Organization Announcements
Paul G. Allen Frontiers Group [to 23 Jan 2021]
https://alleninstitute.org/what-we-do/frontiers-group/news-press/
News
No new digest content identified.
BARDA – U.S. Department of HHS [to 23 Jan 2021]
https://www.phe.gov/about/barda/Pages/default.aspx
BARDA News
No new digest content identified.
BMGF – Gates Foundation [to 23 Jan 2021]
http://www.gatesfoundation.org/Media-Center/Press-Releases
Press Releases and Statements
No new digest content identified.
Bill & Melinda Gates Medical Research Institute [to 23 Jan 2021]
https://www.gatesmri.org/
The Bill & Melinda Gates Medical Research Institute is a non-profit biotech organization. Our mission is to develop products to fight malaria, tuberculosis, and diarrheal diseases—three major causes of mortality, poverty, and inequality in developing countries. The world has unprecedented scientific tools at its disposal; now is the time to use them to save the lives of the world’s poorest people
No new digest content identified.
CARB-X [to 23 Jan 2021]
https://carb-x.org/
News
No new digest content identified.
Center for Vaccine Ethics and Policy – GE2P2 Global Foundation [to 23 Jan 2021]
https://centerforvaccineethicsandpolicy.net/
News/Analysis/Statements
No new digest content identified.
CEPI – Coalition for Epidemic Preparedness Innovations [to 23 Jan 2021]
http://cepi.net/
Latest News
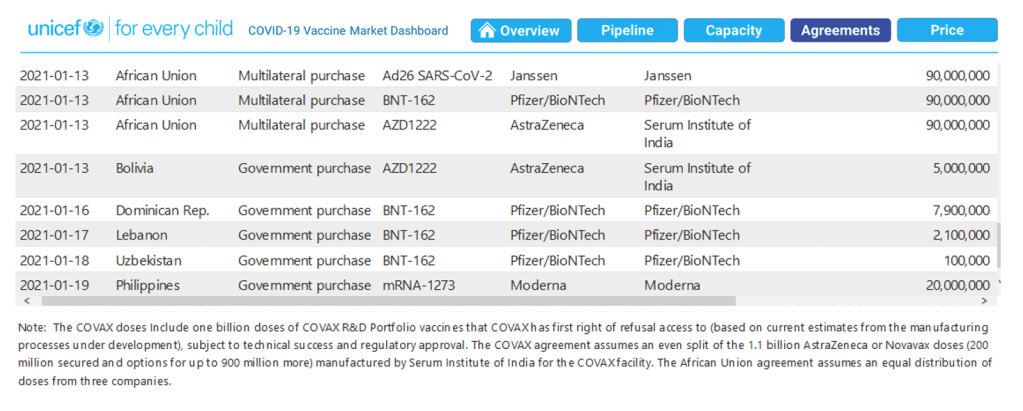
COVAX announces new agreement, plans for first deliveries
22 Jan 2021
[See Milestones above for detail]
Upcoming CEPI-CIHR grant funding opportunity for Canadian researchers
19 Jan 2021 By Jodie Rogers
CEPI and the Canadian Institutes of Health Research (CIHR) Institute of Infection and Immunity (III) will soon launch a new grant funding opportunity for researchers in Canada to pursue projects advancing CEPI’s work to expedite the development of vaccines against emerging infectious diseases, including COVID-19.
EDCTP [to 23 Jan 2021]
http://www.edctp.org/
The European & Developing Countries Clinical Trials Partnership (EDCTP) aims to accelerate the development of new or improved drugs, vaccines, microbicides and diagnostics against HIV/AIDS, tuberculosis and malaria as well as other poverty-related and neglected infectious diseases in sub-Saharan Africa, with a focus on phase II and III clinical trials
No new digest content identified.
Emory Vaccine Center [to 23 Jan 2021]
http://www.vaccines.emory.edu/
Vaccine Center News
No new digest content identified.
European Medicines Agency [to 23 Jan 2021]
http://www.ema.europa.eu/ema/
News & Press Releases
News: Extra dose from vials of Comirnaty COVID-19 vaccine (updated)
Last updated: 20/01/2021
EMA’s human medicines committee (CHMP) has recommended updating the product information for Comirnaty to clarify that each vial contains 6 doses of the vaccine…
News: Global regulators highlight key role of healthcare professionals in fostering confidence in COVID-19 vaccines
Last updated: 19/01/2021
EMA has endorsed a joint statement published today by the International Coalition of Medicines Regulatory Authorities (ICMRA) to inform and help healthcare professionals answer questions about the evaluation, approval and monitoring of safe, effective and high-quality COVID-19 vaccines…
[See COVID above for detail]
European Vaccine Initiative [to 23 Jan 2021]
http://www.euvaccine.eu/
Latest News
No new digest content identified.
FDA [to 23 Jan 2021]
https://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/default.htm
Press Announcements /Selected Details
January 22, 2021 – Coronavirus (COVID-19) Update: January 22, 2021
:: The FDA’s Office of Criminal Investigations recently investigated a case that has led to an arrest and the filing of a criminal complaint by the U.S. Department of Justice for introducing misbranded drugs into interstate commerce. The criminal investigation found that in a variety of online postings from as early as March 2020, the defendant, Johnny T. Stine, claimed to have a COVID-19 vaccine that he offered to inject in customers for $400-$1000 each. Stine’s company, North Coast Biologics, had previously received a warning letter from the agency for promoting an unapproved COVID-19 vaccine product.
“Unproven injectable vaccines purported to prevent or treat COVID-19, made from unknown substances under unknown conditions, present significant health risks in and of themselves. They also can lead consumers to make lifestyle choices that increase their actual risk of infection with COVID-19, or to delay or stop appropriate medical treatment,” said Special Agent in Charge Lisa L. Malinowski, FDA Office of Criminal Investigations Los Angeles Field Office. “The FDA will continue to investigate fraudulent COVID-19 treatments and bring to justice those who try to profit from the pandemic by offering unproven and illegally marketed coronavirus products.”
January 21, 2021 – FDA Approves First Extended-Release, Injectable Drug Regimen for Adults Living with HIV
January 19, 2021 – Coronavirus (COVID-19) Update: January 19, 2021
:: The FDA updated its “Investigational COVID-19 Convalescent Plasma” guidance and corresponding webpage. The revisions provide recommendations regarding when individuals who have received an investigational COVID-19 vaccine as a participant in a clinical trial, or received an authorized or licensed COVID-19 vaccine, qualify as convalescent plasma donors. In addition, the agency has extended the period of enforcement discretion described in the guidance.
FDA – COVID-19 Vaccines [to 23 Jan 2021]
www.fda.gov/covid19vaccines
News and Updates; Upcoming Events
No new digest content identified.
Fondation Merieux [to 23 Jan 2021]
http://www.fondation-merieux.org/
News, Events
No new digest content identified.
Gavi [to 23 Jan 2021]
https://www.gavi.org/
News releases
COVAX announces new agreement, plans for first deliveries
22 Jan 2021
[See Milestones above for detail]
GHIT Fund [to 23 Jan 2021]
https://www.ghitfund.org/newsroom/press
GHIT was set up in 212 with the aim of developing new tools to tackle infectious diseases that
Press Releases
No new digest content identified.
Global Fund [to 23 Jan 2021]
https://www.theglobalfund.org/en/news/
News
No new digest content identified.
Global Research Collaboration for Infectious Disease Preparedness [GloPID-R] [to 23 Jan 2021]
https://www.glopid-r.org/news/
News
No new digest content identified.
Hilleman Laboratories [to 23 Jan 2021]
http://www.hillemanlabs.org/
No new digest content identified.
Human Vaccines Project [to 23 Jan 2021]
http://www.humanvaccinesproject.org/media/press-releases/
Press Releases
HVP COVID Report
Issue 25: Can President Biden’s Team Speed Vaccine Delivery?
Jan 22, 2021
By Michael Osterholm, Ph.D.
Professor, University of Minnesota and Director of the Center for Infectious Disease Research and Policy
IAVI [to 23 Jan 2021]
https://www.iavi.org/newsroom
PRESS RELEASES/FEATURES
January 19, 2021
IAVI at HIVR4P Virtual 2021
IAVI to participate in major HIV prevention research conference beginning January 27
January 13, 2021
Researchers demonstrate protection offered by novel TB vaccine candidate in animal model
:: A study of MTBVAC efficacy in a macaque model of tuberculosis shows that the vaccine candidate protects against aerosol infection with Mycobacterium tuberculosis.
:: Researchers from Europe and the U.S. demonstrate that the MTBVAC vaccine protects better than the current BCG vaccine in a model of tuberculosis in macaques and compare the immunological patterns to those conferred by MTBVAC in clinical trials.
International Coalition of Medicines Regulatory Authorities [ICMRA]
http://www.icmra.info/drupal/en/news
Selected Statements, Press Releases, Research
No new digest content identified.
International Generic and Biosimilar Medicines Association [IGBA]
https://www.igbamedicines.org/
News
Press Releases/Announcements
No new digest content identified.
IFFIm
http://www.iffim.org/
Press Releases/Announcements
No new digest content identified.
IFRC [to 23 Jan 2021]
http://media.ifrc.org/ifrc/news/press-releases/
Selected Press Releases, Announcements
No new digest content identified.
Institut Pasteur [to 23 Jan 2021]
https://www.pasteur.fr/en/press-area
Press release 19.01.2021
COVID-19: a lentiviral vaccine candidate for intranasal administration provides sterilizing protection in animal models
Scientists from the Institut Pasteur-TheraVectys Joint Laboratory recently published in Cell Host & Microbe1 the results of tests, in two preclinical models, of a COVID-19 vaccine candidate using a lentiviral vector administered by nasal route. Their study demonstrates that the vaccine candidate induces antibody response with strong neutralizing activity against SARS-CoV-2, as well as substantial cell-mediated responses. Intranasal vaccination with this vector drastically reduces lung viral loads and prevents pathogenic pulmonary inflammation. This research proves the high vaccine efficacy and the fact that its intranasal administration attracts the effectors from the protective immune response into the upper respiratory tract through which the virus enters the body.
IRC International Rescue Committee [to 23 Jan 2021]
http://www.rescue.org/press-release-index
Media highlights [Selected]
Press Release
New IRC report: Nine ways Biden can help 235 million of world’s most vulnerable, restoring “soul of America,” and advancing US interests
January 21, 2021
Press Release
The IRC applauds the Biden Administration’s measures on Day One to restore US humanity and leadership; urges action on refugee resettlement
January 20, 2021
IVAC [to 23 Jan 2021]
https://www.jhsph.edu/research/centers-and-institutes/ivac/index.html
Updates; Events
No new digest content identified.
IVI [to 23 Jan 2021]
http://www.ivi.int/
Selected IVI News, Announcements, Events
No new digest content identified.
JEE Alliance [to 23 Jan 2021]
https://www.jeealliance.org/
Selected News and Events
No new digest content identified.
Johns Hopkins Center for Health Security [to 23 Jan 2021]
https://www.centerforhealthsecurity.org/news/center-news/
Center News
No new digest content identified.
MSF/Médecins Sans Frontières [to 23 Jan 2021]
http://www.msf.org/
Latest [Selected Announcements]
Rohingya refugee crisis
Relocations, reduced services leave Rohingya communities at breaking point in Bangladesh
Project Update 21 Jan 2021
Brazil
COVID-19 leaves Amazonas health system saturated, overloaded and struggling
Project Update 21 Jan 2021
South Sudan
Falling international funding exposes communities in Jonglei to a lack of healthcare
Project Update 19 Jan 2021
Yemen
Yemen: “These sanctions have to make clear that they do not apply to humanitarian aid”
Interview 18 Jan 2021
National Vaccine Program Office – U.S. HHS [to 23 Jan 2021]
https://www.hhs.gov/vaccines/about/index.html
Upcoming Meetings/Latest Updates
No new digest content identified.
NIH [to 23 Jan 2021]
http://www.nih.gov/news-events/news-releases
News Releases
Full-dose blood thinners decreased need for life support and improved outcome in hospitalized COVID-19 patients
January 22, 2021 — Full doses of blood thinners, in addition to being safe, were superior to the doses normally given to prevent blood clots in hospitalized patients.
PATH [to 23 Jan 2021]
https://www.path.org/media-center/
Press Release
No new digest content identified
Sabin Vaccine Institute [to 23 Jan 2021]
http://www.sabin.org/updates/pressreleases
Statements and Press Releases
No new digest content identified.
UNAIDS [to 23 Jan 2021]
http://www.unaids.org/en
Selected Press Releases/Reports/Statements
21 January 2021
Remembering Andrew Mosane, AIDS activist and progressive “radical”
20 January 2021
Challenge the stigma, pursue your right to health
19 January 2021
The road to PrEP in Guyana
19 January 2021
Modelling shows the value of favourable societal environments
UNHCR Office of the United Nations High Commissioner for Refugees [to 23 Jan 2021]
http://www.unhcr.org/en-us/media-centre.htmlS
Selected Announcements
No new digest content identified.
UNICEF [to 23 Jan 2021]
https://www.unicef.org/media/press-releases
Selected Press releases, Statements
Press release
01/22/2021
COVAX announces new agreement, plans for first deliveries
[See Milestones above for detail]
Statement
01/22/2021
Remarks by Henrietta Fore, UNICEF Executive Director, at press briefing on COVAX
As delivered
[See Milestones above for detail]
Press release
01/20/2021
UN agencies warn economic impact of COVID-19 and worsening inequalities will fuel malnutrition for billions in Asia and the Pacific — FAO-UNICEF-WFP-WHO
Child and maternal diets particularly vulnerable
Unitaid [to 23 Jan 2021]
https://unitaid.org/
Featured News
22 January 2021
Cost of rapid COVID-19 tests halved as global investment ensures availability of high volumes for low- and middle-income countries
:: Over 250 million antigen rapid tests (Ag RDTs) every year to be made available for low- and middle-income countries for a unit price of less than US$2.50, thanks to set of new agreements, the first of which with Premier Medical Corporation (PMC) of India who will provide up to 120 million tests
:: Agreements follow an open call for Expressions of Interest (EOI), launched last year by FIND and Unitaid on behalf of the Access to COVID-19 Tools (ACT) Accelerator, to drive equitable access to fit-for-purpose Ag RDTs for COVID-19
:: Further announcements focused on technology transfer and scale up of regional manufacturing capacity expected in the coming weeks
Vaccination Acceptance Research Network (VARN) [to 23 Jan 2021]
https://vaccineacceptance.org/news.html#header1-2r
Announcements
No new digest content identified.
Vaccine Confidence Project [to 23 Jan 2021]
http://www.vaccineconfidence.org/
News, Research and Reports
No new digest content identified.
Vaccine Education Center – Children’s Hospital of Philadelphia [to 23 Jan 2021]
http://www.chop.edu/centers-programs/vaccine-education-center
News
No new digest content identified.
Wellcome Trust [to 23 Jan 2021]
https://wellcome.ac.uk/news
News
Explainer | 22 January 2021
When will the world be vaccinated against Covid-19?
…Current estimates are that it will probably take well into 2023-24 for everyone who needs a vaccine to receive one(opens in a new tab)….
Explainer | 22 January 2021
Principles and guidelines for reporting on Covid-19 vaccines
Explainer | 22 January 2021
What are ‘adverse events’ and ’emergency use authorisation’ in relation to vaccination?
Explainer | 21 January 2021
How have Covid-19 vaccines been made quickly and safely?
The Wistar Institute [to 23 Jan 2021]
https://www.wistar.org/news/press-releases
Press Releases
Jan. 21, 2021
Wistar Scientists Make Pivotal Discovery on the Mechanism of Epstein-Barr Virus Latent Infection
A previously unknown enzymatic function of the EBNA1 viral protein might instruct new approaches for EBV-associated cancer.
WFPHA: World Federation of Public Health Associations [to 23 Jan 2021]
https://www.wfpha.org/
Latest News
WFPHA Welcomes the Entry into Force of the Treaty on the Prohibition of Nuclear Weapons
Jan 21, 2021 | News
WFPHA at EB148: Statement on Oral Health
Jan 20, 2021 | News
WFPHA at EB148: Statement on COVID-19 Response
Jan 19, 2021 | News
“…To protect people’s lives, an effective and safe COVID-19 immunization should be considered a global public good, and vaccines should be produced at larger volumes, distributed equitably between countries, and provided at no cost to the user. Furthermore, without an equitable distribution of COVID-19 vaccines between countries, the world economy will not recover, causing greater human suffering and premature mortality.
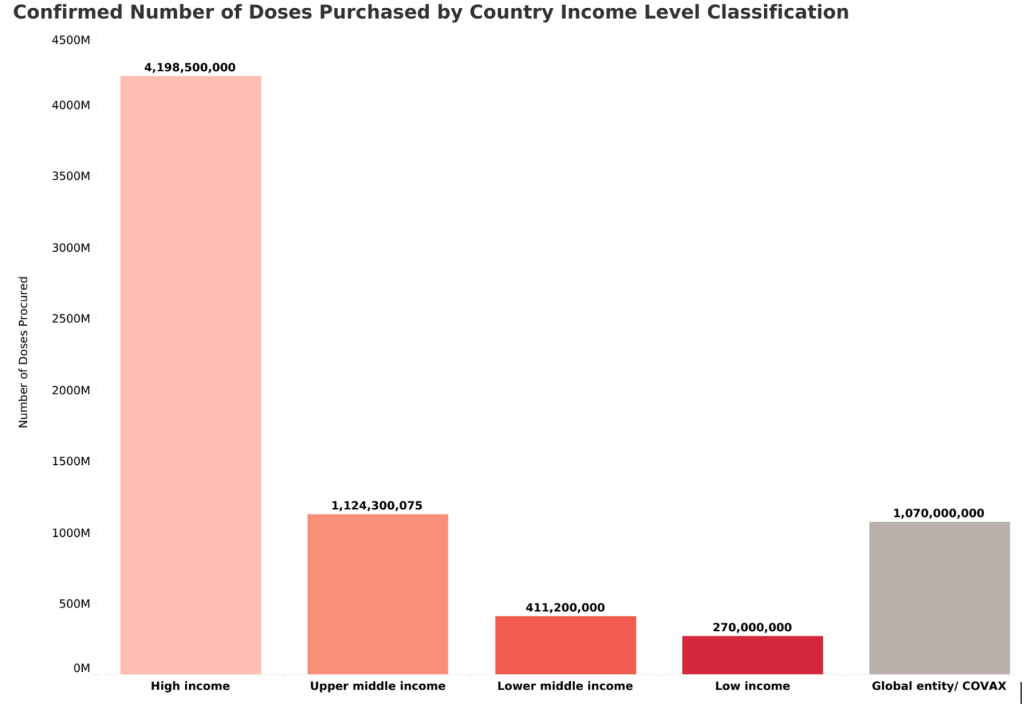
Unless governments and the pharmaceutical industry take urgent action to ensure that enough COVID-19 vaccine doses are produced, 92 low- and middle-income countries will be able to vaccinate against COVID-19 only one in five people in 2021. On the other hand, high-income countries, which account for just 14% of the world’s population, have already purchased 48% of the 8.6 billion doses of the most promising vaccines.
This can be achieved by waiving intellectual property rights to vaccines, tests, and treatments related to COVID-19, openly sharing the vaccines’ technology and intellectual property through the WHO, fully funding COVAX, stopping bilateral deals, and investing in WHO efforts and in the strengthening of national health systems…”
World Organisation for Animal Health (OIE) [to 23 Jan 2021]
https://www.oie.int/en/for-the-media/press-releases/2021/
Press Releases
Launch of the multi-year, multi-partner Global Burden of Animal Diseases programme
Animal health leaders and researchers from the Global Burden of Animal Diseases (GBADs) programme have secured US$7 million from the Bill & Melinda Gates Foundation and UK’s Foreign, Commonwealth and Development Office, to rollout a framework on measuring animal health burdens and their impacts on human lives and economies. The information provided by GBADs will guide public policy and private sector strategy, contributing to improve animal health and welfare more effectively. It will also be a basis for further academic research.
19 January 2021
[See Milestones above for detail]
::::::
ARM [Alliance for Regenerative Medicine] [to 23 Jan 2021]
Press Releases – Alliance for Regenerative Medicine (alliancerm.org)
Press Releases
No new digest content identified.
BIO [to 23 Jan 2021]
https://www.bio.org/press-releases
Press Releases
BIO Welcomes U.S. Involvement in COVAX, ACT Accelerator Programs
January 21, 2021
The Biden administration just announced its intent for the United States to join the COVAX vaccine facility, which aims to deliver COVID-19 vaccines to poor and under-developed countries… “As the organization representing U.S. biotechnology companies who are leading the world in the of development COVID vaccines and therapeutics, BIO welcomes Dr. Anthony Fauci’s statement today that the United States will be joining the global COVAX and ACT Accelerator programs, which are the chief international efforts charged with getting COVID treatments to patients around the world…:
Biotechnology’s Leading Trade Group Restructures for the Future
January 19, 2021
Washington, DC – The Biotechnology Innovation Organization (BIO), which announced its transition to a new CEO in June to Dr. Michelle McMurry-Heath, is announcing a new leadership team and organizational structure that promises to provide long-term stability for the organization’s ambitious agenda. Some staff reductions were made to align the organization for the new strategic direction…
BIO Applauds Incoming Administration’s Decision Elevating Key White House Science Post to Cabinet-level Agency
January 16, 2021
DCVMN – Developing Country Vaccine Manufacturers Network [to 23 Jan 2021]
http://www.dcvmn.org/
News; Upcoming events
No new digest content identified.
ICBA – International Council of Biotechnology Associations [to 23 Jan 2021]
https://internationalbiotech.org/news/
News
No new digest content identified.
IFPMA [to 23 Jan 2021]
http://www.ifpma.org/resources/news-releases/
Selected Press Releases, Statements, Publications
Biopharmaceutical global body kicks off 2021 with new CEO leadership line up
21 January 2021
The biopharmaceutical global industry, represented by the International Federation of Pharmaceutical Manufacturers and Associations (IFPMA), is pleased to announce that Jean-Christophe Tellier, UCB Chief Executive Officer & Chairman of the Executive Committee has assumed the role of IFPMA President. Jean-Christophe Tellier succeeds David A. Ricks, Chairman and Chief Executive Officer, Eli Lilly and Company. Albert Bourla, Chairman and CEO of Pfizer, has been appointed IFPMA Vice President and will together with Isao Teshirogi, President and CEO of Shionogi, complement the IFPMA CEO leadership team. They will serve as the Chair and Vice Chairs of the IFPMA CEO Steering Committee…
IFPMA, GSCF, ICBA Joint Statement on the item 14.2 Strengthening preparedness for health emergencies: implementation
20 January 2021
“…The COVID-19 pandemic has highlighted the importance of timely pathogen sharing in the context of public health emergencies. The criticality of fast, timely and unrestricted access to pathogen information in allowing the development of medical countermeasures and understanding the genomic epidemiology of the virus has been recognised by both the Committee’s interim report, under paragraph 42, and the Director-General’s report on the ‘Public Health Implications of Implementation of the Nagoya Protocol’, under paragraph 3.
Though the preamble to the Nagoya Protocol recognises the IHR and “the importance of ensuring access to human pathogens for public health preparedness and response purposes”, currently, the global ability to share pathogen samples and their related data in a timely manner is being impacted due to the inefficient and bureaucratically burdensome bilateral system envisaged under the same Protocol, most notably for seasonal influenza viruses. Navigating a system in which each country has different access requirements that must be negotiated bilaterally is simply not feasible when dealing with the emergence of a novel pathogen.
In light of this, IFPMA fully supports the Committee’s approach of “conducting an article-by-article analysis” and “considering how the Regulations could facilitate the rapid sharing of scientific findings and samples within the global scientific community under Article 6.”
IFPMA Statement for EB148 agenda item 14.1 Covid-19 Response
19 January 2021
“We share the deep sense of responsibility that together we have to end this pandemic and are proud to be a founding member of the ACT A. We are keenly aware that science and innovation are only part of the equation. As part of our responsibility towards society, we must also ensure equitable global access to safe, high quality, effective, and affordable COVID-19 vaccines and therapeutics. The biopharmaceutical industry has shown its openness to new approaches and numerous collaborations with all stakeholders to respond to the urgent needs of the pandemic. This, however, should not come at the cost of diluting quality and safety of vaccines and medicines, nor should it impact key incentives that foster innovation, namely intellectual property rights. The existing IP framework has enabled the pharmaceutical industry to respond swiftly to the global crisis and develop several new vaccines with remarkable speed, by engaging in new collaborations to leverage both new approaches and existing molecules. This scientific excellence would not have been possible without the important incentives such as those provided by IP rights. Indeed, our expertise and risk-taking ability has allowed us to roll out several new vaccines in less than a year…”
PhRMA [to 23 Jan 2021]
http://www.phrma.org/
Selected Press Releases, Statements
Research shows gene therapies can offer cost savings potential of up to $1.8M over five years for patients with certain blood diseases
January 15, 2021
A recent analysis by Health Advances looks at two particularly burdensome hematological (or blood) diseases — hemophilia A and beta thalassemia — and finds that potential gene therapies in the pipeline could reduce per patient costs by as much as 41% and 90%, respectively, over five years. Research-based knowledge gained in recent years about the genetic foundation for these diseases has facilitated the pursuit of several promising gene therapy approaches.
Blog Post