISAID. Their fast sharing of the data has bought us all time in responding to and potentially controlling this new variant. The current situation reflects the advantage of fast and effective sharing of harmful pathogens and variants, which allows us to develop effective vaccines, treatments, and diagnostics.
Biopharmaceutical companies have hit the ground running in response to the Omicron variant, as they have done with previous variants (Beta and Delta). Crucially, they are looking at how both authorized and unapproved vaccines and treatments can stand up to this latest variant. A number of vaccine developers have already stated that they will develop Omicron targeting candidates.
The emergence of Omicron variant comes on top of a surge in COVID-19 cases in many regions of the world, caused by the dominant Delta variant at a time when health systems are already working hard to cope with seasonal endemic infections. Reducing the toll of the pandemic on lives and livelihoods requires not only continued innovation in the development and production of vaccines, treatments, and diagnostics, but also urgently addressing inequality in vaccine distribution and country readiness for vaccination. Our Five Steps to urgently advance COVID-19 vaccine equity announced in May 2022 are as relevant and as demanding of action today as they were when they were first launched.
Joint Statement on Patient Solidarity Day
Geneva, December 3rd, 2021 – On Patient Solidarity Day, the International Alliance of Patients’ Organizations (IAPO), the International Pharmaceutical Federation (FIP), the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA), the International Hospital Federation (IHF) and the International Council of Nurses (ICN), join patients and families all over the world, in calling for governments and all health stakeholders to collaborate in the implementation of the WHO Flagship Global Patient Safety Action Plan 2021-2030 (GPSAP 2021-30) that was adopted at the 74th World Health Assembly this year.
Every year, large numbers of patients are harmed or die because of unsafe health care, creating a high burden of death and disability worldwide, especially in low- and middle-income countries. On average, an estimated one in 10 patients is subject to an adverse event while receiving hospital care in high-income countries. Available evidence suggests that 134 million adverse events due to unsafe care occur in hospitals in low- and middle-income countries, contributing to around 2.6 million deaths every year. According to recent estimates, the social cost of patient harm can be valued at US$ 1 trillion to 2 trillion a year.[1]
Patient safety is fundamental to the provision of health care in all settings. However, avoidable adverse events, errors and risks associated with health care remain major challenges for patient safety globally. The global COVID-19 pandemic has highlighted the importance of building a culture of trust in healthcare systems, health professionals and medical products. Trust can be undermined by products and interventions that do not adhere to the highest standards of quality and ethical standards.
We therefore call on all governments and health stakeholders to deploy patient safety as a health priority in health sector policies and programmes to achieve universal health coverage; and for all health stakeholders to uphold the guiding principles of GPSAP 2021-30, by working to:
Implement policies to eliminate avoidable harm in health
Foster high-reliability systems
Ensure safety of clinical processes
Maintain patient and family engagement
Secure continuous health worker education, skills and safety
Today, representing key actors in health systems, we reaffirm our commitment to provide the highest quality health care and health products to patients and to work towards the GPSAP 2021-30 vision of “a world in which no one is harmed in health care, and every patient receives safe and respectful care, every time, everywhere”.
[1] Global Patient Safety Action Plan 2021–2030 Towards eliminating avoidable harm in health care
Biopharmaceutical industry collaborates on the implementation of the G7 100 Days Mission to improve readiness
Published on: 03 December 2021
The biopharmaceutical industry welcomes the publication of the 100 Days Mission – First Implementation Report released on 2 December, which maps out key actions needed from all stakeholders to build global pandemic preparedness. As one of the implementation partners, we welcome the strong sense of public-private work involved to achieve this shared ambition is important and will be critical to future success.
The industry has played a full and integral role in tackling COVID-19 and we are committed to working with all partners – including the German G7 Presidency – to ensure the world is better prepared against future pandemics.
Following the World Health Assembly’s decision this week to launch a process to seek a global agreement on pandemic prevention, preparedness and response and we also support working with governments, international organizations and other stakeholders to make sure that policies and plans are developed in a way that achieves the effective response the world requires to tackle any future pandemic.
The 100 Day Mission roadmap indicates that it is well underway towards completion. It should provide useful insights for the global pandemic preparedness process that will be negotiated over the coming years. Both processes should recognise that emerging pathogens must be shared on or before Day 0 of future pandemics, and that any delays due to conditions applied to such sharing must be avoided if we are to achieve the 100 days objectives.
As a global community, we applaud the speed with which scientists are currently sharing pathogens. Their efforts should not be restricted by unclear legislation or unnecessary paperwork.
From the very beginning of the pandemic the biopharmaceutical industry has worked in global partnerships with governments, health systems and across industry to deliver safe and effective COVID-19 vaccines and treatments.
We must now build on the successes of COVID-19, exploring new public-private partnerships to accelerate vaccine and drug discovery for priority pathogens and prepare for new pathogens.
COVID-19 has clearly demonstrated the importance of investment into R&D against future pathogens and COVID variants. These investments from across the R&D ecosystem – including large biopharmaceutical companies, small biotechs and academic researchers – is completely dependent on strong protection of intellectual property.
International Generic and Biosimilar Medicines Association [IGBA]
https://www.igbamedicines.org/
News
No new digest content identified.
International Alliance of Patients’ Organizations – IAPO [to 4 Dec 2021]
https://www.iapo.org.uk/news/topic/6
Press and media [Selected]
Joint Statement On Patient Solidarity Day 2021
London, December 3rd, 2021 – On Patient Solidarity Day, the International Alliance of Patients’ Organizations (IAPO), the International Pharmaceutical Federation (FIP), the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA) and the International Hospital…
Over 3000 patient advocates in Asia-Pacific come together calling for recovering together through compassion, insight and co-creation
On 16 and 17 November 2021, 3800 patient advocates, government and policy makers, industry representatives, healthcare professionals, academia representatives, researchers and media came together for the virtual 3rd Asia-Pacific Patients Congress (APPC 2021) to share their vision and lived…
PhRMA [to 4 Dec 2021]
http://www.phrma.org/
Latest News [Selected]
No new digest content identified.
Contents [click to move among sections]
:: Milestones – Perspectives
:: Coronavirus [COVID-19] – WHO
:: Emergencies
:: Organization Announcements
:: Journal Watch
Journal Watch
Vaccines and Global Health: The Week in Review continues its weekly scanning of key peer-reviewed journals to identify and cite articles, commentary and editorials, books reviews and other content supporting our focu-s on vaccine ethics and policy. Journal Watch is not intended to be exhaustive, but indicative of themes and issues the Center is actively tracking. We selectively provide full text of some editorial and comment articles that are specifically relevant to our work. Successful access to some of the links provided may require subscription or other access arrangement unique to the publisher.
If you would like to suggest other journal titles to include in this service, please contact David Curry at: david.r.curry@centerforvaccineethicsandpolicy.org
American Journal of Infection Control
December 2021 Volume 49Issue 12 p1457-1578
http://www.ajicjournal.org/current
Major Articles
Impact of In-Hospital infection with SARS-CoV-2 among Inpatients at a university hospital
Josni Tauffer, Thais Claudia Roma de Oliveira Konstantyner, Maria Claudia Stockler de Almeida, …
Suely Miyuki Yashiro, Antonia Maria de Oliveira Machado, Eduardo Alexandrino Medeiros
Published online: September 19, 2021
p1464-1468
American Journal of Infection Control
December 2021 Volume 49Issue 12 p1457-1578
http://www.ajicjournal.org/current
Brief Reports
COVID-19 vaccination coverage among hospital-based healthcare personnel reported through the Department of Health and Human Services Unified Hospital Data Surveillance System, United States, January 20, 2021-September 15, 2021
Hannah E. Reses, Emma S. Jones, Donald B. Richardson, Kristopher M. Cate, David W. Walker, Craig N. Shapiro
Published online: November 17, 2021
p1554-1557
American Journal of Preventive Medicine
December 2021 Volume 61 Issue 6 p771-928
https://www.ajpmonline.org/current
Research Articles
Human Papillomavirus Vaccination and Human Papillomavirus–Associated Cancer Rates Within Florida Counties
Stephanie A.S. Staras, Tianyao Huo, Sarah M. Rothbard,…Lindsay A. Thompson, Natalie L. Silver,
Elizabeth A. Shenkman
Published online: August 09, 2021
p812-820
American Journal of Tropical Medicine and Hygiene
Volume 105 (2021): Issue 6 (Dec 2021)
https://www.ajtmh.org/view/journals/tpmd/105/6/tpmd.105.issue-6.xml
Editorial
Open access
Tropical Infectious Diseases: Still Here, Still Raging, Still Killing
Peter J. Hotez
In our time of COVID-19, practically every public health agency—from local and state health departments, to the U.S. CDC, to the WHO—has concentrated its efforts on slowing SARS-2 coronavirus transmission. This occurred initially through nonpharmaceutical interventions and then, in the second year of the pandemic, through administering vaccinations. Despite these efforts, the consequences of the COVID-19 pandemic of 2019–2021 have been devastating. The most recent estimates from the Institute of Health Metrics and Evaluation at the University of Washington indicate that up to 6.5 million people will have lost their lives from COVID-19 by the end of 2021.1
Tragically, the deaths and disability-adjusted life years (DALYs) lost over this period will extend beyond the direct effects of SARS-2 coronavirus. For instance, in the United States and globally, the ensuing social disruptions slowed or even halted childhood vaccination programs.2,3 Although childhood vaccinations are rebounding as waves of the COVID-19 epidemic pass, one worry is that all of the antivaccine aggression now directed at COVID-19 vaccines may spill over to other programs. In such a case, we might not achieve pre-pandemic immunization levels for many months or even years; we might experience resurgence of measles and other vaccine-preventable infectious diseases.4
Another concern is the diversion of global health programs toward COVID-19 at the expense of tropical infectious diseases such as the neglected tropical diseases (NTDs) and malaria. Since 2000, with the start of Millennium Development Goals programs, there have been enormous strides made in disease burden reductions for these conditions. For the NTDs, donor support from the governments of the United States and United Kingdom and operational research from the Bill & Melinda Gates Foundation, together with an ecosystem of nongovernmental development organizations (NGDOs), health ministries, and the WHO, have contributed to enormous reductions in the prevalence of NTDs through mass drug administration and preventive treatments.5 This is also true for global infection control programs supported by the Global Fund to Fight AIDS, Tuberculosis and Malaria; the U.S. President’s Emergency Plan for AIDS Relief and Malaria Initiative; as well as efforts to unify these programs under the banner of universal health coverage.6
Two new papers in the American Journal of Tropical Medicine and Hygiene (AJTMH) examine the consequences of the COVID-19 pandemic and its adverse impact on global efforts to control tropical infectious diseases in Africa.7,8…
American Journal of Tropical Medicine and Hygiene
Volume 105 (2021): Issue 6 (Dec 2021)
https://www.ajtmh.org/view/journals/tpmd/105/6/tpmd.105.issue-6.xml
Perspective Pieces
The Value and Interpretation of Race and Ethnicity Data in the Era of Global Migration: A Change Is in Order
Serin Edwin Erayil, M. Kumi Smith, Tsige Gebreslasse, Patricia F. Walker, Erin M. Mann, Syreeta Wilkins, and William M. Stauffer
Volume/Issue: Volume 105: Issue 6
DOI: https://doi.org/10.4269/ajtmh.21-0665
Page(s): 1453–1455
American Journal of Tropical Medicine and Hygiene
Volume 105 (2021): Issue 6 (Dec 2021)
https://www.ajtmh.org/view/journals/tpmd/105/6/tpmd.105.issue-6.xml
Sex Workers Should Be Included in COVID-19 Vaccination Efforts
Gladson Vaghela, Catherine Hermoso, Attaullah Ahmadi, and Don Eliseo Lucero-Prisno III
Volume/Issue: Volume 105: Issue 6
DOI: https://doi.org/10.4269/ajtmh.21-0438
American Journal of Tropical Medicine and Hygiene
Volume 105 (2021): Issue 6 (Dec 2021)
https://www.ajtmh.org/view/journals/tpmd/105/6/tpmd.105.issue-6.xml
Articles
Restarting Neglected Tropical Diseases Programs in West Africa during the COVID-19 Pandemic: Lessons Learned and Best Practices
Achille Kabore, Stephanie L. Palmer, Ernest Mensah, Virginie Ettiegne-Traore, Rose Monteil, Franck Sintondji, Justin Tine, Daniel Tesfaye, Kisito Ogoussan, Diana Stukel, Brian B. Fuller, Katherine Sanchez, Bolivar Pou, Benoit Dembele, Angela Weaver, Steven Reid, Marie Denise Milord, Yao Kassankogno, Anders Seim, and Joseph Shott
Volume/Issue: Volume 105: Issue 6
DOI: https://doi.org/10.4269/ajtmh.21-0408
Page(s): 1476–1482
American Journal of Tropical Medicine and Hygiene
Volume 105 (2021): Issue 6 (Dec 2021)
https://www.ajtmh.org/view/journals/tpmd/105/6/tpmd.105.issue-6.xml
Open access
Relative Burdens of the COVID-19, Malaria, Tuberculosis, and HIV/AIDS Epidemics in Sub-Saharan Africa
David Bell and Kristian Schultz Hansen
Volume/Issue: Volume 105: Issue 6
DOI: https://doi.org/10.4269/ajtmh.21-0899
Page(s): 1510–1515
American Journal of Tropical Medicine and Hygiene
Volume 105 (2021): Issue 6 (Dec 2021)
https://www.ajtmh.org/view/journals/tpmd/105/6/tpmd.105.issue-6.xml
Assessing Vaccine Coverage and Timeliness in Bamako, Mali after the Introduction of Rotavirus Vaccine: A Modified Immunization Cluster Survey
Anna Roose, Uma Onwuchekwa, Milagritos Tapia, Samba Sow, T. Christopher Mast, and Karen Kotloff
Volume/Issue: Volume 105: Issue 6
DOI: https://doi.org/10.4269/ajtmh.21-0148
Page(s): 1594–1601